Preparation and electrochemical properties of novel silicon-carbon composite anode materials with a core-shell structure
-
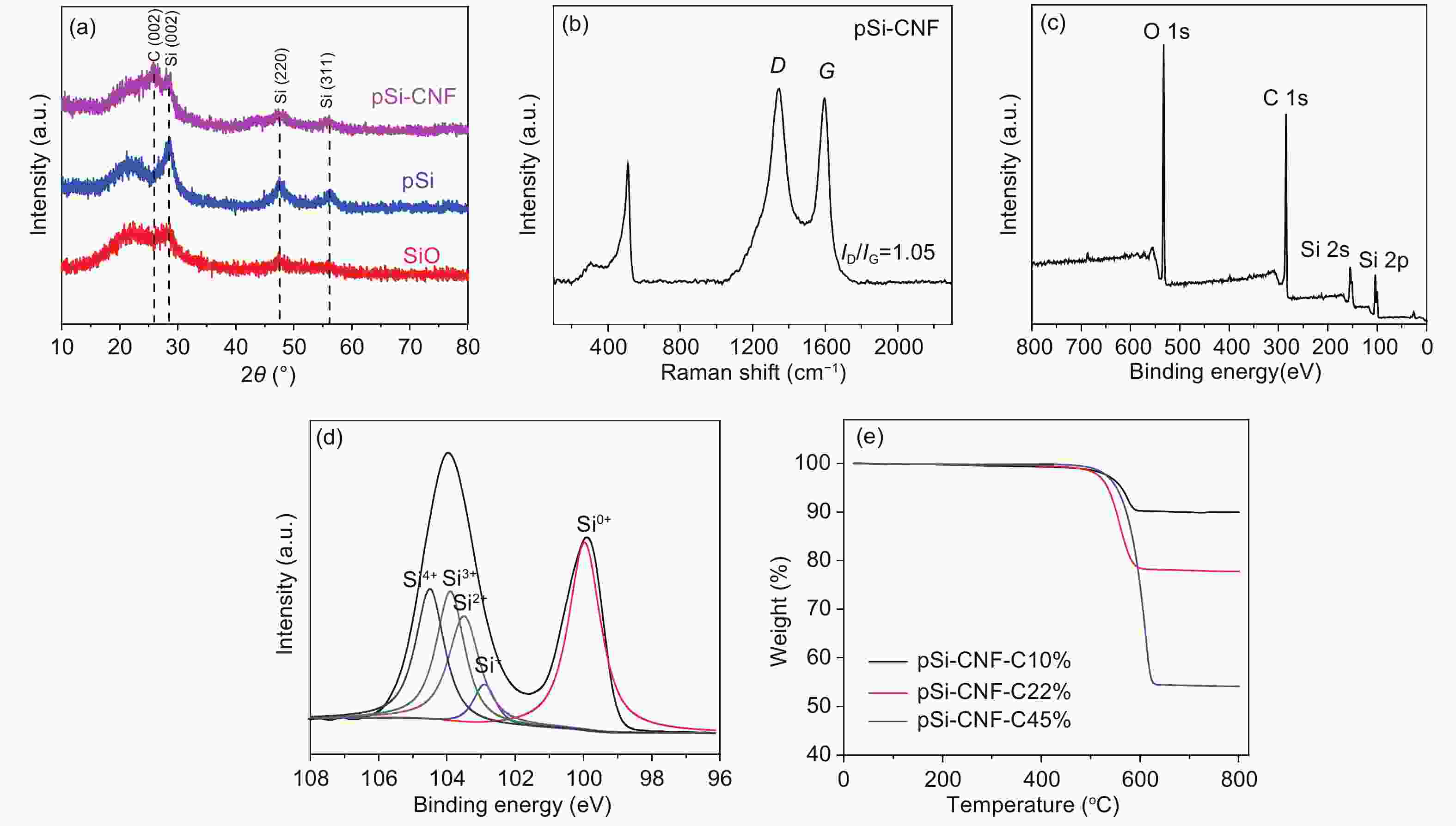
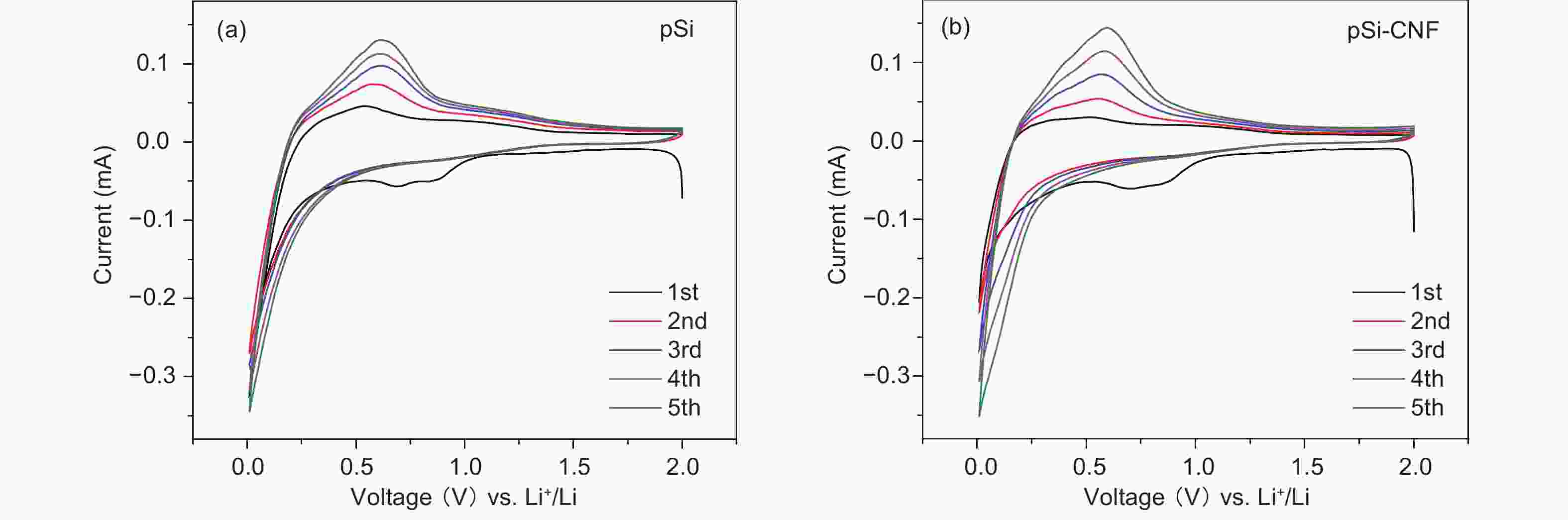
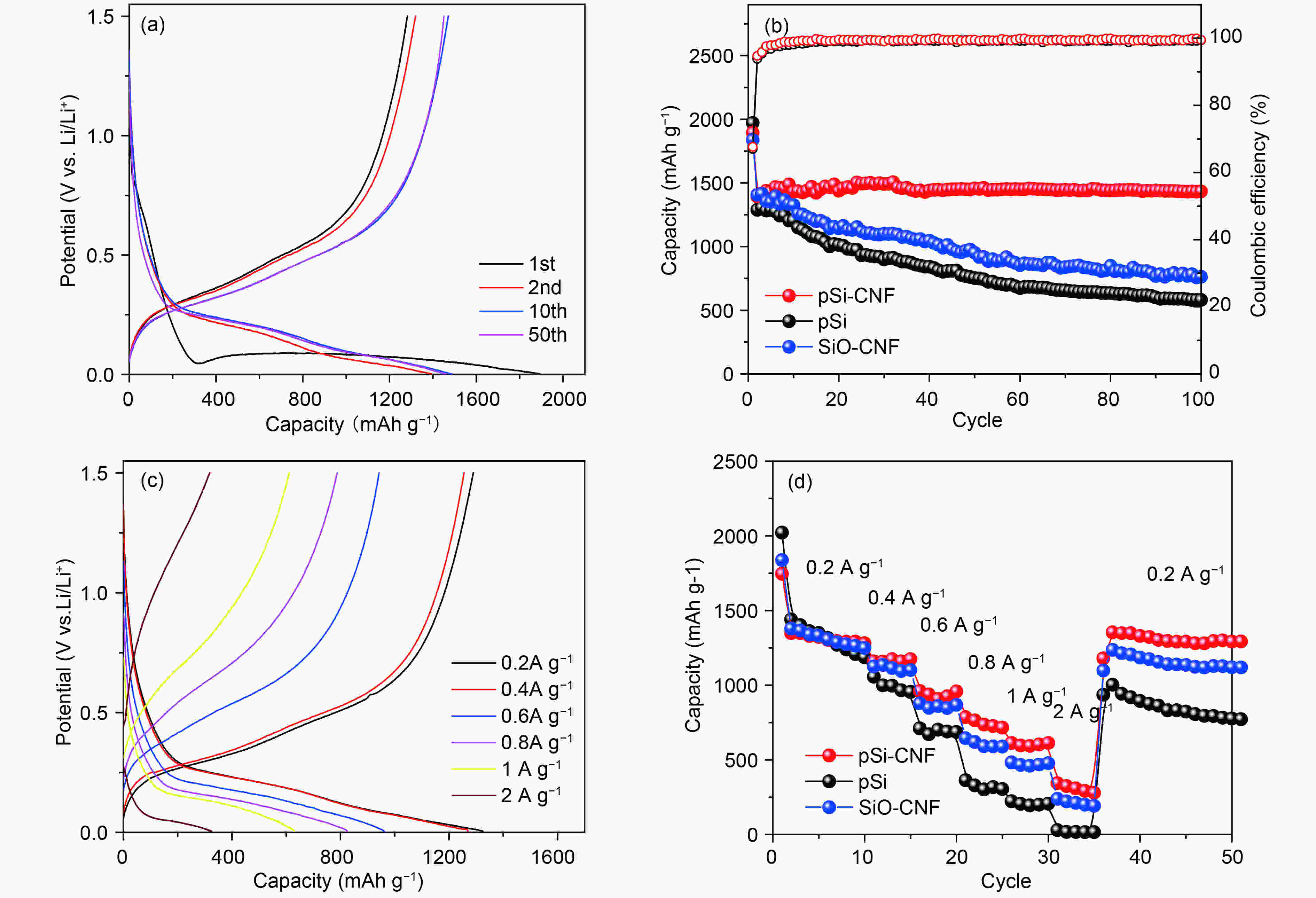
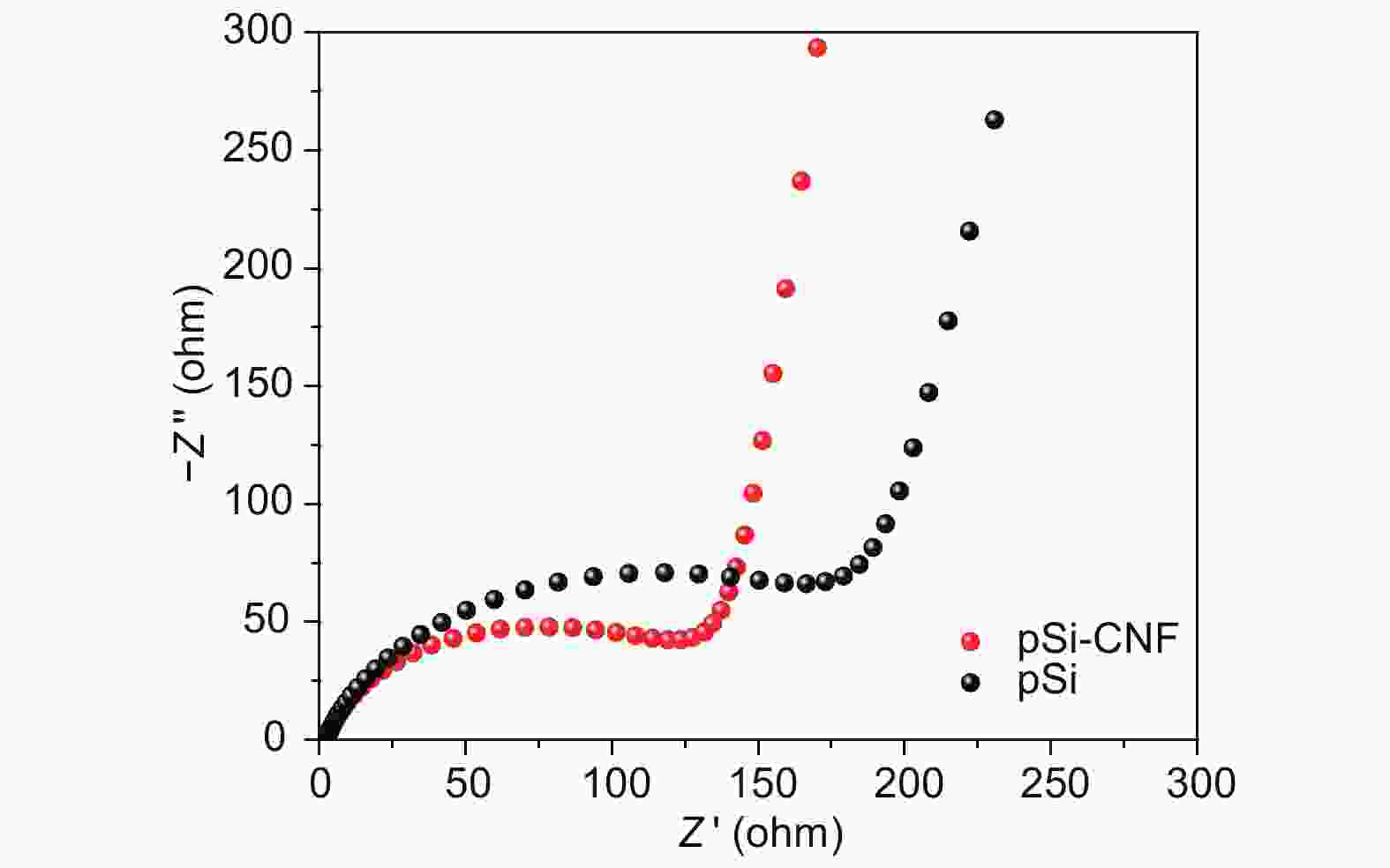
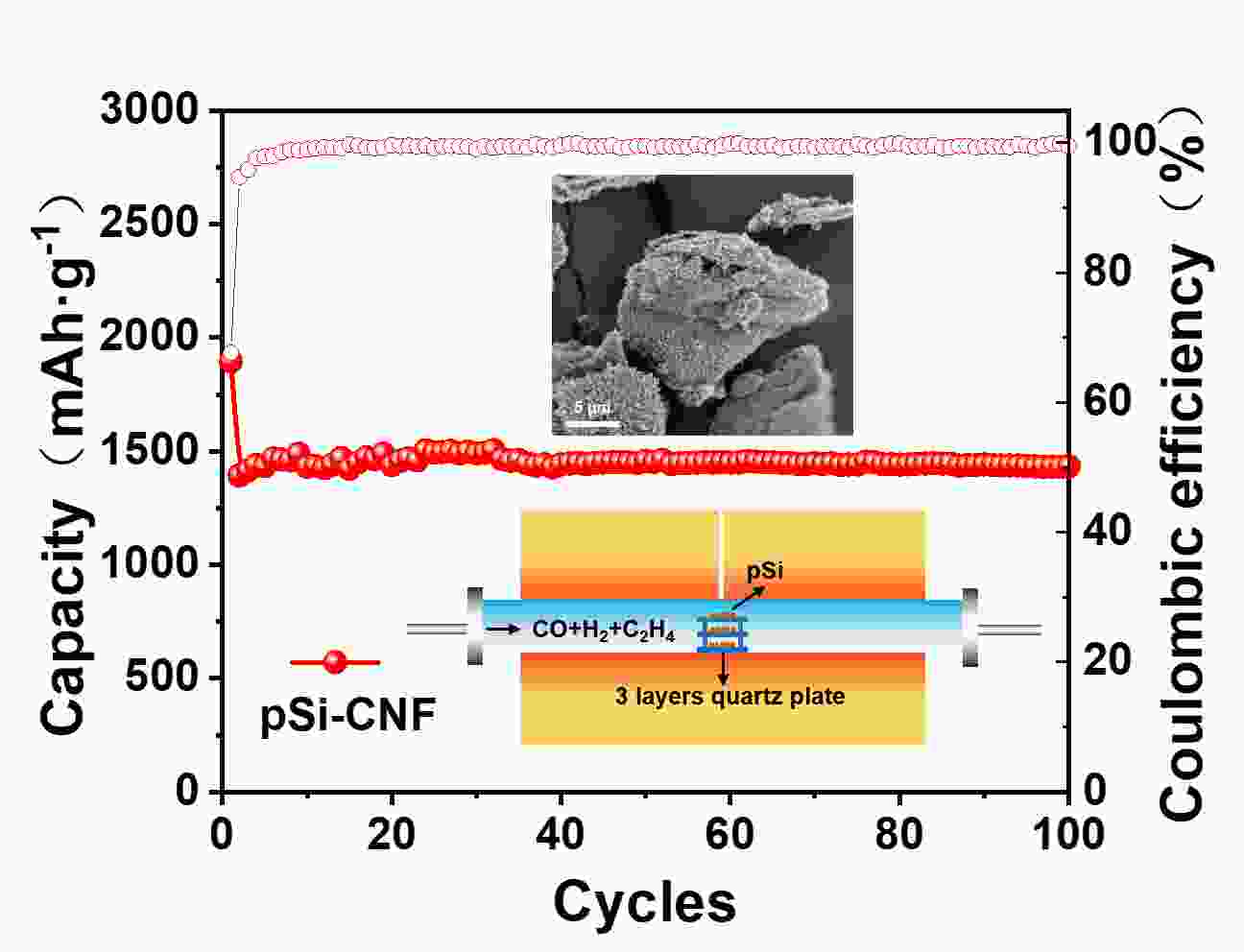
摘要: 通过对氧化硅预处理得到多组分硅pSi(Si、SiO、SiO2),再利用化学气相沉积法(CVD)设计了具有核壳结构的pSi与碳纳米纤维(CNF)的复合材料(pSi-CNF)。多组分硅中Si、SiO提供电化学可逆容量,SiO2可以抑制硅的体积膨胀;碳纳米纤维包覆形成的壳层结构可以有效提高复合材料的导电性,同时进一步抑制硅的体积膨胀保持核壳结构的完整。通过SEM、TEM、EDS、XRD、Raman和XPS对复合物的微观结构进行分析。结果表明:pSi-CNF的粒径为5~20 µm,碳纳米纤维的直径为5~40 nm, pSi-CNF复合材料中含有Si、SiO和SiO2多种组分硅,有明显特征峰;碳纳米纤维均匀包覆于硅表面,形成核壳结构。电化学性能测试表明,在0.2 A·g−1的电流密度下,经100次循环后其可逆容量为1 411 mAh·g−1,容量保持率为74%,具有良好的循环稳定性和较高的可逆容量;在1 A·g−1的电流密度下,经300次循环后其可逆容量为735 mAh·g−1,容量保持率为86%,且具有良好的倍率性能。Abstract: Multi-component porous Si-SiOx (pSi) consisting of Si, SiO and SiO2 was formed by the pretreatment of SiO at 950 °C for 3 h in an inert atmosphere (He) using a disproportionation reaction. Hybrids of pSi and carbon nanofibers (pSi-CNFs) with a core-shell structure were prepared by catalytic chemical vapor deposition (CVD) using Fe-Ni species as the catalyst and a mixture of CO/H2/C2H4 (volumetric ratio 3∶1∶1) as the reactant for 0.5, 1 and 2 h, and were characterized by SEM, TEM, EDS, XRD, Raman spectroscopy and XPS. Results indicate that the pSi-CNF particle sizes are 5−20 μ m with the diameters of the CNFs being 5−40 nm. The CNFs are uniformly coated on the surface of the pSi to form a core-shell structure. Electrochemical performance testing shows that the reversible capacity of the pSi-CNF (0.5 h) remains at 1 411 mAh.g−1 and the capacity retention is 74% after 100 cycles at a current density of 0.2 A.g−1. The reversible capacity remains at 735 mAh.g−1 at a current density of 1 A g−1 after 300 cycles with a capacity retention of 86%. In the pSi, Si and SiO provide the electrochemical reversible capacity. The core-shell structure with the CNF coating effectively improves the conductivity of the composites, and also inhibits the volume expansion of silicon to maintain the integrity of the core shell structure.
-
Key words:
- Silicon carbon composite /
- Anode material /
- Chemical vapor deposition /
- Carbon nanofiber
-
Table 1. Comparison of electrochemical performance between pSi-CNF materials and other silicon-carbon composite anode materials.
Categories of Si Synthesis method Cycling stability Specific capacity (mAh g−1) Cycle number Current/rate Ref. Micrometer-sized Si Si@SiO2 cluster formation and etching 1160 1000 0.5 C [27] Simple Si/C composite Pyrolysis of polymers with Si 1200 30 0.1 C [28] Yolk-shell Si/C SiO2 and carbon coating 1500 1000 1 C [29] Si/graphene Freeze-drying 840 300 1.4 A g−1 [30] Si/CNT Growth of CNT on substrate and sputtering of Si 2500 100 0.2 C [31] pSi-CNF (this work) Disproportionation and CVD 1411 100 0.2 A g−1 -
[1] Shen X H, Tian Z Y, Fan R J, et al. Research progress on silicon/carbon composite anode materials for lithium-ion battery[J]. Journal of Energy Chemistry,2018,27(4):1067-1090. doi: 10.1016/j.jechem.2017.12.012 [2] Li C, Zhang H P, Fu L J, et al. Cathode materials modified by surface coating for lithium ion batteries[J]. Electrochimica Acta,2006,51(19):3872-3883. doi: 10.1016/j.electacta.2005.11.015 [3] Sun X L, Qin X J, Bu L M, et al. Research progress on carbon anode materials for lithium ion batteries[J]. Nonferrous Metals,2011,63(2):147-151. [4] Aurbach D, Zinigrad E, Cohen Y, et al. A short review of failure mechanisms of lithium metal and lithiated graphite anodes in liquid electrolyte solutions[J]. Solid State Ionics,2002,148(3-4):405-416. doi: 10.1016/S0167-2738(02)00080-2 [5] Li Y M, Xu S Y, Wu X Y, et al. Amorphous monodispersed hard carbon micro-spherules derived from biomass as a high performance negative electrode material for sodium-ion batteries[J]. Journal of Materials Chemistry A,2015,3(1):71-77. doi: 10.1039/C4TA05451B [6] Ren X X, Xu S D, Liu S B, et al. Lath-shaped biomass derived hard carbon as anode materials with super rate capability for sodium-ion batteries[J]. Journal of Electroanalytical Chemistry,2019,841:63-72. doi: 10.1016/j.jelechem.2019.04.033 [7] Casimir A, Zhang H G, Ogoke O, et al. Silicon-based anodes for lithium-ion batteries: Effectiveness of materials synthesis and electrode preparation[J]. Nano Energy,2016,27:359-376. doi: 10.1016/j.nanoen.2016.07.023 [8] Kim H, Lee E J, Sun Y K. Recent advances in the Si-based nanocomposite materials as high capacity anode materials for lithium ion batteries[J]. Materials Today,2014,17(6):285-297. doi: 10.1016/j.mattod.2014.05.003 [9] Liu N, Lu Z, Zhao J, et al. A pomegranate-inspired nanoscale design for large-volume-change lithium battery anodes[J]. Nature Nanotechnology,2014,9(3):187-192. doi: 10.1038/nnano.2014.6 [10] Park H, Choi S, Lee S J, et al. Design of an ultra-durable silicon-based battery anode material with exceptional high-temperature cycling stability[J]. Nano Energy,2016,26:192-199. doi: 10.1016/j.nanoen.2016.05.030 [11] Ji L, Zhou W, Chabot V, et al. Reduced graphene oxide/tin-antimony nanocomposites as anode materials for advanced sodium-ion batteries[J]. ACS Applied Materials & Interfaces,2015,7(44):24895-24901. [12] Zhang J J, Yu A S. Nanostructured transition metal oxides as anode materials for lithium-ion batteries[J]. Science Bulletin,2015,60(9):823-838. doi: 10.1007/s11434-015-0771-6 [13] Liu Z H, Yu Q, Zhao Y L, et al. Silicon oxides: A promising family of anode materials for lithium-ion batteries[J]. Chemical Society Reviews,2019,48(1):285-309. doi: 10.1039/C8CS00441B [14] Qiu D F, Ma X, Zhang J D, et al. Mesoporous silicon microspheres produced from in situ magnesiothermic reduction of silicon oxide for high-performance anode material in sodium-ion batteries[J]. Nanoscale Research Letters,2018,13:275. doi: 10.1186/s11671-018-2699-7 [15] LIN J L, SU S M, HE Y M, et al. Improving the thermal and mechanical properties of an alumina-filled silicone rubber composite by incorporating carbon nanotubes[J]. New Carbon Materials,2020,35(1):66-72. doi: 10.1016/S1872-5805(20)60476-0 [16] Al-Salch M H, Sundararaj U. A review of vapor grown carbon nanofiber/polymer conductive composites[J]. Carbon,2009,47(1):2-22. doi: 10.1016/j.carbon.2008.09.039 [17] Jang S M, Miyawaki J, Tsuji M, et al. The preparation of a novel Si-CNF composite as an effective anodic material for lithium-ion batteries[J]. Carbon,2009,47(15):3383-3391. doi: 10.1016/j.carbon.2009.07.018 [18] Zhu X Y. Synthesis of silicon-carbon composite anode materials for lithium ion batteries by chemical vapor deposition[D]. Qingdao University, 2013. [19] Liu H P, Qiao W M, Zhan L, et al. In situ growth of a carbon nanofiber/Si composite and its application in Li-ion storage[J]. New Carbon Materials,2009,24(2):124-130. doi: 10.1016/S1872-5805(08)60042-6 [20] Yamamura H, Nobuhara K, Nakanishi S, et al. Investigation of the irreversible reaction mechanism and the reactive trigger on SiO anode material for lithium-ion battery[J]. Journal of the Ceramic Society of Japan,2011,119(1395):855-860. doi: 10.2109/jcersj2.119.855 [21] Park C M, Choi W, Hwa Y, et al. Characterization and electrochemical behaviors of disproportionated SiO and its composite for rechargeable Li-ion batteries[J]. Journal of Materials Chemistry,2010,20(23):4854-4860. doi: 10.1039/b923926j [22] Tan T, Lee P K, Yu D Y W, et al. Probing the reversibility of silicon monoxide electrodes for lithium-ion batteries[J]. Journal of the Electrochemical Society,2019,166(3):A5210-A5214. doi: 10.1149/2.0321903jes [23] Nagao Y, Sakaguchi H, Honda H, et al. Structural analysis of pure and electrochemically lithiated SiO using neutron elastic scattering[J]. Journal of the Electrochemical Society,2004,151(10):A1572-A1575. doi: 10.1149/1.1787173 [24] Shi Z Q, Guo C Y, Yi W, et al. Catalytic graphitization of MCMB as anode material for lithium ion batteries[J]. Power Source Technology,2009,33(12):1061-1063. [25] Li C L, Tang F J, Cui X L, et al. Research progress on the composition and modification of SEI films in lithium ion batteries[J]. Power Source Technology,2016,40(10):2079-2081. [26] Yamasaki S, Nishino T, Asada A. Nonaqueous secondary battery with lithium titanium cathode [P]. U.S. Patent 6759168, 2004. [27] Jo Y N, Kim Y, Kim J S, et al. Si-graphite composites as anode materials for lithium secondary batteries[J]. Journal of Power Sources,2010,195(18):6031-6036. doi: 10.1016/j.jpowsour.2010.03.008 [28] Si Q, Hanai K, Imanishi N, et al. Highly reversible carbon-nano-silicon composite anodes for lithium rechargeable batteries[J]. Journal of Power Sources,2009,189(1):761-765. doi: 10.1016/j.jpowsour.2008.08.007 [29] Liu Y, Wen Z Y, Wang X Y, et al. Electrochemical behaviors of Si/C composite synthesized from F-containing precursors[J]. Journal of Power Sources,2009,189(1):733-737. doi: 10.1016/j.jpowsour.2008.08.016 [30] Chou S L, Wang J Z, Choucair M, et al. Enhanced reversible lithium storage in a nanosize silicon/graphene composite[J]. Electrochemistry Communications,2010,12(2):303-306. doi: 10.1016/j.elecom.2009.12.024 [31] Abel P R, Chockla A M, Lin Y M, et al. Nanostructured Si1-xGex for tunable thin film lithium-ion battery anodes[J]. ACS Nano,2013,7(3):2249-2257. doi: 10.1021/nn3053632 -





 下载:
下载: