-
摘要: 煤炭清洁高效利用是煤炭产业精细化、高值化发展的必由之路。作为自然界广泛存在的高碳资源,以煤炭为原料开发煤基新型炭材料,拓展煤炭利用新途径是一个具有前景而富有挑战性的课题。近年来,研究者以煤炭及其衍生物为含碳前驱体,设计并构筑了具有丰富形态和结构的纳米炭材料,进一步考察了其在储能、催化、吸附与分离等领域的应用。本文综述了基于煤炭及其衍生物开发新型炭纳米材料的最新研究进展,重点介绍了针对不同煤阶煤制备炭纳米材料的设计合成方法和结构调控策略,讨论了煤基炭纳米材料在二次电池和超级电容器为主的储能过程中的应用。最后,对煤炭材料化利用的未来发展进行了展望,以期为先进煤衍生炭纳米材料的精细设计和可控制备等方面提供新的研究思路。Abstract: As a natural abundant high-carbon resource, the use of coal to develop carbon nanomaterials is an important research topic. In recent years, a variety of carbon materials with different morphologies and nanotextures have been designed and constructed using coal and their derivatives as precursors, and their use in energy storage, catalysis, adsorption and absorption have been explored. State-of-the-art research on carbon nanomaterials derived from coals of different rank and their derivatives are summarized with specific attention to the synthesis strategies and structure control. The use of these coal-derived carbons for energy storage, such as secondary batteries and supercapacitors, is also discussed in terms of their structural features. The review aims to provide valuable insight into the present challenges and inspire new ideas for the development of advanced coal-derived carbon materials.
-
Key words:
- Coal /
- Carbon materials /
- Carbon nanotubes /
- Graphene /
- Porous carbon /
- Energy storage
-
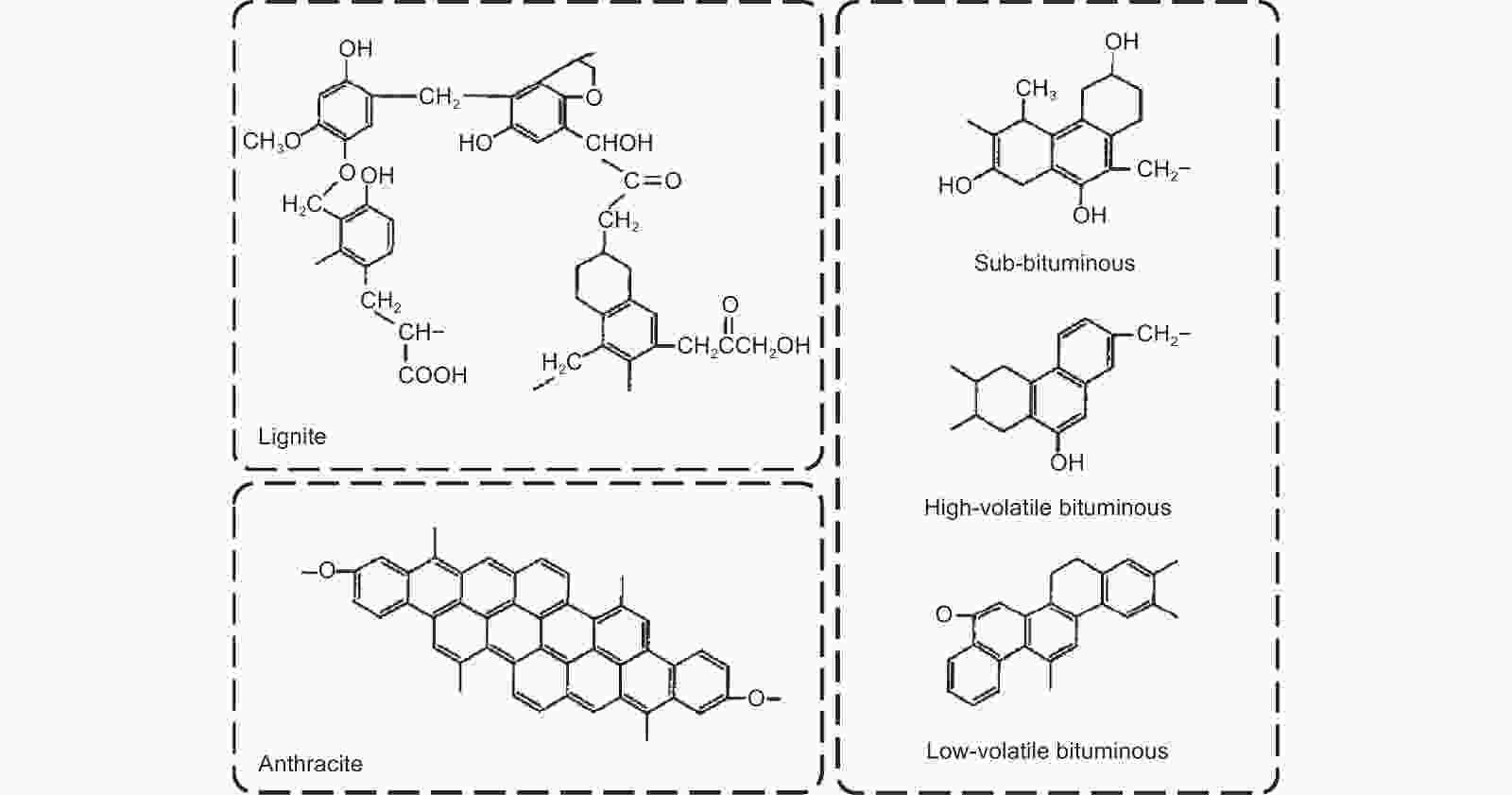
Figure 1. Representative structures of different rank coals[29]. Reprinted with permission.
Figure 2. (a) Macroscale image and illustrative nanostructure of coal. (b) SEM image of ground bituminous coal, (c) Schematic illustration of the synthesis of b-GQDs (Oxygenated sites are shown in red), (d) TEM image of b-GQDs showing a regular size and shape distribution,(e) HRTEM image of representative b-GQDs from (d) (the inset is the 2D FFT image that shows the crystalline hexagonal structure of these quantum dots) and (f) AFM image of b-GQDs showing height of 1.5−3 nm[45]. Reprinted with permission.
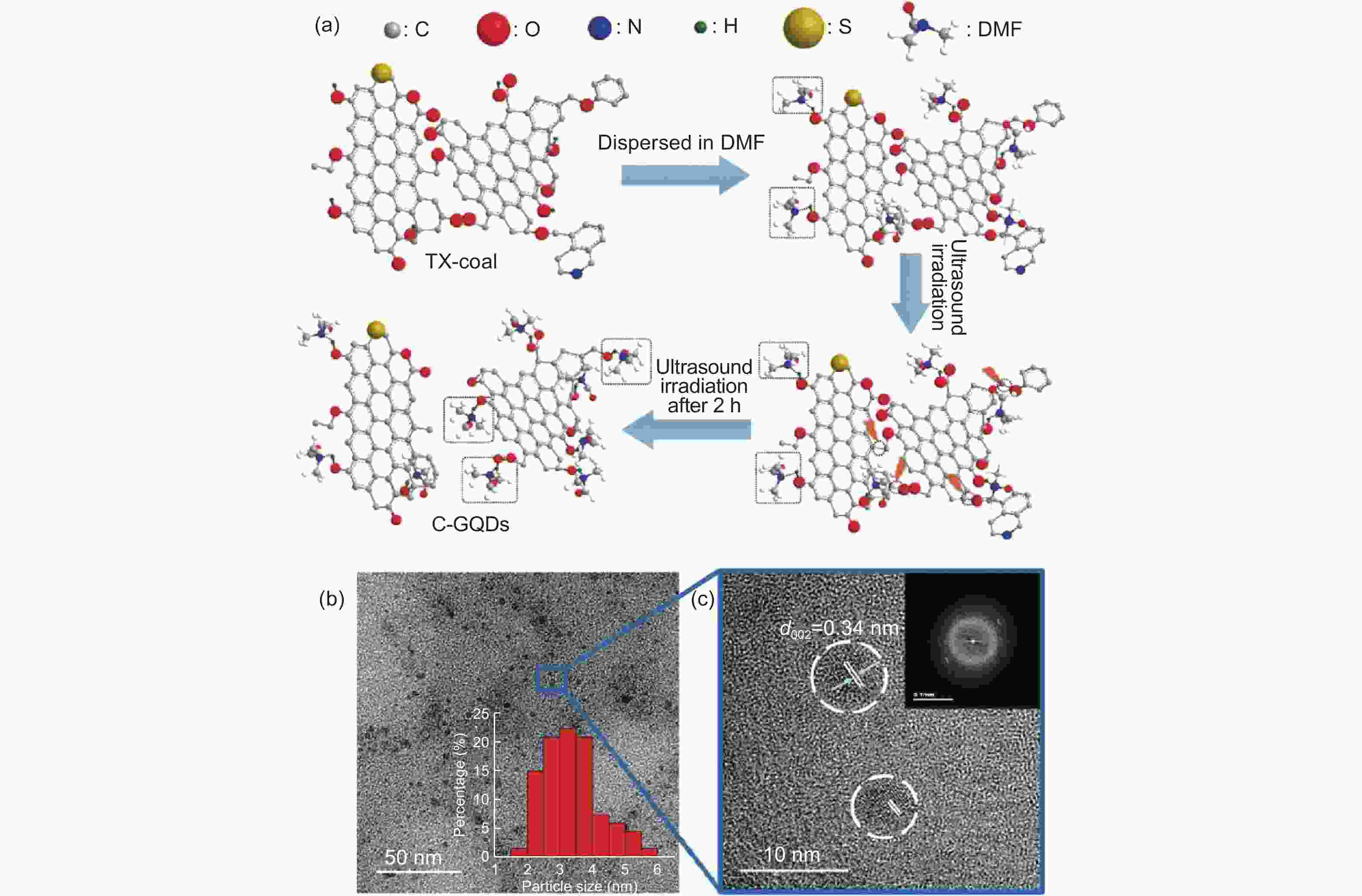
Figure 3. (a) Schematic illustration of C-GQDs synthesis, (b) TEM image of C-GQDs, (c) HRTEM image of C-GQDs. Inset is the FFT pattern of C-GQDs[55]. Reprinted with permission.
Figure 4. TEM images of iron carbide-oxide filled CNTs obtained via CVD of coal-gas: (a) low-resolution TEM image of partly filled CNTs and (b) high-resolution TEM image of one filled CNT[67]. Reprinted with permission.
Figure 5. (a, b, c) FESEM images of CPCFs at low magnification: PVA/Coal = (a) 2/1, (b) 1/1 and (c) 1/2 and (d, e, f) magnified FESEM images of CPCFs: PVA/Coal = (d) 2/1, (e) 1/1 and (f) 1/2[73]. Reprinted with permission.
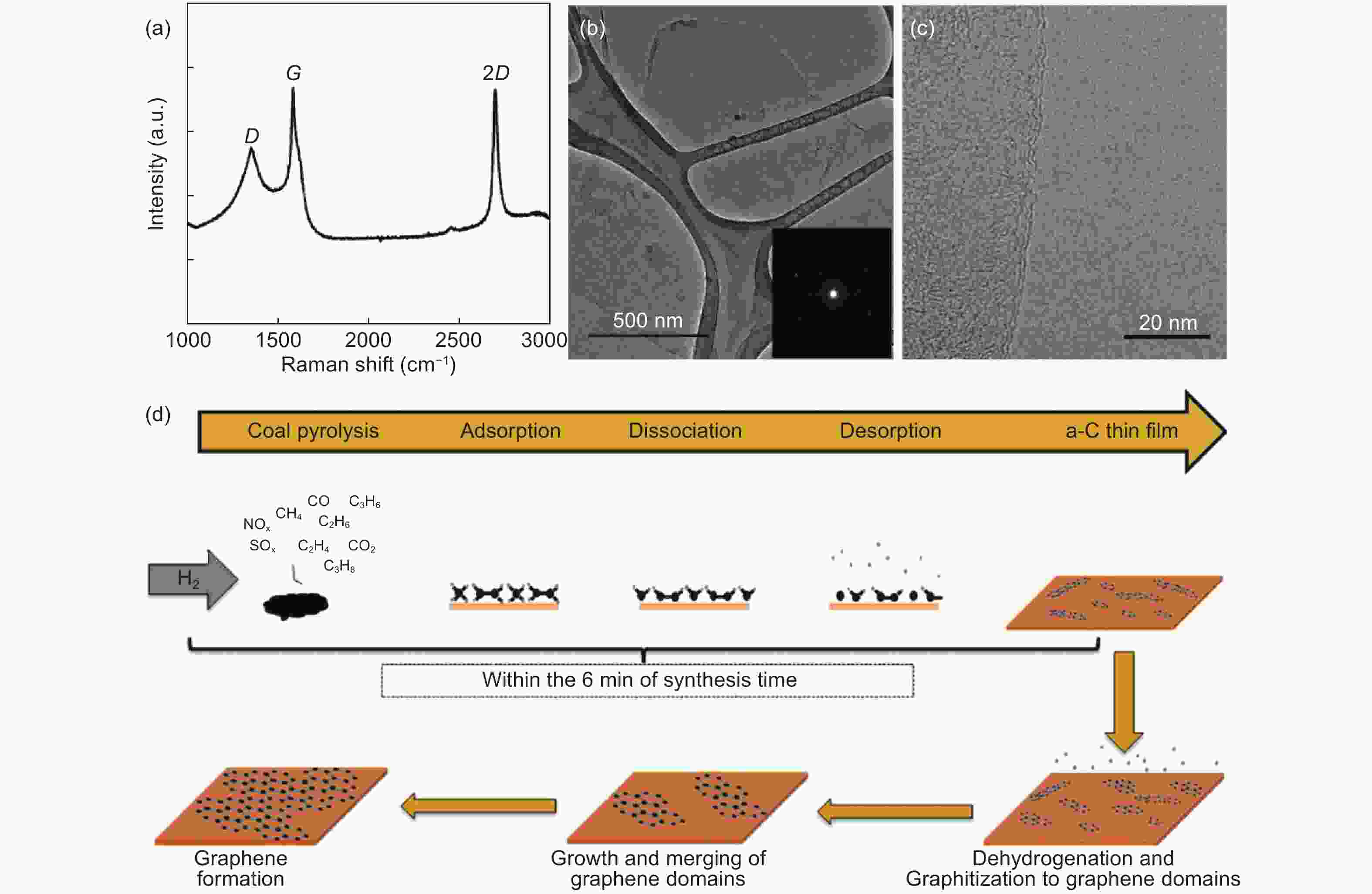
Figure 6. (a) Raman spectra of coal derived graphene film synthesized at 1055 oC for 30 min, (b) Large area of coal derived graphene films on TEM grid, (Inset: SAED with six diffraction spots demonstrating crystalline nature of coal derived graphene film), (c) High magnification TEM image with the edge of the coal derived graphene film and (d) schematic of growth mechanism of coal derived graphene films [78]. Reprinted with permission.
Figure 7. (a, b) Digital photos of samples before and after treatment with H2 discharge plasma, and SEM and TEM images of (c, d, e) TX-NC-GS (graphene sheets obtained from Taixi coal without catalytic graphitization) and (f, g, h) TX-C-GS (graphene sheets obtained from Taixi coal with catalytic graphitization,) with different magnifications[79]. Reprinted with permission.
Figure 8. (a, b) FESEM images of ISPCs and (c, d) TEM images of ISPCs[93]. Reprinted with permission.
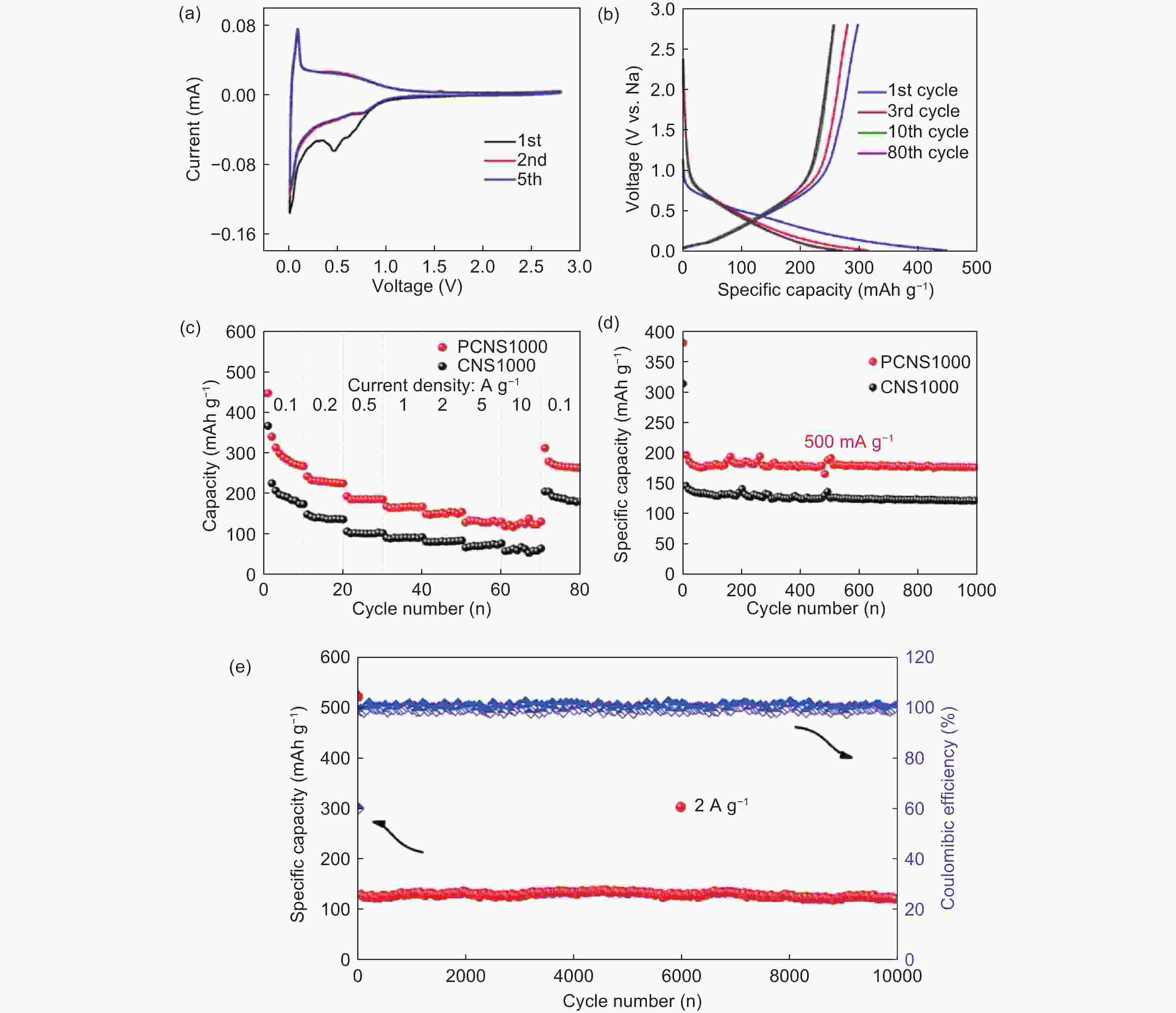
Figure 9. (a) CV curves of PCNS1000 between 0.01 and 2.8 V at 0.1 mV s−1, (b) Galvanostatic discharge-charge profiles of PCNS1000 at 100 mA g−1, (c) Rate capability of the electrodes at different current densities, (d) Cycling performance of the electrodes at 0.5 A g−1 and (e) Long-term cycling stability of PCNS1000 at 2 A g−1[89]. Reprinted with permission.
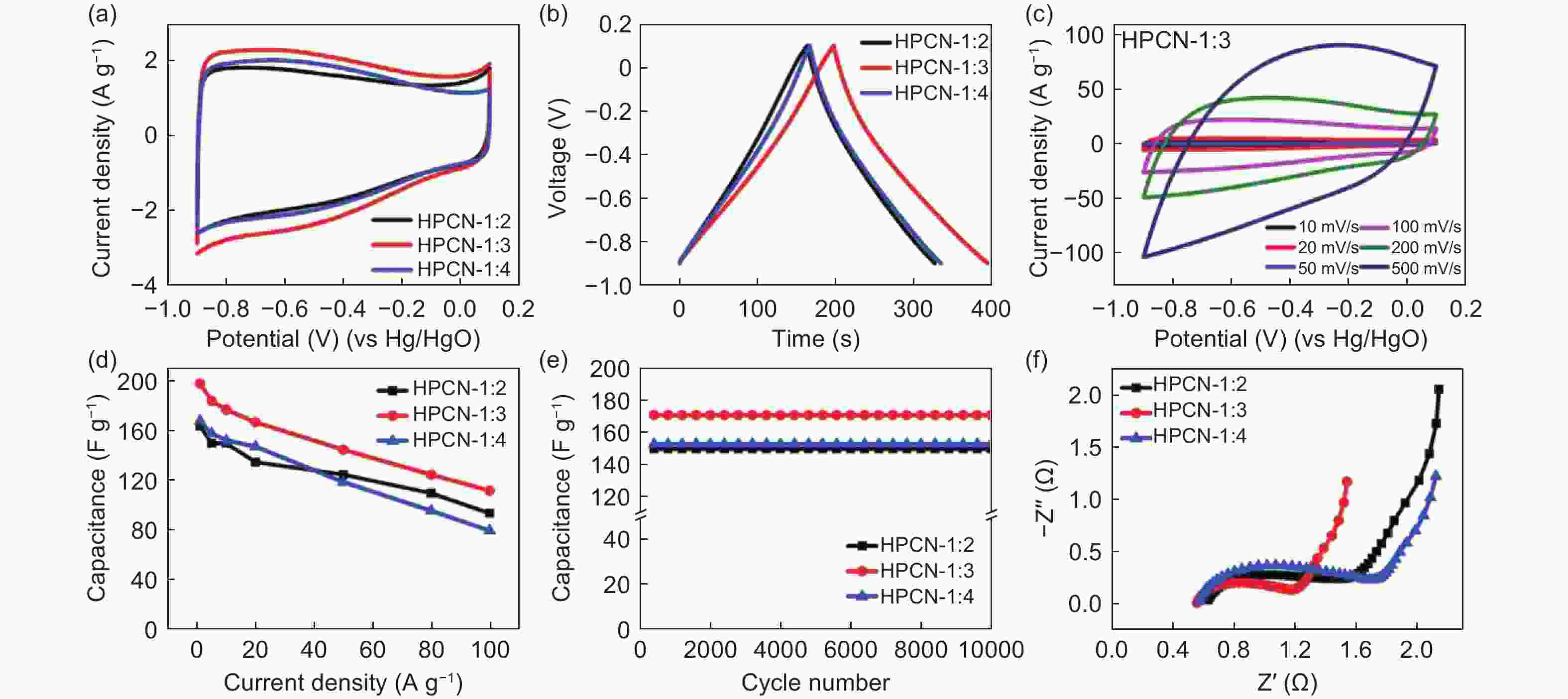
Figure 10. (a) CV curves under the scan rate of 10 mV s−1, (b) Galvanostatic charge-discharge curves under 1 A g−1 of the HPCNs prepared with different template addition ratios, (c) CV curves of the HPCN-1∶3 under different scan rates from 10 to 500 mV s−1, (d) The rate performance, (e) The cycle performance under 10 A g−1, (f) The EIS spectra of the HPCNs prepared with different template addition ratios[105]. Reprinted with permission.
-
[1] Jorge A B, Jervis R, Periasamy A P, et al. 3D carbon materials for efficient oxygen and hydrogen electrocatalysis[J]. Advanced Energy Materials,2019:1902484. [2] Zhang L H, Shi Y M, Wang Y, et al. Nanocarbon catalysts: Recent understanding regarding the active sites[J]. Advanced Science,2020:1902126. [3] Borchardt L, Zhu Q, Casco M E, et al. Toward a molecular design of porous carbon materials[J]. Material Today,2017,20(10):592-610. doi: 10.1016/j.mattod.2017.06.002 [4] Kang B T, Ai H Q, Lee J L. Single-atom vacancy induced changes in electronic and magnetic properties of graphyne[J]. Carbon,2017,116:113-119. doi: 10.1016/j.carbon.2017.01.068 [5] Hou H S, Qiu X Q, Wei W F, et al. Carbon anode materials for advanced sodium-ion batteries[J]. Advanced Energy Materials,2017:1602898. [6] Lv Q, Si W Y, He J J, et al. Selectively nitrogen-doped carbon materials as superior metal-free catalysts for oxygen reduction[J]. Nature Communications,2018,9:3376. doi: 10.1038/s41467-018-05878-y [7] Zhang Y T, Wang P, Yang J, et al. Fabrication of core-shell nanohybrid derived from iron-based metal-organic framework grappled on nitrogen-doped graphene for oxygen reduction reaction[J]. Chemical Engineering Journal,2020,401:126001. doi: 10.1016/j.cej.2020.126001 [8] Hu C, Li M Y, Qiu J S, et al. Design and fabrication of carbon dots for energy conversion and storage[J]. Chemical Society Reviews,2019,48(8):2315-37. doi: 10.1039/C8CS00750K [9] Wang M, Yang J, Liu S Y, et al. Nitrogen-doped hierarchically porous carbon nanosheets derived from polymer/graphene oxide hydrogels for high-performance supercapacitors[J]. Journal of Colloid and Interface Science,2020,560:69-75. doi: 10.1016/j.jcis.2019.10.037 [10] Yang J, Liu S Y, Wang M, et al. Fabrication of porous carbon nanosheets with the engineered graphitic structure for electrochemical supercapacitors[J], Industrial & Engineering Chemistry Research, 2020, DOI: 10.1021/acs.iecr.0c02394. [11] Huang H G, Shen K, Chen F F, et al. Metal-organic frameworks as a good platform for the fabrication of single-atom catalysts[J]. ASC Catalysis,2020,10:6579-6586. [12] Yang J, Jia K L, Wang M, et al. Fabrication of nitrogen-doped porous graphene hybrid nanosheets from metal-organic frameworks for lithium-ion batteries[J]. Nanotechnology,2020,31(14):145402. doi: 10.1088/1361-6528/ab6475 [13] Wang Y L, Qu Q L, Gao S T, et al. Biomass derived carbon as binder-free electrode materials for supercapacitors[J]. Carbon,2019,155:706-726. doi: 10.1016/j.carbon.2019.09.018 [14] He L, Sun Y R, Wang C H, et al. High performance sulphur-doped pitch-based carbon materials as anode materials for sodium-ion batteries[J]. New Carbon Materials,2020,35(4):420-427. doi: 10.1016/S1872-5805(20)60499-1 [15] Wang H, Shao Y, Mei S L, et al. Polymer-derived heteroatom-doped porous carbon materials[J]. Chemical Reviews,2020,120(17):9363-9419. doi: 10.1021/acs.chemrev.0c00080 [16] Liu F, Liu S Y, Meng J S, et al. Stabilizing conversion reaction electrodes by MOF derived N-doped carbon shell for highly reversible lithium storage[J]. Nano Energy,2020,73:104758. [17] Hu H, Zhao Z B, Wan W B, et al. Ultralight and highly compressible graphene aerogels[J]. Advanced Materials,2013,25:2219-2223. doi: 10.1002/adma.201204530 [18] Li H Q, Xiao N, Wang Y W, et al. Promoted electroreduction of CO2 with oxygen vacancies on plasma-activated SnOx/carbon foam monolithic electrode[J]. Journal of Materials Chemistry A,2020,8:1779-1786. doi: 10.1039/C9TA12401B [19] Qin Z H. New advances in coal structure model[J]. International Journal of Mining Science and Technology,2018,28(4):541-559. doi: 10.1016/j.ijmst.2018.06.010 [20] Ma X M, Dong X S, Fan Y P. Prediction and characterization of the microcrystal structures of coal with molecular simulation[J]. Energy & Fuels,2018,32(2):3097-3107. [21] Mathews J P, Chaffee A L. The molecular representations of coal-A review[J]. Fuel,2012,96:1-14. doi: 10.1016/j.fuel.2011.11.025 [22] Vasireddy S, Morreale B, Cugini A, et al. Clean liquid fuels from direct coal liquefaction: chemistry, catalysis, technological status and challenges[J]. Energy & Environmental Science,2011,4(2):311-345. [23] Haenel M W. Recent progress in coal structure research[J]. Fuel,1992,71(11):1211-1223. doi: 10.1016/0016-2361(92)90046-Q [24] Pang L S K, Vassallo A M, Wilson M A. Fullerenes from coal[J]. Nature,1991,352(6335):480-480. [25] Xu G Z, Yang T, Fang Z G, et al. Preparation and characterization of coal-based carbon foams by microwave heating process under ambient pressure[J]. Diamond & Related Materials,2018,86:63-70. [26] Nilewski L, Mendoza K, Jalilov A S, et al. Highly oxidized graphene quantum dots from coal as efficient antioxidants[J]. ACS Applied Materials & Interfaces,2019,11(18):16815-16821. doi: 10.1016/j.apsusc.2018.03.156 [27] Moothi K, Iyuke S E, Meyyappan M, et al. Coal as a carbon source for carbon nanotube synthesis[J]. Carbon,2012,50:2679-2690. doi: 10.1016/j.carbon.2012.02.048 [28] Sakurovs R, Koval L, Grigore M, et al. Nanometre-sized pores in coal: Variations between coal basins and coal origin[J]. International Journal of Coal Geology,2018,186:126-134. doi: 10.1016/j.coal.2017.11.010 [29] Klein J, Pfeifer F, Schacht, Sinder Ch. Environmental aspects of bioconversion processes[J]. Fuel Processing Technology,1997,52:17-25. doi: 10.1016/S0378-3820(97)00012-X [30] Zhang Y T, Zhang X Q, Liu G Y, et al. Graphitization of Shenfu coal[J]. CIESC Journal,2015,66(4):1514-1520. [31] Inagaki M, Toyoda M, Soneda Y, et al. Nitrogen-doped carbon materials[J]. Carbon,2018,132:104-140. doi: 10.1016/j.carbon.2018.02.024 [32] Kroto H W, Heath J R, Brien S C O, et al. C60: Buckminsterfullerene[J]. Nature,1985,318(14):162-163. [33] Bloodworth S, Sitinova G, Alom S, et al. First synthesis and characterization of CH4@C60[J]. Angewandte Chemie International Edition,2019,58(15):5038-5043. doi: 10.1002/anie.201900983 [34] Coro J, Suárez M, Silva L S. R., et al. Fullerene applications in fuel cells: A review[J]. International Journal of Hydrogen Energy,2016,41(40):17944-17959. doi: 10.1016/j.ijhydene.2016.08.043 [35] Weston A, Murthy M, Lalvani S. Synthesis of fullerenes from coal[J]. Fuel Processing Technology,1995,45(3):203-212. doi: 10.1016/0378-3820(95)00042-6 [36] QIU Jie-shan, ZHOU Ying, WANG Lin-na, et al. Preparation of fullerenes from coals[J]. Journal of Dalian University of Technology,2000,40(S1):41-45. [37] QIU Jie-shan, LUO Chang-qi, GUO Shu-cai. Feasibility for developing advanced carbon materials from coals[J]. Chemical Industry and Engineering Progress,1995,4:15-22. doi: 10.3321/j.issn:1000-6613.1995.02.003 [38] WU Hao, QIAN Wei, YU Xiu-ling, et al. Research progress in the preparation of fullerenes[J]. Environment and Sustainable Development,2010,35(4):34-37. doi: 10.3969/j.issn.1673-288X.2010.04.012 [39] QIU Jie-shan, ZHOU Ying, ZHANG Hong-bin, et al. Preparation and purification of coal-based fullerene (C60)[J]. CIESC Journal,1997(02):252-256. doi: 10.3321/j.issn:0438-1157.1997.02.020 [40] WANG Xiao-min, WANG Hai-ying, ZHANG Hai-xia, et al. HREM analysis of onion-like fullerenes prepared from coal[J]. Journal of Chinese Electron Microscopy Society,2004(02):159-162. doi: 10.3969/j.issn.1000-6281.2004.02.014 [41] Ugarte Daniel. Formation mechanism of quasi-spherical carbon particles induced by electron bombardment[J]. Chemical Physics Letters,1993,207:4-6. [42] LIU Xu-guang, DU Ai-bin, FU Dong-ju, et al. Study on the preparation of onion-like fullerenes from Chinese coal[J]. Transactions of Materials and Heat Treatment,2005(03):31-33+105. [43] GUO Ming-cong, YANG Yong-zhen, REN Wen-fang, et al. In-situ modification of onion-like fullerenes in oxygen radio frequency plasma[J]. Coal Conversion,2009,32(02):1-4. doi: 10.3969/j.issn.1004-4248.2009.02.001 [44] Hu C, Mu Y, Li M Y, et al. Recent advances in the synthesis and applications of carbon dots[J]. Acta Phys -Chim Sin,2019,35(6):572-590. doi: 10.3866/PKU.WHXB201806060 [45] Ye R Q, Peng Z W, Metzger A, et al. Bandgap engineering of coal-derived graphene quantum dots[J]. ACS Applied Materials & Interfaces,2015,7(12):7041-7048. [46] Ye R Q, Xiang C S, Lin J, et al. Coal as an abundant source of graphene quantum dots[J]. Nature Communications,2013,4(1):3546-3558. [47] Hu C, Yu C, Li M Y, et al. Chemically tailoring coal to fluorescent carbon dots with tuned size and their capacity for Cu(II) detection[J]. Small,2015,10(23):4926-4933. [48] Geng B J, Yang D W, Zheng F F, et al. Facile conversion of coal tar to orange fluorescent carbon quantum dots and their composite encapsulated by liposomes for bioimaging[J]. New Journal of Chemistry,2017(41):14444-14451. [49] Xu Y, Wang S G, Hou X Y, et al. Coal-derived nitrogen, phosphorus and sulfur co-doped graphene quantum dots: A promising ion fluorescent probe[J]. Applied Surface Science,2018(445):519-526. [50] Hu S L, Wei Z J, Chang Q, et al. A facile and green method towards coal-based fluorescent carbon dots with photocatalytic activity[J]. Applied Surface Science,2016(378):402-407. [51] Das T, Saikia Binoy K, Dekaboruah H P, et al. Blue-fluorescent and biocompatible carbon dots derived from abundant low-quality coals[J]. Journal of photochemistry and photobiology. B, Biology,2019(195):1-11. [52] Meng X, Chang Q, Xue C R, et al. Full-colour carbon dots: from energy-efficient synthesis to concentration-dependent photoluminescence properties[J]. Chemical Communications,2017(53):3074-3077. [53] Kundu N, Bhunia P, Sarkar S, et al. Highly fluorescent carbon dots from quinoline insoluble residues in coal tar[J]. Optical Materials,2020,100:109638. doi: 10.1016/j.optmat.2019.109638 [54] Liu X X, Hao J Y, Liu J H, et al. Green synthesis of carbon quantum dots from lignite coal and the application in Fe3+ detection[J]. IOP Conference Series: Earth and Environmental Science,2018,113:012063. doi: 10.1088/1755-1315/113/1/012063 [55] Zhang Y T, Li K K, Ren S Z, et al. Coal-derived graphene quantum dots produced by ultrasonic physical tailoring and their capacity for Cu(II) detection[J]. ACS Sustainable Chemistry & Engineering,2019,7:9793-9799. [56] Li M Y, Yu C, Hu C, et al. Solvothermal conversion of coal into nitrogen-doped carbon dots with singlet oxygen generation and high quantum yield[J]. Chemical Engineering Journal,2017(320):570-575. [57] Cheng Y, Li P, Zhang Q B, et al. Top-down fabrication of small carbon nanotubes[J]. Nanoscale Horizons,2019,4:1310-1317. doi: 10.1039/C9NH00285E [58] Iijima S. Helical microtubules of graphite carbon[J]. Nature,1991,354(6438):56-58. [59] Farrera C, Andon, F T, Feliu N. Carbon nanotubes as optical sensors in biomedicine[J]. ACS Nano,2017,11:10637-10643. doi: 10.1021/acsnano.7b06701 [60] Shandakov S D, Kosobutsky A V, Rybakov M S, et al. Effect of gaseous and condensate products of ethanol decomposition on aerosol CVD synthesis of single-walled carbon nanotubes[J]. Carbon,2018,126:522-531. doi: 10.1016/j.carbon.2017.10.064 [61] Williams K A, Tachibana M, Allen J L, et al. Single-wall carbon nanotubes from coal[J]. Chemical Physics Letters,1999,310(1):31-37. [62] Qiu J S, Li Y F, Wang Y P, et al. High-purity single-wall carbon nanotubes synthesized from coal by arc discharge[J]. Carbon,2003,41(11):2159-2179. doi: 10.1016/S0008-6223(03)00215-X [63] Qiu J S, An Y L, Zhao Z B, et al. Catalytic synthesis of single-walled carbon nanotubes from coal gas by chemical vapor deposition method[J]. Fuel Processing Technology,2003,85(8):913-920. [64] Qiu J S, Li Y F, Wang Y P, et al. Production of carbon nanotubes from coal[J]. Fuel Processing Technology,2004,85:1663-1670. doi: 10.1016/j.fuproc.2003.12.010 [65] Qiu J S, Wang Z Y, Zhao Z B, et al. Synthesis of double-walled carbon nanotubes from coal in hydrogen-free atmosphere[J]. Fuel,2006,86(1):282-286. [66] Qiu J S, Zhang F, Zhou Y, et al. Carbon nanomaterials from eleven caking coals[J]. Fuel,2002,81:1509-1514. doi: 10.1016/S0016-2361(02)00069-8 [67] Qiu J S, Li Q X, Wang Z Y, et al. CVD synthesis of coal-gas-derived carbon nanotubes and nanocapsules containing magnetic iron carbide and oxide[J]. Carbon,2006,44(12):2565-2568. doi: 10.1016/j.carbon.2006.05.030 [68] Tian Y J, Zhang Y L, Wang B J, et al. Coal-derived carbon nanotubes by thermal plasma jet[J]. Carbon,2004,42(12):2597-2601. [69] Yadav D, Amini Fedi, Ehrmann A. Recent advances in carbon nanofibers and their applications-A review[J]. European Polymer Journal,2020,138:109963. doi: 10.1016/j.eurpolymj.2020.109963 [70] Lu W Y, He T, Xu B, et al. Progress in catalytic synthesis of advanced carbon nanofibers[J]. Journal of Materials Chemistry A,2017,5:13863-13881. doi: 10.1039/C7TA02007D [71] Yue L C, Zhao H T, Wu Z G, et al. Recent advances in electrospun one-dimensional carbon nanofibers structures/heterostructures as anode materials for sodium ion batteries[J]. Journal of Materials Chemistry A,2020,8:11493-11510. doi: 10.1039/D0TA03963B [72] He Y T, Wang L X, Jia D Z. Coal/PAN interconnected carbon nanofibers with excellent energy storage performance and electrical conductivity[J]. Electrochimica Acta,2016,194:239-245. doi: 10.1016/j.electacta.2016.01.191 [73] Guo M X, Guo J X, Jia D Z, et al. Coal derived porous carbon fibers with tunable internal channels for flexible electrodes and organic matter absorption[J]. Journal of Materials Chemistry A,2015,3:21178-21184. doi: 10.1039/C5TA05743D [74] Zhao X F, Qiu J S, Sun Y C, et al. Preparation of coal-based carbon nanofibers and open bamboo-shaped carbon nanotubes by arc discharge method[J]. New Carbon Materials,2009,24(02):109-113. [75] Inagaki M, Kang F Y. Materials Science and Engineering of Carbon: Fundamentals[M]. (Second edition), Beijing: Tsinghua University Press, 2014. [76] Olabi A. G., Abdelkareem M A, Wilberforce T, et al. Application of graphene in energy storage device-A review[J]. Renewable and Sustainable Energy Reviews,2021,135:110026. doi: 10.1016/j.rser.2020.110026 [77] Wang D, Botte G G, Vijapur S H. Raw coal derived large area and transparent graphene films[J]. ECS Solid State Letters,2013,2(7):M45-M47. doi: 10.1149/2.005307ssl [78] Vijapur S H, Wang D, Ingram D C, et al. An investigation of growth mechanism of coal derived graphene films[J]. Materials Today Communications,2017,11:147-155. doi: 10.1016/j.mtcomm.2017.04.003 [79] Zhou Q, Zhao Z B, Qiu J S, et al. Graphene sheets from graphitized anthracite coal: preparation, decoration, and application[J]. Energy & Fuels,2012,26:5186-5192. [80] Bhaviripudi S, Jia X T, Dresselhaus M S, et al. Role of kinetic factors in chemical vapor deposition synthesis of uniform large area graphene using copper catalyst[J]. Nano Letters,2010,10(10):4128-4133. doi: 10.1021/nl102355e [81] ZHANG Ya-ting. Preparation, modification, and application of coal-based-graphene[D]. Xi'an University of science and technology, 2015. [82] Sierra U, Álvarez P, Blanco C, et al. Cokes of different origin as precursors of graphene oxide[J]. Fuel,2016,166:400-403. doi: 10.1016/j.fuel.2015.10.112 [83] He X J, Ma H, Wang J X, et al. Porous carbon nanosheets from coal tar for high-performance supercapacitors[J]. Journal of Power Sources,2017,357:41-46. doi: 10.1016/j.jpowsour.2017.04.108 [84] Pakhira B, Ghosh S, Maity S, et al. Extraction of preformed graphene oxide from coal: its clenched fist form entrapping large molecules[J]. RSC Advances,2013,5:89076-89082. [85] Xie L L, Jin Z H, Dai Z D, et al. Porous carbons synthesized by templating approach from fluid precursors and their applications in environment and energy storage: A review[J]. Carbon,2020,170:100-118. doi: 10.1016/j.carbon.2020.07.034 [86] Huang G X, Kang W W, Xing B L, et al. Oxygen-rich and hierarchical porous carbons prepared from coal based humic acid for supercapacitor electrodes[J]. Fuel Processing Technology,2016,142:1-5. doi: 10.1016/j.fuproc.2015.09.025 [87] Teng H S, Yeh T S. Preparation of activated carbons from bituminous coals with zinc chloride activation[J]. Industrial & Engineering Chemistry Research,1998,37(1):58-65. [88] Wang Y W, Xiao N, Wang Z Y, et al. Rational design of high-performance sodium-ion battery anode by molecular engineering of coal tar pitch[J]. Chemical Engineering Journal, 2018, 342: 52-60. [89] Hao M Y, Xiao N, Wang Y W, et al. Pitch-derived N-doped porous carbon nanosheets with expanded interlayer distance as high-performance sodium-ion battery anodes[J]. Fuel Processing Technology,2018,177:328-335. doi: 10.1016/j.fuproc.2018.05.007 [90] XIAO Nan, QIU Jieshan. Progress in synthesis and applications of functional carbon materials from coal tar pitch[J]. Chemical Industry and Engineering Progress,2016,35(6):1804-1811. [91] He X J, Li X J, Wang J X, et al. From diverse polycyclic aromatic molecules to interconnected graphene nanocapsules for supercapacitors[J]. Microporous and Mesoporous Materials,2017,245:73-81. doi: 10.1016/j.micromeso.2017.02.078 [92] Xie X Y, He X J, Shao X L, et al. Synthesis of layered microporous carbons from coal tar by directing, space-confinement and self-sacrificed template strategy for supercapacitors[J]. Electrochimica Acta,2017,246:634-642. doi: 10.1016/j.electacta.2017.06.092 [93] Xie X Y, He X J, Zhang H F, et al. Interconnected sheet-like porous carbons from coal tar by a confined soft-template strategy for supercapacitors[J]. Chemical Engineering Journal,2018,350:49-56. doi: 10.1016/j.cej.2018.05.011 [94] Gao S S, Tang Y K, Wang L, et al. Coal-based hierarchical porous carbon synthesized with a soluble salt self-assembly-assisted method for high performance supercapacitors and Li-ion batteries[J]. ACS Sustainable Chemisrty & Engineering,2018,6(3):3255-3263. [95] Xie J, Lu Y C. A retrospective on lithium-ion batteries[J]. Nature Communications,2020,11:2499. doi: 10.1038/s41467-020-16259-9 [96] Yuan Y, Chen Z W, Yu H X, et al. Heteroatom-doped carbon-based materials for lithium and sodium ion batteries[J]. Energy Storage Materials,2020,32:65-90. doi: 10.1016/j.ensm.2020.07.027 [97] Zhao X J, Jia W, Wu X Y, et al. Ultrafine MoO3 anchored in coal-based carbon nanofibers as anode for advanced lithium-ion batteries[J]. Carbon,2020,156:445-452. doi: 10.1016/j.carbon.2019.09.065 [98] Zhang Y T, Zhang K B, Jia K L, et al. Preparation of coal-based graphene quantum dots/α-Fe2O3 nanocomposites and their lithium-ion storage properties[J]. Fuel,2019,241:646-653. doi: 10.1016/j.fuel.2018.12.030 [99] Zhang Y T, Zhang K B, Ren S Z, et al. 3D nanoflower-like composite anode of α-Fe2O3/coal-based graphene for lithium-ion batteries[J]. Journal of Alloys and Compounds,2019,794:828-834. [100] Borenstein A, Hanna O, Attias R, et al. Carbon-based composite materials for supercapacitor electrodes: a review[J]. Journal of Materials Chemistry A,2017,5:12653-12672. doi: 10.1039/C7TA00863E [101] Pei T, Sun F, Gao J H, et al. Introducing catalytic gasification into chemical activation for the conversion of natural coal into hierarchically porous carbons with broadened pore size for enhanced supercapacitive utilization[J]. RSC Advances,2018,8:37880-37889. doi: 10.1039/C8RA07308B [102] Gao F, Qu J Y, Zhao Z B, et al. A green strategy for the synthesis of graphene supported Mn3O4 nanocomposites from graphitized coal and their supercapacitor application[J]. Carbon,2014,80:640-650. doi: 10.1016/j.carbon.2014.09.008 [103] He X J, Li X J, Ma H, et al. ZnO template strategy for the synthesis of 3D interconnected graphene nanocapsules from coal tar pitch as supercapacitor electrode materials[J]. Journal of Power Sources,2017,340:183-191. doi: 10.1016/j.jpowsour.2016.11.073 [104] ZHANG Yating, REN Shaozhao, LI Jinkai, et al. Fabrication and electrochemical capacitive performance of PANI/coal-based three-dimensional graphene[J]. CIESC Journal,2017,68(11):4316-4322. [105] Zhang S, Zhu J Y, Qing Y, et al. Construction of hierarchical porous carbon nanosheets from template-assisted assembly of coal-based graphene quantum dots for high performance supercapacitor electrodes[J]. Materials Today Energy,2017,6:36-45. doi: 10.1016/j.mtener.2017.08.003 [106] GUO Ji-xi, SONG Xian-li, GUO Ming-xi, et al. Preparation of MnO2/CBCNFs composite materials and its electrochemical performance for flexible supercapacitor[J]. Chemistry,2016,79(10):942-951. -






 下载:
下载: