A review of covalent organic framework electrode materials for rechargeable metal-ion batteries
-
摘要: 共价有机框架具有强健的骨架、丰富的电化学活性位点、便于金属离子传输的可控孔道以及利于优化电化学性能的可调控的分子结构,因此是理想的下一代可充电金属离子电池电极材料。此外,共价有机框架电极材料没有传统无机电极材料价格昂贵及含有毒金属的问题,也不存在有机小分子循环稳定性差的问题,在下一代可充电金属离子电池中具有巨大的应用潜力。因此,本文总结了共价有机框架电极材料的电化学活性位点,并着重讨论了通过调节共价有机框架的骨架结构、孔道、活性位点和电子结构提高共价有机框架电极材料电化学性能(包括:能量密度、倍率性能和循环寿命)的策略。为了开发高性能的共价有机框架电极材料,未来的工作需着重于优化它们的离子和电子导电性,进一步提高它们的工作电压以及探明它们的储能机制。本文将有助于开发用于下一代金属离子电池的高性能共价有机框架电极材料。Abstract: Covalent organic frameworks (COFs) are highly promising electrode materials for next-generation rechargeable metal-ion batteries owing to their robust framework, abundant electrochemically active sites, well-defined and tunable pores and channels for metal ion transfer, and adjustable molecular structures for improving electrochemical performance. Moreover, COFs do not have the problems caused by expensive or toxic elements in conventional inorganic electrode materials or the cycling stability challenges existing in small organic molecules, and thus have great potential as electrode materials in next-generation rechargeable metal-ion batteries. We summarize the electrochemically active sites of these materials for charge storage, and most importantly, we focus on strategies for improving their electrochemical performance, including energy density, rate performance and cycling life by changing their frameworks, pores, active sites, and electronic structures. To fabricate high performance COF electrodes, much more effort is needed to improve their ionic and electronic conductivities, increase their operating voltage, and reveal their mechanisms of energy storage. This review may shed light on developing high performance COF electrode materials for next-generation rechargeable metal-ion batteries.
-
Key words:
- Covalent organic frameworks /
- Rechargeable metal-ion batteries /
- Energy density /
- Rate performance /
- Cyclic lifespan
-
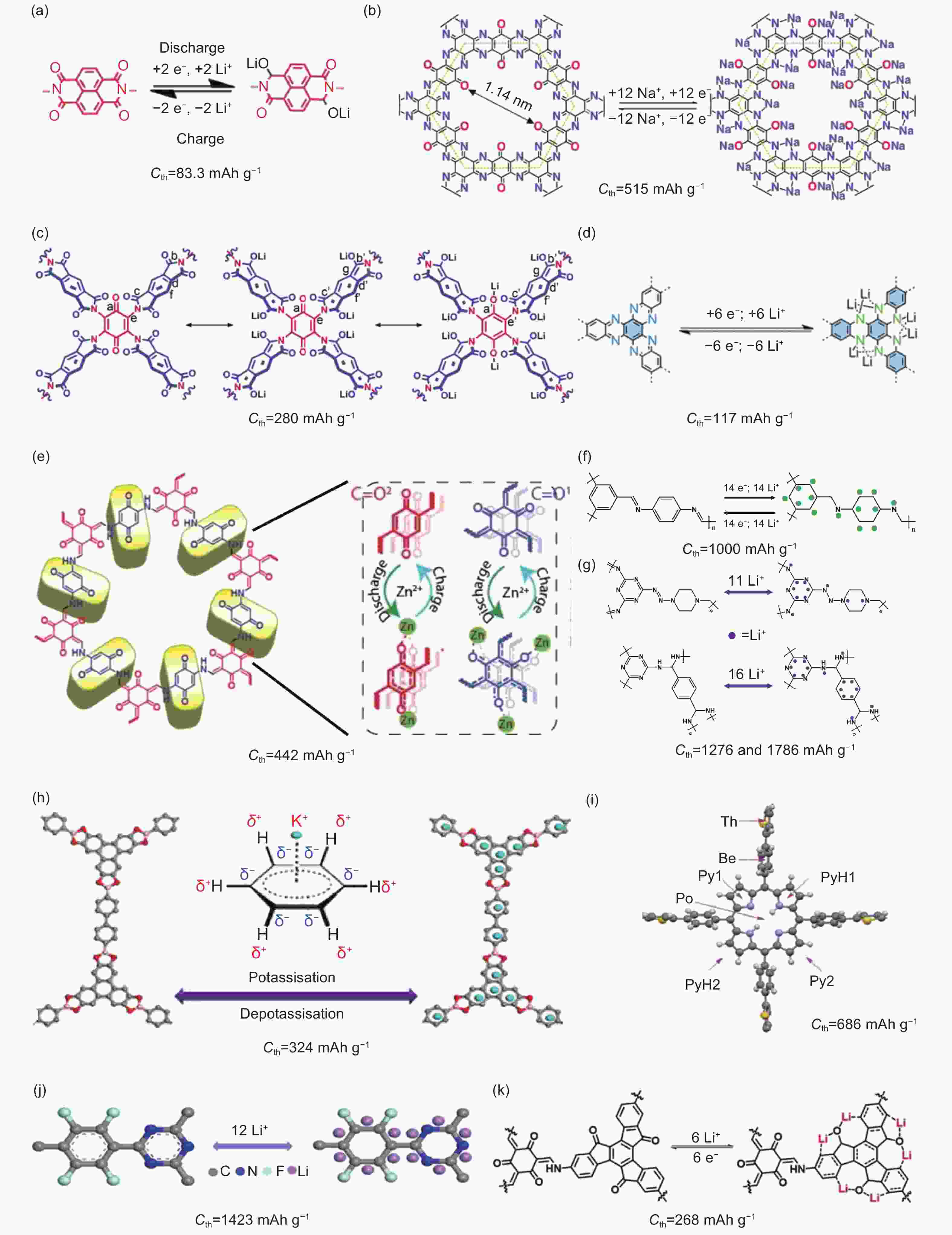
Figure 1. Various electrochemical active sites in reported COFs and corresponding theoretical specific capacity (Cth)[52-62]. (a, b, f) reproduced by permission of Nature Publishing Group, (c, d, g, k) reproduced by permission of Wiley-VCH, (e) reproduced by permission of the Royal Society of Chemistry and (h, i, j) reproduced by permission of American Chemical Society.
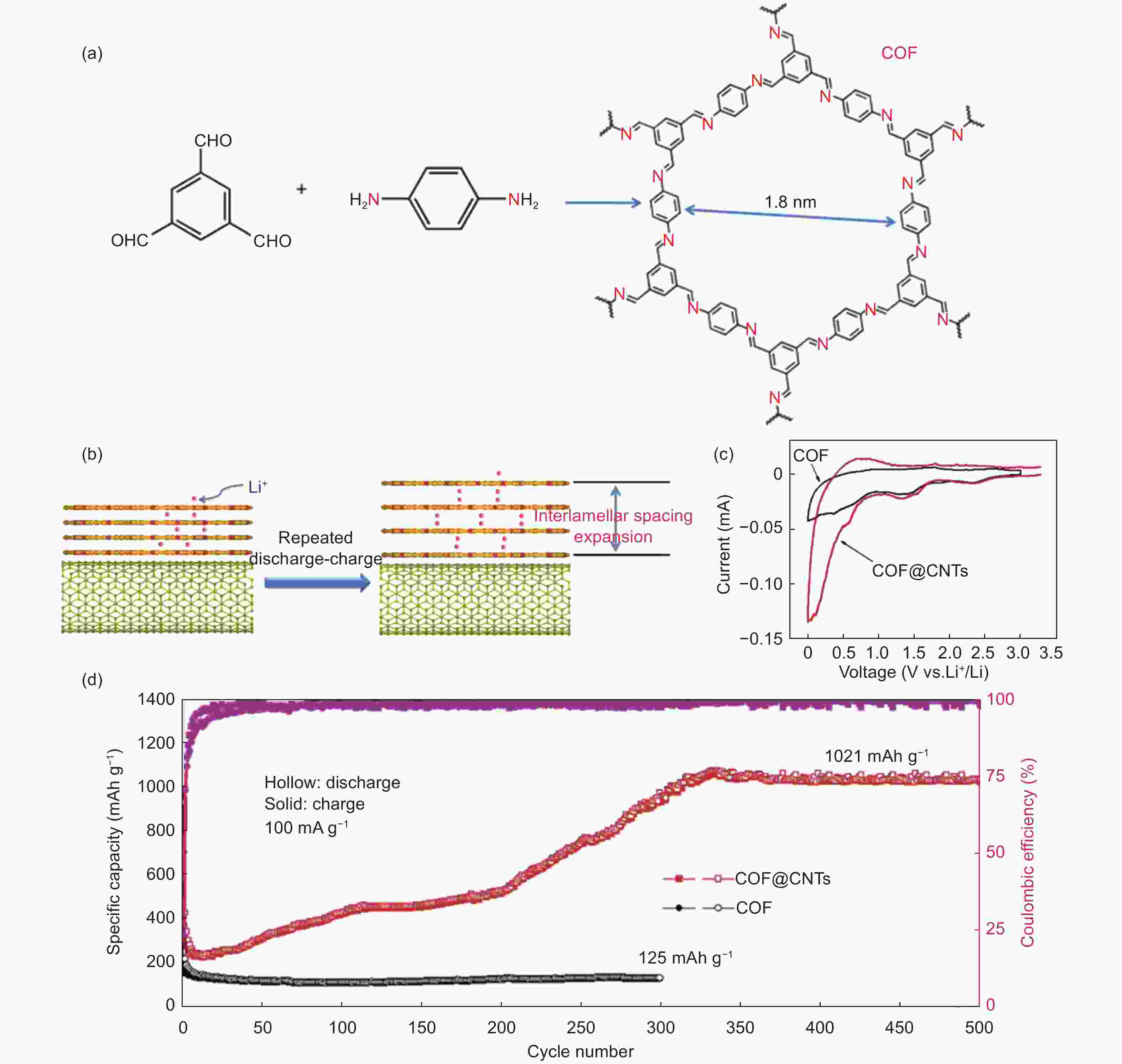
Figure 2. The composite COF@CNT electrode[57]: (a) structure of the COF, (b) interlayer spacing expansion, (c) the cyclic voltammetry curves and (d) the electrochemical performance of the composite electrode (Reproduced by permission of Nature Publishing Group).
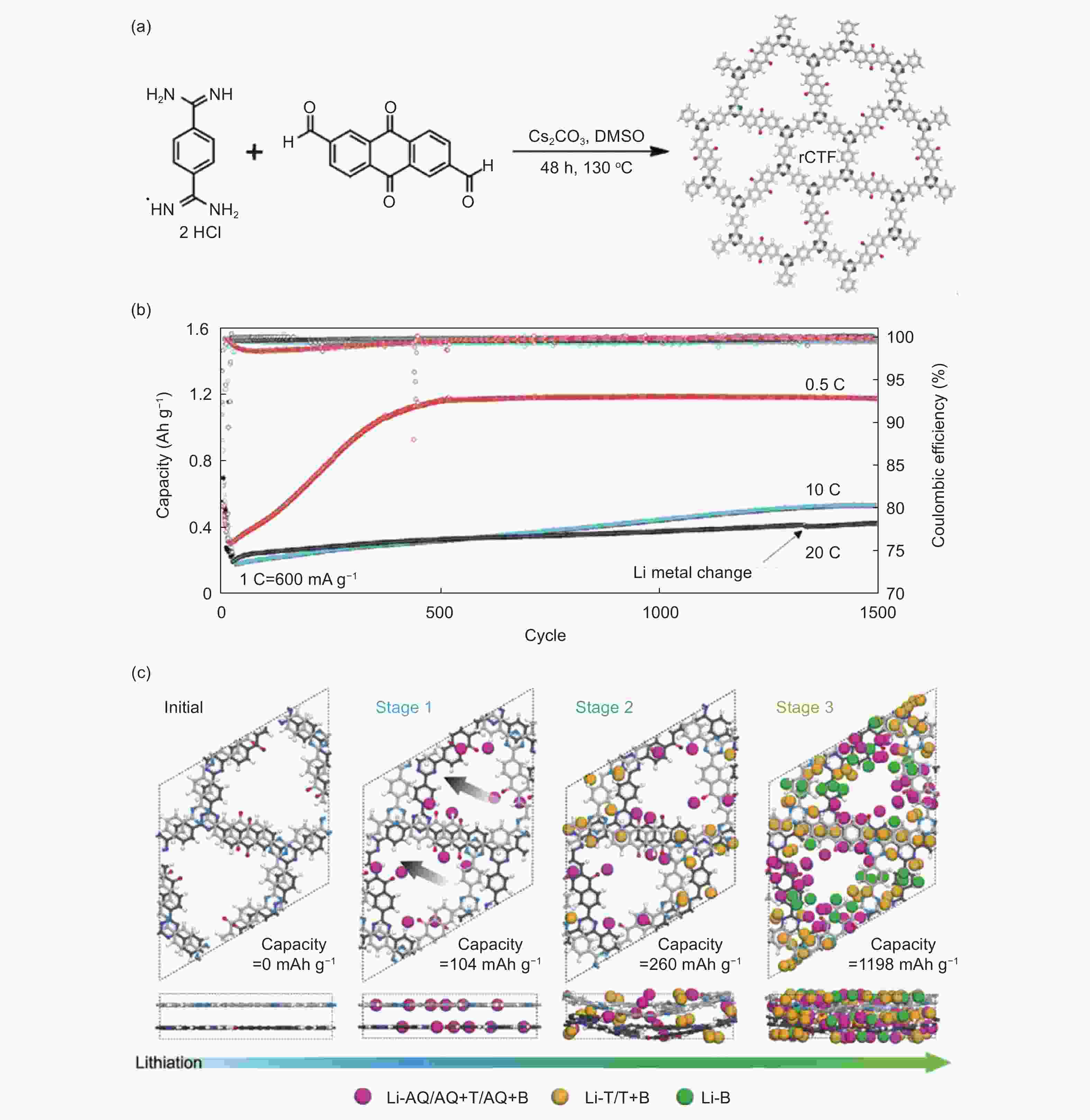
Figure 3. (a) The structure and synthetic method for rCTF, (b) the electrochemical performance of rCTF and (c) the simulated lithium storage mechanism[69] (Reproduced by permission of Wiley-VCH).
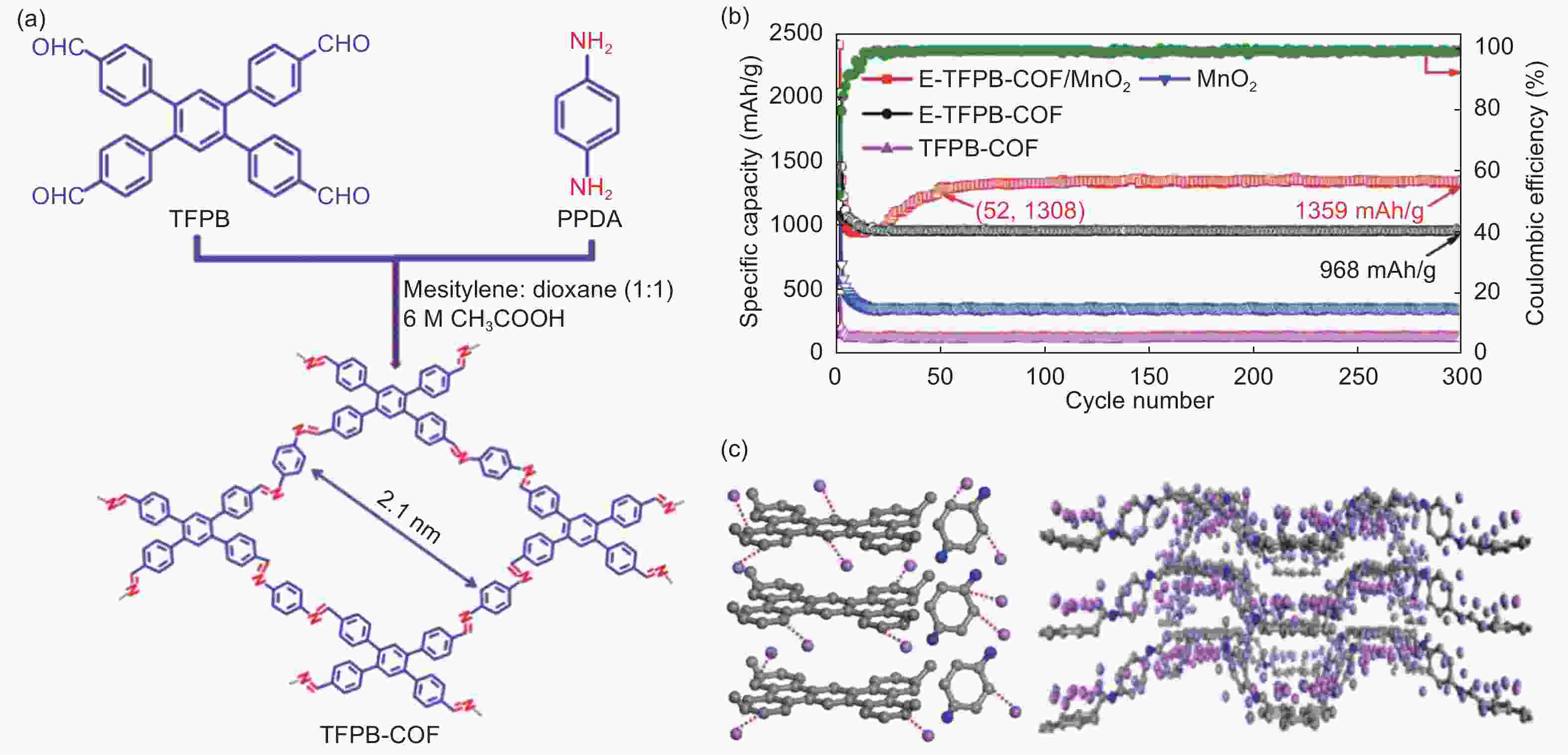
Figure 4. (a) The synthetic route of TFPB-COF, (b) the electrochemical performance of TFPB-COF and (c) the lithium storage mechanism[88] (Reproduced by permission of Wiley-VCH).
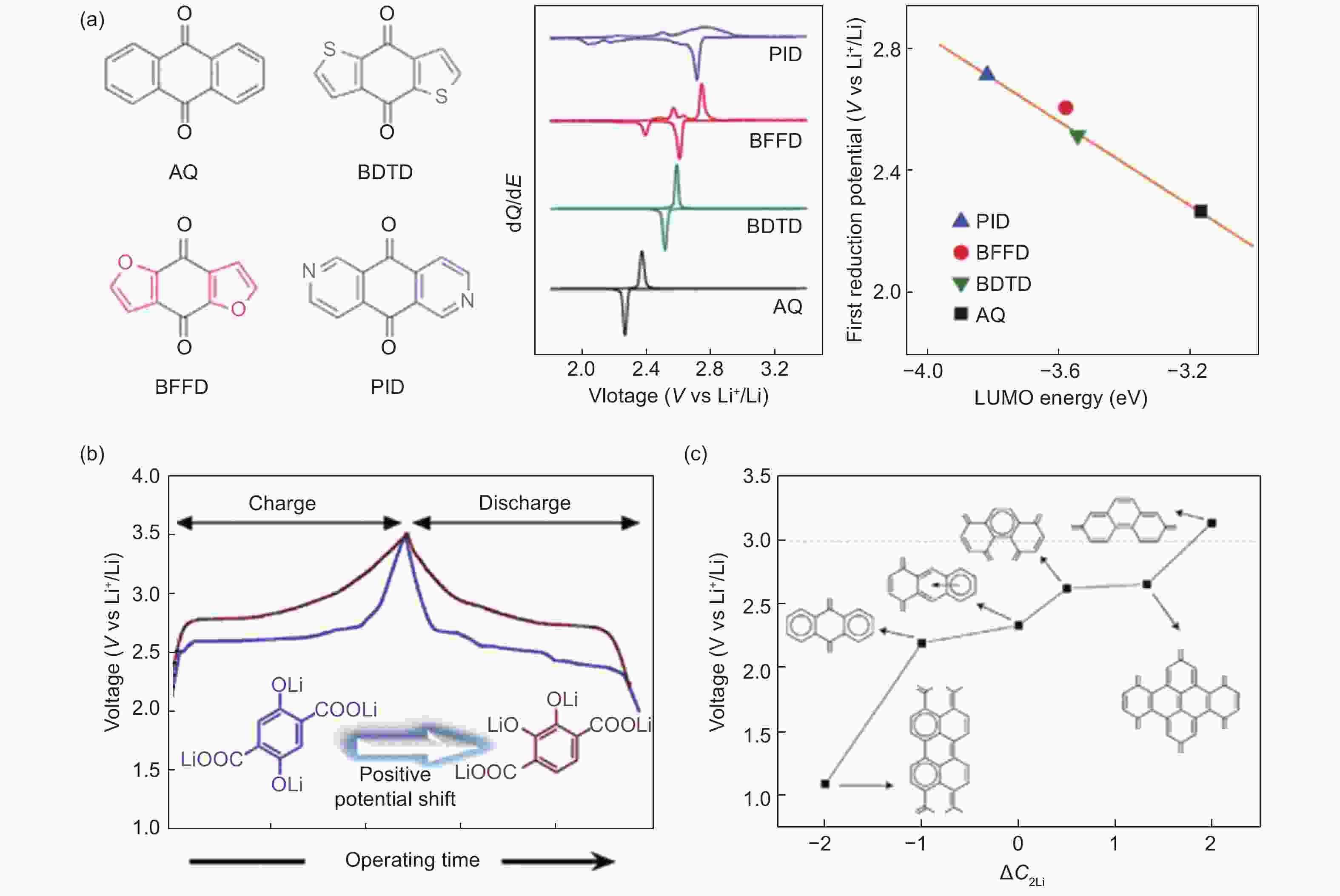
Figure 5. The relationship between voltage and molecular structure: (a) small molecules as the cathode in a LIB: molecular structures, differential capacity curves and correlation between the first reduction potentials and the calculated LUMO energies[94], (b) the effect of active sites position[95] and (c) conjugated structure[96] on voltage ((a) Reproduced by permission of Wiley-VCH, (b) Reproduced by permission of American Chemical Society and (c) Reproduced by permission of the Royal Society of Chemistry.
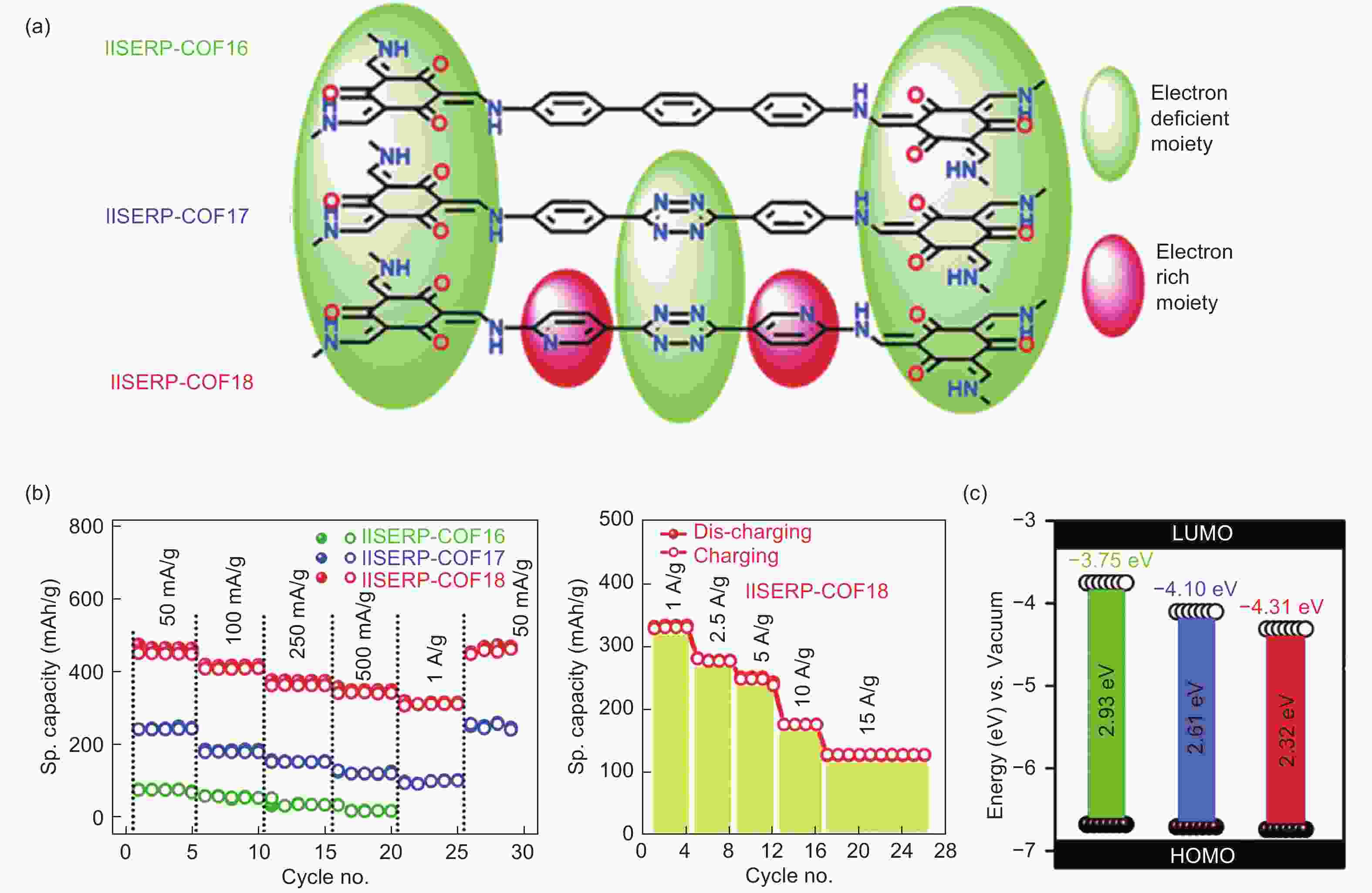
Figure 6. (a) Different molecular configurations, (b) the electrochemical performance of three COFs, (c) the molecular orbital energy level of three different COFs[79] (Reproduced by permission of the Royal Society of Chemistry).
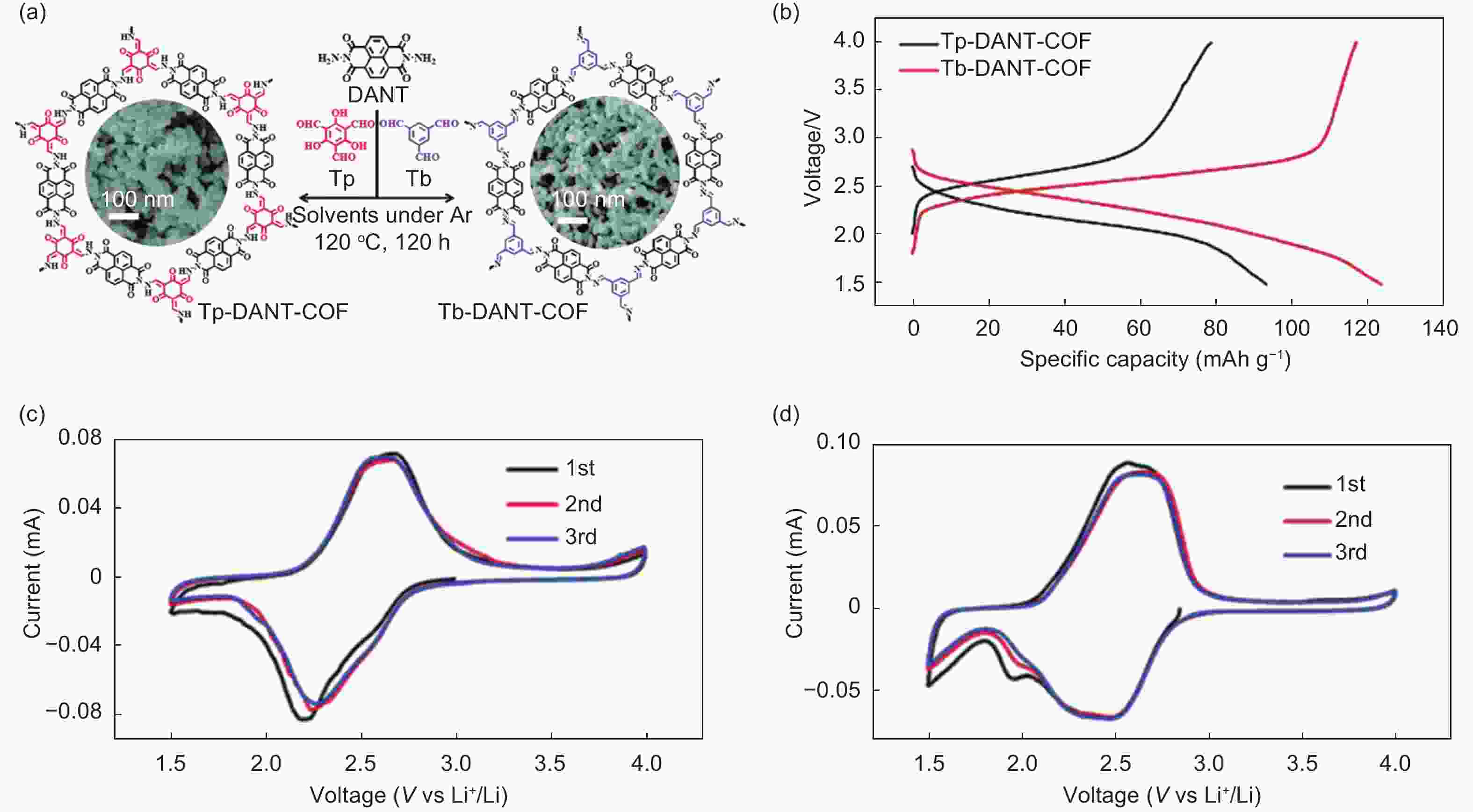
Figure 7. (a) The microstructures of two COFs, (b) the specific capacity, the cyclic voltammetry curves for (c) Tp-DANT-COF and (d)Tb-DANT-COF[64] (Reproduced by permission of the Royal Society of Chemistry).
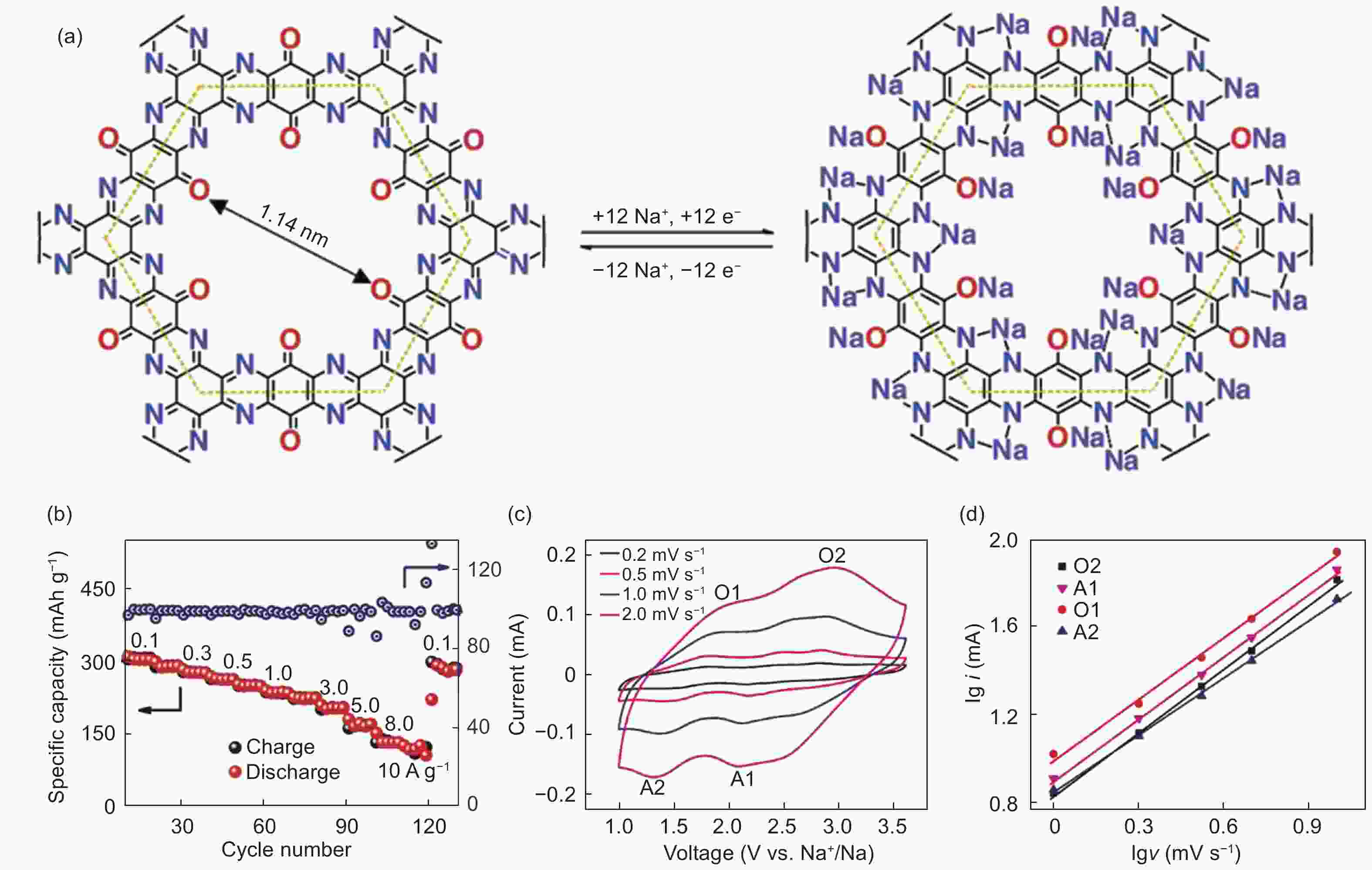
Figure 8. (a) The structure of a nitrogen-rich COF, (b) the rate performance, (c) the cyclic voltammetry curves for the nitrogen-rich COF and (d) lgi-lgv curves for the b evaluation[53] (Reproduced by permission of Nature Publishing Group).
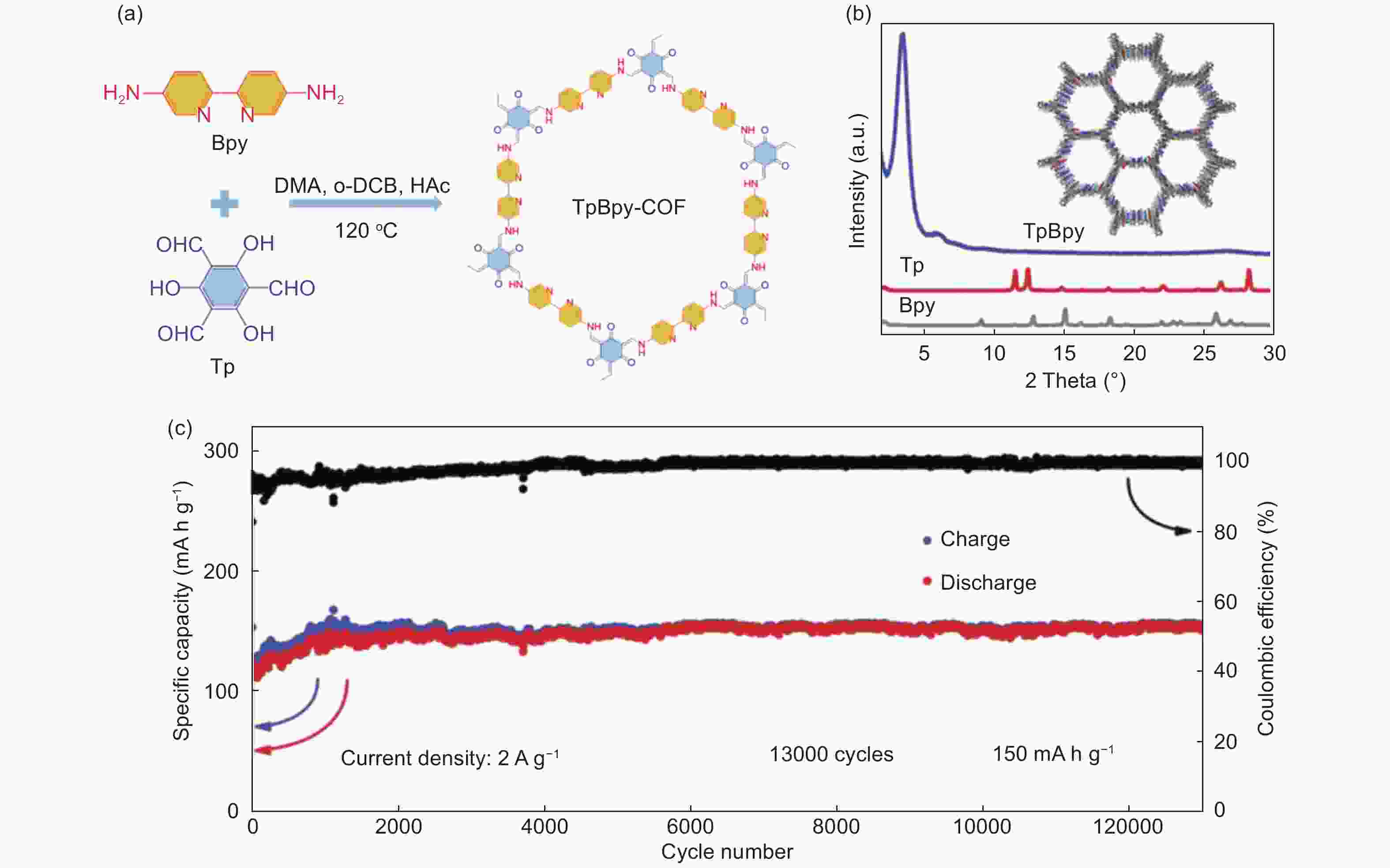
Figure 9. (a) The synthetic route of TpBpy-COF, (b) the XRD pattern of TpBpy-COF and (c) cyclic performance of TpBpy-COF[84] (Reproduced by permission of Wiley-VCH).
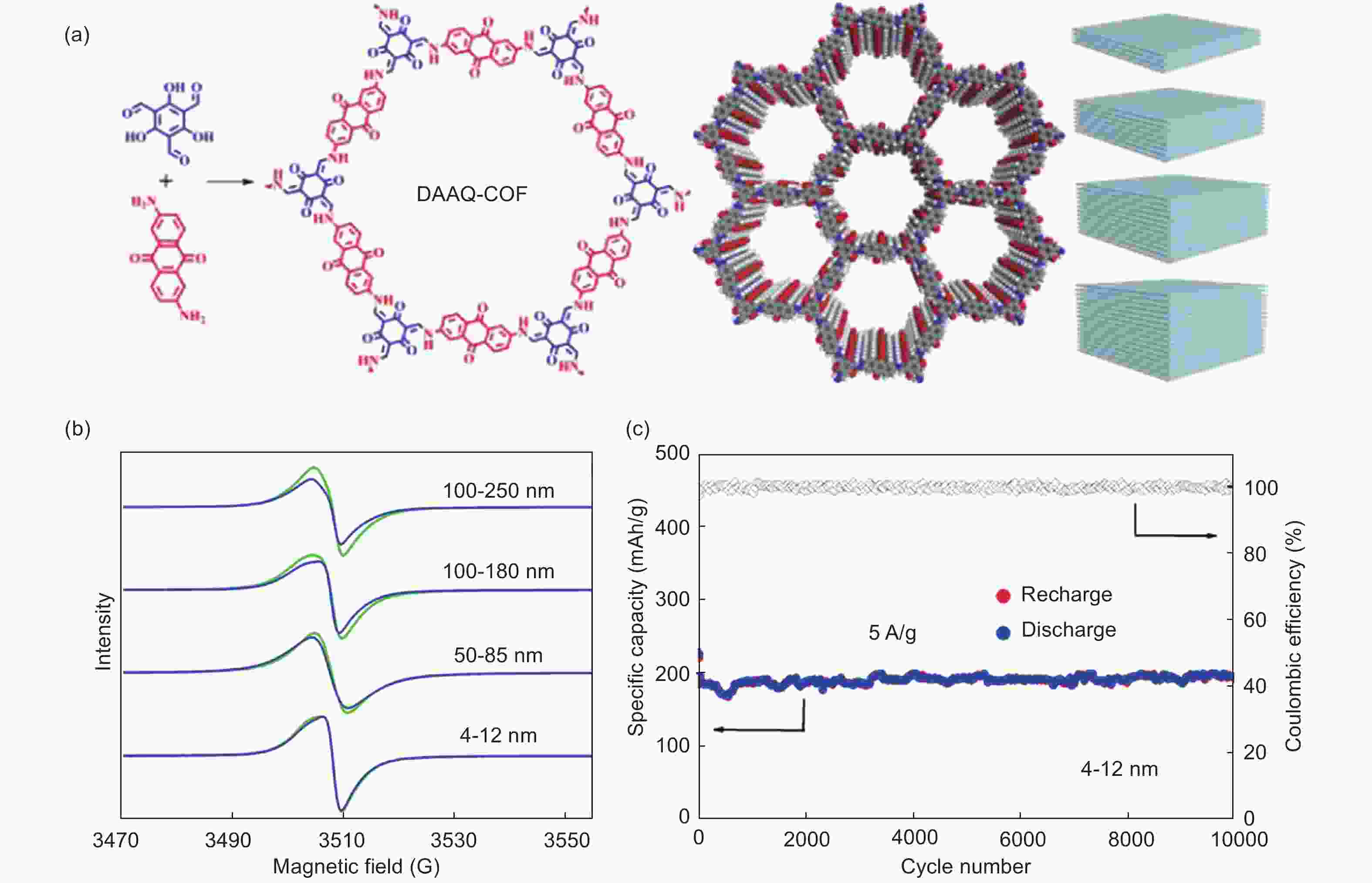
Figure 10. (a) Structures of DAAQ-COF with different thicknesses, (b) change of EPR signal for COFs with different thicknesses and (c) the cyclic performance for DAAQ-COF with a thickness of 4-12 nm[67] (Reproduced by permission of American Chemical Society).
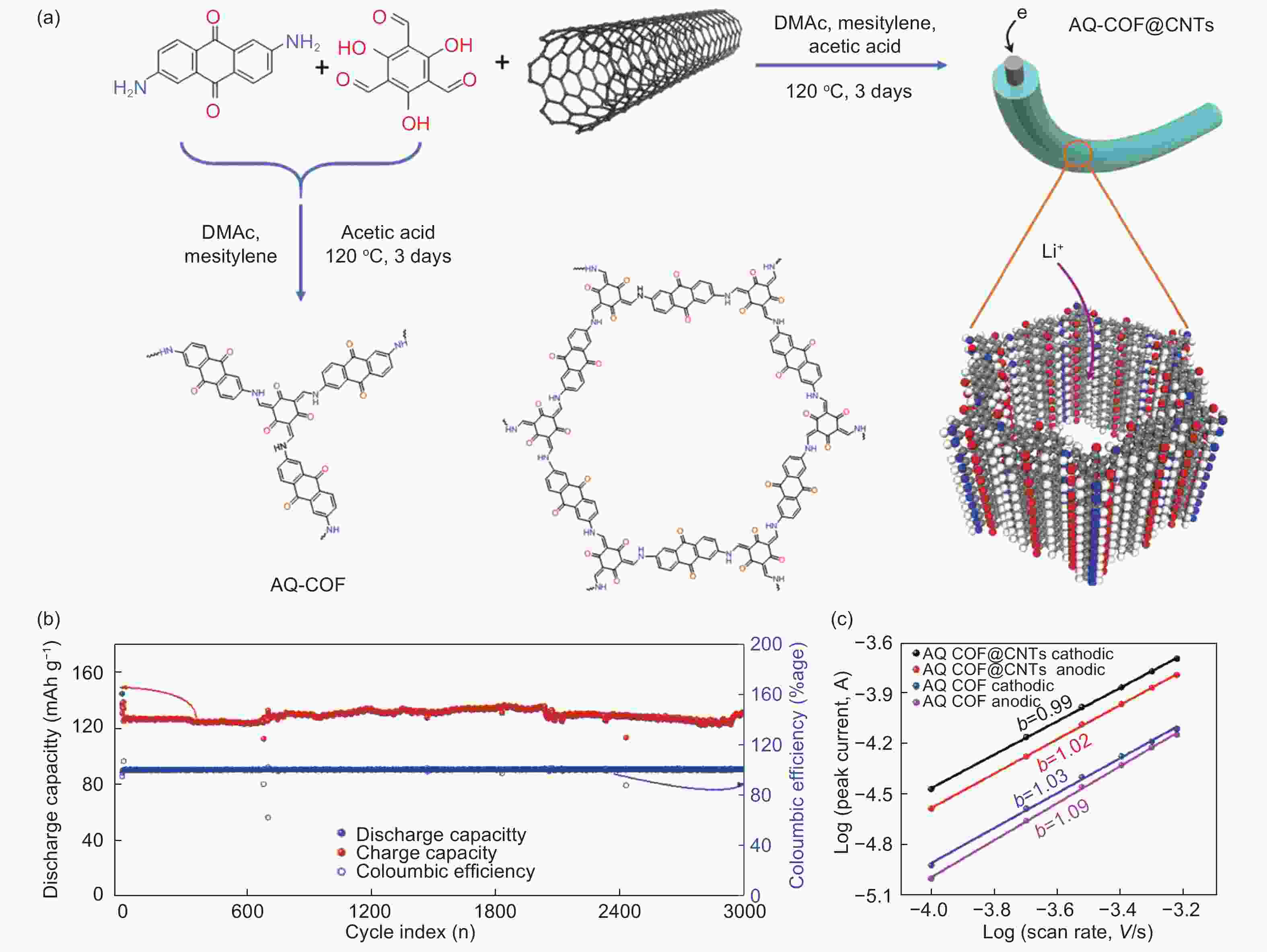
Figure 11. (a) Synthesis of AQ-COF@CNTs, (b) the cyclic performance of AQ-COF@CNTs and (c) lgi-lgv curves[75] (Reproduced by permission of the Royal Society of Chemistry).
Table 1. The representative COF electrode materials in RMBs.
COFs’ name Application
ScenariosCurrent density (mA g−1)/
Specific capacity (mAh g−1)Voltage
Window (V)Lifespan (cycles)/
Capacity retention (%)Current density (A g−1)/
Specific capacity (mAh g−1)Ref. DTP–ANDI–COF@CNT LIB 200/70 1.5−3.5 700/100 1/60 [52] PIBN-COF@Graphene LIB 28/271 1.5−3.5 300/86 2.8/200 [54] DAAQ-TFP–COF LIB 20/110 1.5−4 1000/100 3/20 [66] DAAQ-ECOF LIB 20/145 1.5−4 1800/100 3/50 [66] DAAQ-COF SIB 100/420 0.05−3 10000/99 5/200 [67] DABQ-ECOF LIB 20/~200 1.5−3.5 / / [66] TEMPO–ECOF LIB 20/~100 2−4 / / [66] TQBQ-COF SIB 20/350 1−3.6 1000/96 10/134 [53] TRO–COF LIB 27.4/268 0.5−4.5 100/99.9 0.548/100 [62] N2–COF LIB 1000/700 0.05−3 500/75 5/500 [68] N3–COF LIB 1000/700 0.05−3 500/65 5/550 [68] Tp-DANT-COF LIB 200/78.9 1.5−4 600/90 2/70 [64] Tb-DANT-COF LIB 50/135 1.5−4 300/83 2/67 [64] rCTF LIB 300/1190 0.005−3 1500/100 12/500 [69] PI-ECOF-1 LIB 14.2/112 1.5−3.5 300/75 0.284/70 [65] PI-ECOF-2 LIB 12.8/103 1.5−3.5 300/75 0.256/40 [65] TFPB-TAPT COF SIB 30/200 0.05−1.6 500/50.8 0.2/145 [70] BQ1-COF LIB 39/502 1.2−3.5 1000/81 7.73/171 [71] C2N-450 LIB 37.2/933 0.02−3 500/130.6 3.72/40.1 [72] C3N LIB 37.2/787 0.02−3 500/91 3.72/180 [72] PGF-1 LIB 500/480 1−3.5 1400/78.3 5/200 [73] DBA-COF 3 LIB 50/200 0.05−3 90/100 1/90 [74] AQ-COF@CNT LIB 50/71 1.5−3.5 3000/100 2.5/11 [75] TThPP LIB 1000/400 0.005−3 200/100 4/200 [60] JUC-526 LIB 200/441.2 0.01−3 500/100 2/200 [76] HATN-CMP LIB 100/150 1.5−4 50/60 1/50 [77] IISERP-CON1 LIB 100/720 0.01−3 1000/100 2/460 [78] IISERP-COF16 SIB 100/80 0.01−3 500/100 / [79] IISERP-COF17 SIB 100/200 0.01−3 500/100 1/100 [79] IISERP-COF18 SIB 100/400 0.01−3 1400/92 15/127 [79] Pa-COF LIB 1000/401.3 0.01−3.5 2000/74.8 5/141.8 [80] Tb-COF LIB 1000/379.1 0.01−3.5 2000/72.7 5/144 [80] TAPB-terephthal Aldehyde COFs SIB 100/303 0−3 1000/85 5/150 [81] Cz-COF1 LIB 200/300 0.01−3 400/59 1/200 [82] Cz-COF2 200/250 400/50 1/100 [82] Tp-Azo-COF LIB 1000/306 0.01−3 3000/100 2.4/100 [83] TpBpy AIB 2000/150 0.01−2.3 13000/100 5/113 [84] CTF-0 KIB 100/100 0.01−3 350/67 1/63 [85] CTF-1 100/50 350/67 1/31 [85] DAPT-TFP-CPF SIB 100/150 0.8−3.2 1400/94.7 5/100 [86] PDI-Tc LIB 190/45 1.8−3.2 500/80 0.482/22 [87] HqTp-COF ZIB 625/150 0.2−1.8 1000/95 1.25/125 [56] COF-CNT LIB 100/1536 0.005−3 500/510 5/200 [54] 2D CCP-HATN LIB 500/116 1.2−3.9 1000/91 1/94 [55] E-CIN-1/CNT LIB 100/538 0.001−3 250/124 5/97 [58] E-SNW-1/CNT 100/542 250/91 5/212 [58] COF-10@CNT KIB 100/288 0.005−3 4000/54 5/68 [59] E-TFPB-COF/MnO2 LIB 100/1250 0.005−3 300/135 5/500 [88] Si@COF LIB 1000/2000 0.01−1 1000/60 5/1000 [89] -
[1] Dunn B, Kamath H, Tarascon J. Electrical energy storage for the grid: A battery of choices[J]. Science,2011,334(6058):928. doi: 10.1126/science.1212741 [2] Lukatskaya M, Dunn B, Gogotsi Y. Multidimensional materials and device architectures for future hybrid energy storage[J]. Nature Communications,2016,7(1):12647. doi: 10.1038/ncomms12647 [3] Wang L, Chen B, Ma J, et al. Reviving lithium cobalt oxide-based lithium secondary batteries-toward a higher energy density[J]. Chemical Society Reviews,2018,47(17):6505-6602. doi: 10.1039/C8CS00322J [4] Wang Z, Jin W, Huang X, et al. Covalent organic frameworks as electrode materials for metal ion batteries: A current review[J]. The Chemical Record,2020,20(10):1198-1219. doi: 10.1002/tcr.202000074 [5] Wen Y, Wang X, Yang Y, et al. Covalent organic framework-regulated ionic transportation for high-performance lithium-ion batteries[J]. Journal of Materials Chemistry A,2019,7(46):26540-26548. doi: 10.1039/C9TA09570E [6] Hwang J, Myung S, Sun Y. Sodium-ion batteries: Present and future[J]. Chemical Society Reviews,2017,46:3529-3614. doi: 10.1039/C6CS00776G [7] Song M, Tan H, Chao D, et al. Recent advances in Zn-ion batteries[J]. Advanced Functional Materials,2018,28(41):1802564. doi: 10.1002/adfm.201802564 [8] Muldoon J, Bucur C, Oliver A, et al. Electrolyte roadblocks to a magnesium rechargeable battery[J]. Energy & Environmental Science,2012,5:5941. [9] Wu X, Leonard D, Ji X. Emerging non-aqueous potassium-ion batteries: Challenges and opportunities[J]. Chemistry of Materials,2017,29:5031-5042. doi: 10.1021/acs.chemmater.7b01764 [10] Wang M, Jiang C, Zhang S, et al. Reversible calcium alloying enables a practical room-temperature rechargeable calcium-ion battery with a high discharge voltage[J]. Nature Chemistry,2018,10(6):667-672. doi: 10.1038/s41557-018-0045-4 [11] Zhang Y, Liu S, Ji Y, et al. Emerging nonaqueous aluminum-ion on batteries: Challenges, status, and perspectives[J]. Advanced Materials,2018,30:1706310. doi: 10.1002/adma.201706310 [12] Nishi Y. Lithium ion secondary batteries; past 10 years and the future[J]. Journal of Power Sources,2001,100:101-106. doi: 10.1016/S0378-7753(01)00887-4 [13] Li M, Lu J, Chen Z, et al. 30 Years of lithium-ion batteries[J]. Advanced Materials,2018,30:1800561. doi: 10.1002/adma.201800561 [14] Ozawa K. Lithium-ion rechargeable batteries with LiCoO2 and carbon electrodes: The LiCoO2/C system[J]. Solid State Ionics Diffusion & Reactions,1994,69:212-221. [15] Jiang Q, Han Z, Wang S. Plasma-enhanced low-temperature solid-state synthesis of spinel LiMn2O4 with superior performance for lithium-ion batteries[J]. Green Chemistry,2016,18:662-666. doi: 10.1039/C5GC01563D [16] Prosini P. Iron Phosphate Materials as Cathodes for Lithium Vatteries [M]. Italy: Spring, 2011, 73-82. [17] Yoshio M, Wang H, Fukuda K, et al. Effect of carbon coating on electrochemical performance of treated natural graphite as lithium-ion battery anode material[J]. Journal of the Electrochemical Society,2000,147(4):1245-1250. doi: 10.1149/1.1393344 [18] Lee H, Baek J, Lee S, et al. Effect of carbon coating on elevated temperature performance of graphite as lithium-ion battery anode material[J]. Journal of Power Sources,2004,128(1):61-66. doi: 10.1016/j.jpowsour.2003.09.051 [19] Son I, Hwan Park J, Kwon S, et al. Silicon carbide-free graphene growth on silicon for lithium-ion battery with high volumetric energy density[J]. Nature Communications,2015,6:7393. doi: 10.1038/ncomms8393 [20] Zuo X, Zhu J, Müller-Buschbaum P, et al. Silicon based lithium-ion battery anodes: A chronicle perspective review[J]. Nano Energy,2017,31:113-143. doi: 10.1016/j.nanoen.2016.11.013 [21] Zhang X, Wang D, Qiu X, et al. Stable high-capacity and high-rate silicon-based lithium battery anodes upon two-dimensional covalent encapsulation[J]. Nature Communications,2020,11(1):3826. doi: 10.1038/s41467-020-17686-4 [22] Zeng X, Zhan C, Lu J, et al. Stabilization of a high-capacity and high-power nickel-based cathode for Li-ion batteries[J]. Chem,2018,4(4):690-704. doi: 10.1016/j.chempr.2017.12.027 [23] Xing Z, Wang S, Yu A, et al. Aqueous intercalation-type electrode materials for grid-level energy storage: Beyond the limits of lithium and sodium[J]. Nano Energy,2018,50:229-244. doi: 10.1016/j.nanoen.2018.05.049 [24] Goodenough J, Park K. The Li-ion rechargeable battery: A perspective[J]. Journal of the American Chemical Society,2013,135(4):1167-1176. doi: 10.1021/ja3091438 [25] Wang Y, He P, Zhou H. A lithium–air capacitor–battery based on a hybrid electrolyte[J]. Energy & Environmental Science,2011,4(12):4994-4999. [26] Braun P, Cho J, Pikul J, et al. High power rechargeable batteries[J]. Current Opinion in Solid State and Materials Science,2012,16(4):186-198. doi: 10.1016/j.cossms.2012.05.002 [27] Guerfi A, Trottier J, Boyano I, et al. High cycling stability of zinc-anode/conducting polymer rechargeable battery with non-aqueous electrolyte[J]. Journal of Power Sources,2014,248(15):1099-1104. [28] Zibin L, Chong Q, Wenhan G, et al. Metal-organic frameworks: pristine metal-organic frameworks and their composites for energy storage and conversion[J]. Advanced Materials,2018,30(37):1870276. doi: 10.1002/adma.201870276 [29] Chen N, Zhang H, Li L, et al. Ionogel electrolytes for high‐performance lithium batteries: A review[J]. Advanced Energy Materials,2018,8(12):1702675. doi: 10.1002/aenm.201702675 [30] Wang F, Wu X, Yuan X, et al. Latest advances in supercapacitors: From new electrode materials to novel device designs[J]. Chemical Society Reviews,2017,46(22):6816-6854. doi: 10.1039/C7CS00205J [31] Medina D, Sick T, Bein T. Photoactive and conducting covalent organic frameworks[J]. Advanced Energy Materials,2017,7(16):1700387. doi: 10.1002/aenm.201700387 [32] Pachfule P, Acharjya A, Roeser J, et al. Diacetylene functionalized covalent organic framework (COF) for photocatalytic hydrogen generation[J]. Journal of the American Chemical Society,2017,140(4):1423-1427. [33] Lin C, Zhang L, Zhao Z, et al. Design principles for covalent organic frameworks as efficient electrocatalysts in clean energy conversion and green oxidizer production[J]. Advanced Materials,2017,29(17):1606635. doi: 10.1002/adma.201606635 [34] Wu D, Xu Q, Jing Q, et al. Bimetallic covalent organic frameworks for constructing multifunctional electrocatalyst[J]. Chemistry,2019,26:3105-3111. [35] Yang S, Hu W, Zhang X, et al. 2D covalent organic frameworks as intrinsic photocatalysts for visible light-driven CO2 reduction[J]. Journal of the American Chemical Society,2018,140(44):14614-14618. doi: 10.1021/jacs.8b09705 [36] Fan H, Mundstock A, Feldhoff A, et al. Covalent organic framework-covalent organic framework bilayer membranes for highly selective gas separation[J]. Journal of the American Chemical Society,2018,140:10094-10098. doi: 10.1021/jacs.8b05136 [37] Fu J, Das S, Xing G, et al. Fabrication of COF-MOF composite membranes and their highly selective separation of H2/CO2[J]. Journal of the American Chemical Society,2016,138(24):7673-7680. doi: 10.1021/jacs.6b03348 [38] Kang Z, Peng Y, Qian Y, et al. Mixed matrix membranes (MMMs) comprising exfoliated 2D covalent organic frameworks (COFs) for efficient CO2 separation[J]. Chemistry of Materials,2016,28(5):1277-1285. doi: 10.1021/acs.chemmater.5b02902 [39] Alhmoud H, Delalat B, Elnathan R, et al. Porous silicon nanodiscs for targeted drug delivery[J]. Advanced Functional Materials,2015,25(7):1137-1145. doi: 10.1002/adfm.201403414 [40] Zhang G, Li X, Liao Q, et al. Water-dispersible PEG-curcumin/amine-functionalized covalent organic framework nanocomposites as smart carriers for in vivo drug delivery[J]. Nature Communications,2018,9:2785. doi: 10.1038/s41467-018-04910-5 [41] Rosi N. Hydrogen storage in microporous metal-organic frameworks[J]. Science,2003,300(5622):1127-1129. doi: 10.1126/science.1083440 [42] Furukawa H, Yaghi O. Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications[J]. Journal of the American Chemical Society,2009,131(25):8875-8883. doi: 10.1021/ja9015765 [43] Geng K, He T, Liu R, et al. Covalent organic frameworks: Design, synthesis, and functions[J]. Chemical Reviews,2020,120:8814-8933. doi: 10.1021/acs.chemrev.9b00550 [44] Côté Adrien P, Benin Annabelle I, Ockwig Nathan W, et al. Porous, crystalline, covalent organic frameworks[J]. Science,2005,310(5751):1166-1170. doi: 10.1126/science.1120411 [45] Feng X, Ding X, Jiang D. Covalent organic frameworks[J]. Chemical Society Reviews,2012,41(18):6010-6022. doi: 10.1039/c2cs35157a [46] Sun T, Xie J, Guo W, et al. Covalent-organic frameworks: Advanced organic electrode materials for rechargeable batteries[J]. Advanced Energy Materials,2020,10:1904199. doi: 10.1002/aenm.201904199 [47] Huang N, Wang P, Jiang D. Covalent organic frameworks: A materials platform for structural and functional designs[J]. Nature Reviews Materials,2016,1:16068. doi: 10.1038/natrevmats.2016.68 [48] Geng K, Arumugam V, Xu H, et al. Covalent organic frameworks: Polymer chemistry and functional design[J]. Progress in Polymer Science,2020,108:101288. doi: 10.1016/j.progpolymsci.2020.101288 [49] Yusran Y, Fang Q, Valtchev V. Electroactive covalent organic frameworks: Design, synthesis, and applications[J]. Advanced Materials,2020,32:2002038. doi: 10.1002/adma.202002038 [50] Hu Y, Wayment L, Haslam C, et al. Covalent organic framework based lithium-ion battery: Fundamental, design and characterization[J]. EnergyChem,2020,3(1):100048. [51] Xiao Z, Li L, Tang Y, et al. Covalent organic frameworks with lithiophilic and sulfiphilic dual linkages for cooperative affinity to polysulfides in lithium-sulfur batteries[J]. Energy Storage Materials,2018,12:252-259. doi: 10.1016/j.ensm.2018.01.018 [52] Xu F, Jin S, Zhong H, et al. Electrochemically active, crystalline, mesoporous covalent organic frameworks on carbon nanotubes for synergistic lithium-ion battery energy storage[J]. Scientific Reports,2015,5(1):8225. doi: 10.1038/srep08225 [53] Shi R, Liu L, Lu Y, et al. Nitrogen-rich covalent organic frameworks with multiple carbonyls for high-performance sodium batteries[J]. Nature Communications,2020,11(1):178. doi: 10.1038/s41467-019-13739-5 [54] Luo Z, Liu L, Ning J, et al. A microporous covalent organic framework with abundant accessible carbonyl groups for lithium‐ion batteries[J]. Angewandte Chemie International Edition,2018,57(30):9443-9446. doi: 10.1002/anie.201805540 [55] Xu S, Wang G, Biswal B, et al. A nitrogen-rich 2D sp2-carbon-linked conjugated polymer framework as a high-performance cathode for lithium-ion batteries[J]. Angewandte Chemie International Edition,2018,131(1):859-863. [56] Khayum M, Ghosh M, Vijayakumar V, et al. Zinc ion interactions in a two-dimensional covalent organic framework based aqueous zinc ion battery[J]. Chemical Science,2019,10(38):8889-8894. doi: 10.1039/C9SC03052B [57] Lei Z, Yang Q, Xu Y, et al. Boosting lithium storage in covalent organic framework via activation of 14-electron redox chemistry[J]. Nature Communications,2018,9(1):576. doi: 10.1038/s41467-018-02889-7 [58] Lei Z, Chen X, Sun W, et al. Exfoliated triazine-based covalent organic nanosheets with multielectron redox for high-performance lithium organic batteries[J]. Advaced Energy Materials,2018,9(3):1801010. [59] Chen X, Zhang H, Ci C, et al. Few-layered boronic ester based covalent organic frameworks/carbon nanotube composites for high-performance K-organic batteries[J]. ACS Nano,2019,13(3):3600-3607. doi: 10.1021/acsnano.9b00165 [60] Yang H, Zhang S, Han L, et al. High conductive two-dimensional covalent organic framework for lithium storage with large capacity[J]. ACS Applied Materials & Interfaces,2016,8(8):5366-5375. [61] Zhang H, Sun W, Chen X, et al. Few-layered fluorinated triazine-based covalent organic nanosheets for high-performance alkali organic batteries[J]. ACS Nano,2019,13(12):14252-14261. doi: 10.1021/acsnano.9b07360 [62] Yang X, Hu Y, Dunlap N, et al. A truxenone-based covalent organic framework as all-solid-state Li-ion battery cathode with high capacity[J]. Angewandte Chemie International Edition,2020,59(46):20385-20389. doi: 10.1002/anie.202008619 [63] Sakaushi K, Hosono E, Nickerl G, et al. Aromatic porous-honeycomb electrodes for a sodium-organic energy storage device[J]. Nature Communications,2013,4(1):1485. doi: 10.1038/ncomms2481 [64] Yang D, Yao Z, Wu D, et al. Structure-modulated crystalline covalent organic frameworks as high-rate cathodes for Li-ion batteries[J]. Journal of Materials Chemistry A,2016,4(47):18621-18627. doi: 10.1039/C6TA07606H [65] Wang Z, Li Y, Liu P, et al. Few layer covalent organic frameworks with graphene sheets as cathode materials for lithium-ion batteries[J]. Nanoscale,2019,11(12):5330-5335. doi: 10.1039/C9NR00088G [66] Wang S, Wang Q, Shao P, et al. Exfoliation of covalent organic frameworks into few-layer redox-active nanosheets as cathode materials for lithium-ion batteries[J]. Journal of the American Chemical Society,2017,139(12):4258-4261. doi: 10.1021/jacs.7b02648 [67] Gu S, Wu S, Cao L, et al. Tunable redox chemistry and stability of radical intermediates in 2D covalent organic frameworks for high performance sodium ion batteries[J]. Journal of the American Chemical Society,2019,141(24):9623-9628. doi: 10.1021/jacs.9b03467 [68] Bai L, Gao Q, Zhao Y. Two fully conjugated covalent organic frameworks as anode materials for lithium ion batteries[J]. Journal of Materials Chemistry A,2016,4(37):14106-14110. doi: 10.1039/C6TA06449C [69] Buyukcakir O, Ryu J, Joo S, et al. Lithium accommodation in a redox-active covalent triazine framework for high areal capacity and fast-charging lithium-ion batteries[J]. Advanced Functional Materials,2020,30(36):2003761. doi: 10.1002/adfm.202003761 [70] Patra B, Das S, Ghosh A, et al. Covalent organic framework based microspheres as an anode material for rechargeable sodium batteries[J]. Journal of Materials Chemistry A,2018,6(34):16655-16663. doi: 10.1039/C8TA04611E [71] Wu M, Zhao Y, Sun B, et al. A 2D covalent organic framework as a high-performance cathode material for lithium-ion batteries[J]. Nano Energy,2020,70:104498. doi: 10.1016/j.nanoen.2020.104498 [72] Xu J, Mahmood J, Dou Y, et al. 2D frameworks of C2N and C3N as new anode materials for lithium-ion batteries[J]. Advanced Materials,2017,29(34):1702007. doi: 10.1002/adma.201702007 [73] Li X, Wang H, Chen H, et al. Dynamic covalent synthesis of crystalline porous graphitic frameworks[J]. Chem,2020,6(4):933-944. doi: 10.1016/j.chempr.2020.01.011 [74] Wolfson E, Xiao N, Schkeryantz L, et al. A dehydrobenzoannulene-based two-dimensional covalent organic framework as an anode material for lithium-ion batteries[J]. Molecular Systems Design & Engineering,2020,5(1):97-101. [75] Amin K, Zhang J, Zhou H, et al. Surface controlled pseudo-capacitive reactions enabling ultra-fast charging and long-life organic lithium ion batteries[J]. Sustainable Energy & Fuels,2020,4(8):4179-4185. [76] Yu X, Li C, Ma Y, et al. Crystalline, porous, covalent polyoxometalate-organic frameworks for lithium-ion batteries[J]. Microporous and Mesoporous Materials,2020,299:110105. doi: 10.1016/j.micromeso.2020.110105 [77] Xu F, Chen X, Tang Z, et al. Redox-active conjugated microporous polymers: A new organic platform for highly efficient energy storage[J]. Chemical Communications,2014,50(37):4788-4790. doi: 10.1039/C4CC01002G [78] Haldar S, Roy K, Nandi S, et al. High and reversible lithium ion storage in self-exfoliated triazole-triformyl phloroglucinol-based covalent organic nanosheets[J]. Advanced Energy Materials,2018,8(8):1702170. doi: 10.1002/aenm.201702170 [79] Haldar S, Kaleeswaran D, Rase D, et al. Tuning the electronic energy level of covalent organic frameworks for crafting high-rate Na-ion battery anode[J]. Nanoscale Horizons,2020,5(8):1264-1273. doi: 10.1039/D0NH00187B [80] Chen H, Zhang Y, Xu C, et al. Two π-conjugated covalent organic frameworks with long-term cyclability at high current density for lithium ion battery[J]. Chemistry A European Journal,2019,25(68):15472-15476. doi: 10.1002/chem.201903733 [81] Zhang Y, Gao Z. High performance anode material for sodium-ion batteries derived from covalent-organic frameworks[J]. Electrochimica Acta,2019,301:23-28. doi: 10.1016/j.electacta.2019.01.147 [82] Feng S, Xu H, Zhang C, et al. Bicarbazole-based redox-active covalent organic frameworks for ultrahigh-performance energy storage[J]. Chemical Communications,2017,53(82):11334-11337. doi: 10.1039/C7CC07024A [83] Zhao G, Zhang Y, Gao Z, et al. Dual active site of the Azo and Carbonyl-modified covalent organic framework for high-performance Li storage[J]. ACS Energy Lettters,2020,5(4):1022-1031. doi: 10.1021/acsenergylett.0c00069 [84] Lu H, Ning F, Jin R, et al. Two-dimensional covalent organic frameworks with enhanced aluminum storage properties[J]. ChemSusChem,2020,13(13):3447-3454. doi: 10.1002/cssc.202000883 [85] Li S, Li W, Wu X, et al. Pore-size dominated electrochemical properties of covalent triazine frameworks as anode materials for K-ion batteries[J]. Chemical Science,2019,10(33):7695-7701. doi: 10.1039/C9SC02340B [86] Li H, Tang M, Wu Y, et al. Large π-conjugated porous frameworks as cathodes for sodium-ion batteries[J]. The Journal of Physical Chemistry Letters,2018,9(12):3205. doi: 10.1021/acs.jpclett.8b01285 [87] Schon T, Tilley A, Kynaston E, et al. Three-dimensional arylene diimide frameworks for highly stable lithium ion batteries[J]. ACS Applied Materials & Interfaces,2017,9(18):15631. [88] Chen X, Li Y, Wang L, et al. High-lithium-affinity chemically exfoliated 2D covalent organic frameworks[J]. Advanced Materials,2019,31(29):1901640. doi: 10.1002/adma.201901640 [89] Ai Q, Fang Q, Liang J, et al. Lithium-conducting covalent-organic-frameworks as artificial solid-electrolyte-interphase on silicon anode for high performance lithium ion batteries[J]. Nano Energy,2020,72:104657. doi: 10.1016/j.nanoen.2020.104657 [90] Wu F, Tan G, Chen R, et al. Novel solid-state Li/LiFePO4 battery configuration with a ternary nanocomposite electrolyte for practical applications[J]. Advanced Materials,2011,23(43):5081-5085. doi: 10.1002/adma.201103161 [91] Luo S, Wang K, Wang J, et al. Binder-free LiCoO2/carbon nanotube cathodes for high-performance lithium ion batteries[J]. Advanced Materials,2012,24(17):2294-2298. doi: 10.1002/adma.201104720 [92] Jung Y, Lu P, Cavanagh A, et al. Unexpected improved performance of ALD coated LiCoO2/graphite Li-ion batteries[J]. Advanced Energy Materials,2013,3(2):213-219. doi: 10.1002/aenm.201200370 [93] Shim J, Lee S, Park S. Effects of MgO coating on the structural and electrochemical characteristics of LiCoO2 as cathode materials for lithium ion battery[J]. Chemistry of Materials,2014,26(8):2537-2543. doi: 10.1021/cm403846a [94] Liang Y, Zhang P, Yang S, et al. Fused heteroaromatic organic compounds for high-power electrodes of rechargeable lithium batteries[J]. Advanced Energy Materials,2013,3(5):600-605. doi: 10.1002/aenm.201200947 [95] Gottis S, Barrès A, Dolhem F, et al. Voltage gain in lithiated enolate-based organic cathode materials by isomeric effect[J]. ACS Applied Materials & Interfaces,2014,6(14):10870-10876. [96] Wu D, Xie Z, Zhou Z, et al. Designing high-voltage carbonyl-containing polycyclic aromatic hydrocarbon cathode materials for Li-ion batteries guided by Clar's theory[J]. Journal of Materials Chemistry A,2015,3(37):19137-19143. doi: 10.1039/C5TA05437K [97] Lu Y, Zhang Q, Li L, et al. Design strategies toward enhancing the performance of organic electrode materials in metal-ion batteries[J]. Chem,2018,4(12):2786-2813. doi: 10.1016/j.chempr.2018.09.005 [98] Kim H, Kwon J, Lee B, et al. High energy organic cathode for sodium rechargeable batteries[J]. Chemistry of Materials,2015,27(21):7258-7264. doi: 10.1021/acs.chemmater.5b02569 [99] Patil N, Aqil A, Ouhib F, et al. Bioinspired redox-active catechol-bearing polymers as ultrarobust organic cathodes for lithium storage[J]. Advanced Materials,2017,29(40):1703373. doi: 10.1002/adma.201703373 [100] Lu Y, Chen J. Prospects of organic electrode materials for practical lithium batteries[J]. Nature Reviews Chemistry,2020,4(3):127-142. doi: 10.1038/s41570-020-0160-9 [101] Schmuch R, Wagner R, Hörpel G, et al. Performance and cost of materials for lithium-based rechargeable automotive batteries[J]. Nature Energy,2018,3(4):267-278. doi: 10.1038/s41560-018-0107-2 [102] Pramudita J, Sehrawat D, Goonetilleke D, et al. An initial review of the status of electrode materials for potassium-ion batteries[J]. Advanced Energy Materials,2017,7(24):1602911. doi: 10.1002/aenm.201602911 [103] Kim S, Seo D, Ma X, et al. Electrode materials for rechargeable sodium-ion batteries: Potential alternatives to current lithium-ion batteries[J]. Advanced Energy Materials,2012,2(7):710-721. doi: 10.1002/aenm.201200026 [104] Zhang C, Qiao Y, Xiong P, et al. Conjugated microporous polymers with tunable electronic structure for high-performance potassium-ion batteries[J]. ACS Nano,2019,13(1):745-754. doi: 10.1021/acsnano.8b08046 [105] Song Z, Zhan H, Zhou Y. Polyimides: Promising energy-storage materials[J]. Angewandte Chemie International Edition,2010,49(45):8444-8448. doi: 10.1002/anie.201002439 [106] WangOrcid J, Chen C, Zhang Y. Hexaazatrinaphthylene-based porous organic polymers as organic cathode materials for lithium-ion batteries[J]. ACS Sustainable Chemistry & Engineering,2018,6(2):1772-1779. [107] Wang J, Lee Y, Tee K, et al. A nanoporous sulfur-bridged hexaazatrinaphthylene framework as an organic cathode for lithium ion batteries with well-balanced electrochemical performance[J]. Chemical Communications,2018,54(55):7681-7684. doi: 10.1039/C8CC03801E [108] Ye X, Huang Y, Tang X, et al. Two-dimensional extended π-conjugated triphenylene-core covalent organic polymer[J]. Journal of Materials Chemistry A,2019,7(7):3066-3071. doi: 10.1039/C8TA10554E [109] Kou Y, Xu Y, Guo Z, et al. Supercapacitive energy storage and electric power supply using an Aza-fused π-conjugated microporous framework[J]. Angewandte Chemie International Edition,2011,123(37):8912-8916. [110] Kim H, Seo D, Yoon G, et al. The reaction mechanism and capacity degradation model in lithium insertion organic cathodes, Li2C6O6, using combined experimental and first principle studies[J]. The Journal of Physical Chemistry Letters,2014,5(17):3086-3092. doi: 10.1021/jz501557n [111] Wang Y, Ding Y, Pan L, et al. Understanding the size-dependent sodium storage properties of Na2C6O6-based organic electrodes for sodium-ion batteries[J]. Nano Letters,2016,16(5):3329-3334. doi: 10.1021/acs.nanolett.6b00954 [112] Luo C, Fan X, Ma Z, et al. Self-healing chemistry between organic material and binder for stable sodium-ion batteries[J]. Chem,2017,3(6):1050-1062. doi: 10.1016/j.chempr.2017.09.004 [113] Iordache A, Delhorbe V, Bardet M, et al. Perylene-based all-organic redox battery with excellent cycling stability[J]. ACS Applied Materials & Interfaces,2016,8(35):22762-22767. [114] Shi Y, Tang H, Jiang S, et al. Understanding the electrochemical properties of naphthalene diimide: implication for stable and high-rate lithium-ion battery electrodes[J]. Chemistry of Materials,2018,30(10):3508-3517. doi: 10.1021/acs.chemmater.8b01304 [115] Kundu D, Oberholzer P, Glaros C, et al. Organic cathode for aqueous Zn-ion batteries: Taming a unique phase evolution toward stable electrochemical cycling[J]. Chemistry of Materials,2018,30(11):3874-3881. doi: 10.1021/acs.chemmater.8b01317 [116] Song Z, Qian Y, Gordin M, et al. Polyanthraquinone as a reliable organic electrode for stable and fast lithium storage[J]. Angewandte Chemie International Edition,2015,127(47):14153-14157. [117] Muench S, Wild A, Friebe C, et al. Polymer-based organic batteries[J]. Chemical Reviews,2016,116(16):9438-9484. doi: 10.1021/acs.chemrev.6b00070 [118] Fang L, Cao X, Cao Z. Covalent organic framework with high capacity for the lithium ion battery anode: Insight into intercalation of Li from first-principles calculations[J]. Journal of Physics: Condensed Matter,2019,31(20):205502. doi: 10.1088/1361-648X/ab087e -






 下载:
下载: