-
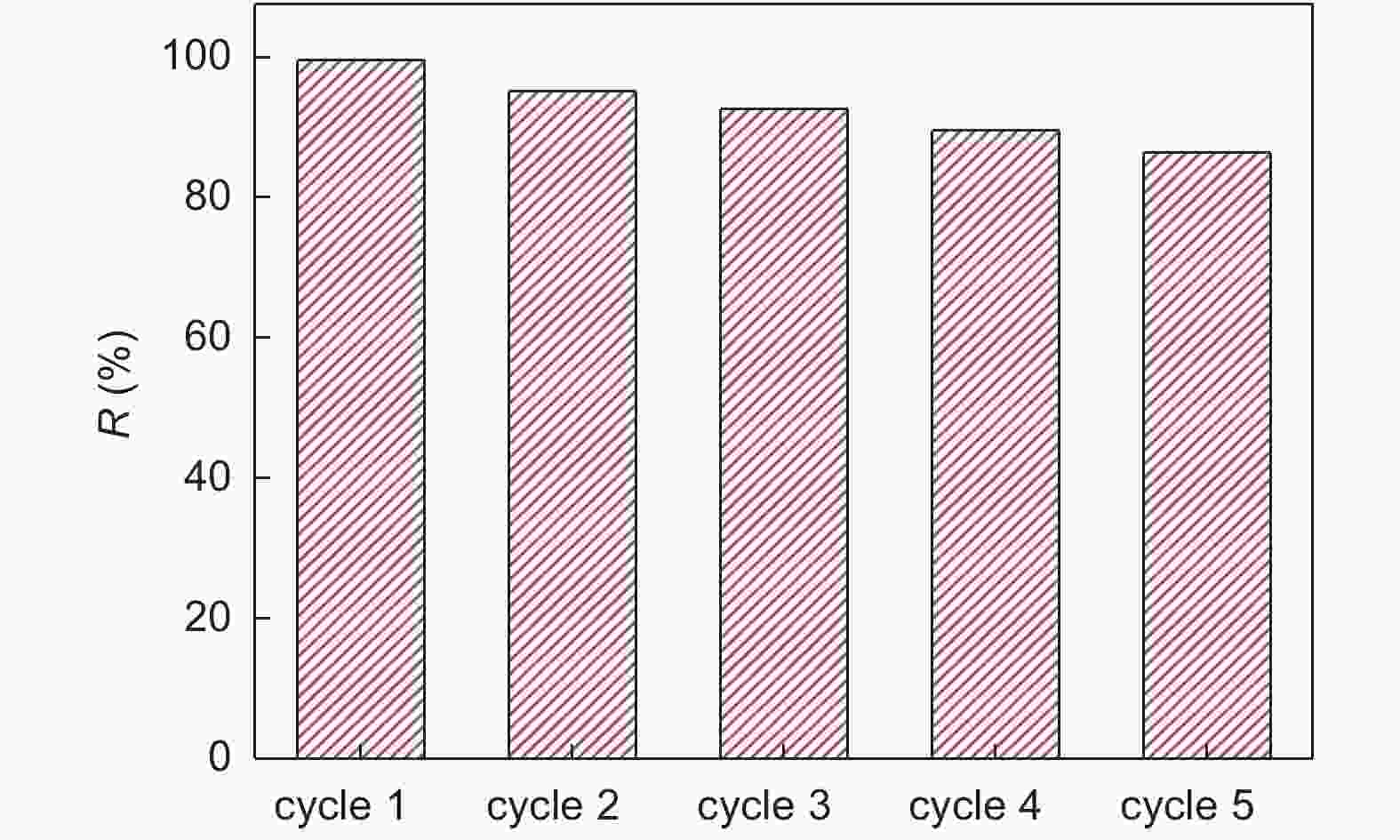
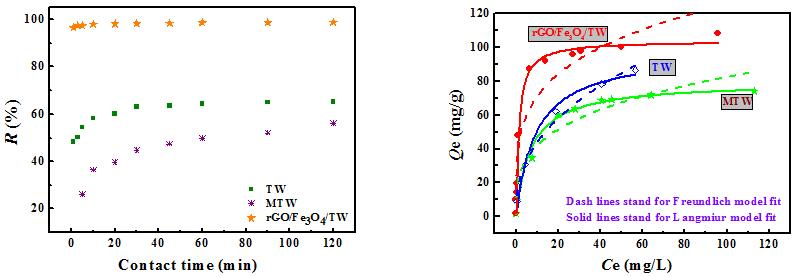
摘要: 将改进Hummers法合成的氧化石墨烯(GO)与废茶渣(TW)、Fe3O4进行复合获得废茶渣磁性改性物rGO/Fe3O4/TW,通过FTIR光谱和XRD对产物结构和晶型进行表征。考察溶液pH 值、振荡时间和铀初始浓度对合成产物吸附行为的影响。采用孔结构分析仪和XPS对rGO/Fe3O4/TW吸附前后样品的孔特性和表面结构进行吸附机理分析。结果表明,rGO/Fe3O4/TW不仅具有优良的去铀性能,短时间内可达近100%的去除率,而且负载铀之后通过磁场作用易从液相中快速分离出来。rGO/Fe3O4/TW对铀的吸附过程符合Langmuir 模型和准二级动力学模型。对于初始浓度为10 mg/L的含铀溶液,TW、MTW和rGO/Fe3O4/TW的最大吸附量分别为97.70 mg g−1、79.46 mg g−1和103.84 mg g−1。同时,rGO/Fe3O4/TW具有良好的循环再利用性,经5个吸附-解吸-再吸附循环之后,仍可达到较好的去铀效果,去铀率约为85%。Abstract: Used tea leaves, or tea waste (TW), were crushed into powder and mixed with graphene oxide (GO) in water, followed by adjusting the pH value of the resulting suspension with ammonia to 11, adding FeCl2·4H2O under magnetic stirring, filtration and drying to prepare a rGO/Fe3O4/TW (with mass ratios of 1∶2∶1) hybrid material. The structure and crystalline phases of the material were characterized by FTIR and XRD. Isotherms for uranium adsorption were obtained and its kinetics were measured in a conical bottle that was placed in a shaker. The effects of the pH value of the uranium solution, adsorption time and initial concentration on the uranium adsorption were investigated. Results indicate that the hybrid has a much faster adsorption rate than TW with an uranium removal rate up to nearly 100% in 20 min for an initial uranium concentration of 10 mg L−1. The maximum adsorption capacity of the hybrid is 103.84 mg g−1 while that of TW is 97.70 mg g−1. The hybrid with adsorbed uranium can be easily separated from the solution by applying a magnetic field. The isotherms and kinetics of uranium adsorption on the hybrid are best fitted by the Langmuir isotherm model and the pseudo-second-order model, respectively. The hybrid has good reusability with an uranium removal rate of about 85% after 5 cycles.
-
Key words:
- Tea waste /
- Graphene oxide /
- Magnetic modification /
- Adsorption /
- Uranium
-
Table 1. Adsorption kinetic model parameters for TW, MTW and rGO/Fe3O4/TW.
Sorbents Pseudo-first-order model Pseudo-second-order model Qe
(mg/g)k1 (g/(mg·
min)R2 Qe (mg/g) k2 (g/(mg·
min)R2 TW 0.5756 0.026 0.9361 19.92 0.233 0.9999 MTW 0.1298 0.018 0.9677 12.35 0.006 0.9945 rGO/Fe3O4/TW 0.2075 0.035 0.9730 20.41 0.027 0.9993 Table 2. Adsorption parameters for langmuir and freundlich isotherm models.
Sorbents Langmuir isotherm Freundlich isotherm Qm
(mg g−1)kL
(L mg−1)R2 n kF (mg1−n Ln g−1) R2 TW 97.70 0.11 0.9899 2.42 16.86 0.9885 MTW 79.46 0.14 0.9969 3.42 21.52 0.8937 rGO/Fe3O4/TW 103.84 0.75 0.9983 4.05 40.27 0.8749 -
[1] Safarik I, Baldikova E, Prochazkova J, et al. Magnetically modified agricultural and food waste: Preparation and application[J]. J Agric Food Chem,2018,66:2538-2552. doi: 10.1021/acs.jafc.7b06105 [2] Sarici-Özdemir Ç, Kiliç F. Kinetics behavior of methylene blue onto agricultural waste[J]. Particul Sci Technol,2018,36(2):194-201. doi: 10.1080/02726351.2016.1240127 [3] Afroze S, Sen T K. A review on heavy metal ions and dye adsorption from water by agricultural solid waste adsorbents[J]. Water Air Soil Pollut,2018,229:225-274. doi: 10.1007/s11270-018-3869-z [4] Wu X X, Zhang C Y, Tian Z W, et al. Large-surface-area carbons derived from lotus stem waste for efficient CO2 capture[J]. New Carbon Mater,2018,33(3):252-261. doi: 10.1016/S1872-5805(18)60338-5 [5] Dieng H, Zawawi R B M, Yusof N I S B M, et al. Green tea and its waste attract workers of formicine ants and kill their workers—implications for pest management[J]. Ind Crop Prod,2016,89:157-166. doi: 10.1016/j.indcrop.2016.05.019 [6] Foroughi-dahr M, Abolghasemi H, Esmaieli M, et al. Experimental study on the adsorptive behavior of Congo red in cationic surfactant-modified tea waste[J]. Process Saf Environ Prot,2015,95:226-236. doi: 10.1016/j.psep.2015.03.005 [7] Yang S X, Wu Y H, Aierken A, et al. Mono/competitive adsorption of Arsenic(Ⅲ) and Nickel(Ⅱ) using modified green tea waste[J]. J Taiwan Inst Chem E,2016,60:213-221. doi: 10.1016/j.jtice.2015.07.007 [8] Gupta A, Balomajumder C. Simultaneous adsorption of Cr(VI) and phenol onto tea waste biomass from binary mixture: multicomponent adsorption, thermodynamic and kinetic study[J]. J Environ Chem Eng,2015,3:785-796. doi: 10.1016/j.jece.2015.03.003 [9] Weng C H, Lin Y T, Hong D Y, et al. Effective removal of copper ions from aqueous solution using base treated black tea waste[J]. Ecol Eng,2014,67:127-133. doi: 10.1016/j.ecoleng.2014.03.053 [10] Tzeng J H, Weng C H, Huang J W, et al. Spent tea leaves: A new non-conventional and low-cost biosorbent for ethylene removal[J]. Int Biodeter Biodegr,2015,104:67-73. doi: 10.1016/j.ibiod.2015.05.012 [11] Ding D X, Liu X T, Hu N, et al. Removal and recovery of uranium from aqueous solution by tea waste[J]. J Radioanal Nucl Chem,2012,293:735-741. doi: 10.1007/s10967-012-1866-z [12] Cai H M, Chen G J, Peng C Y, et al. Removal of fluoride from drinking water using tea waste loaded with Al/Fe oxides: A novel, safe and efficient biosorbent[J]. Appl Surf Sci,2015,328:34-44. doi: 10.1016/j.apsusc.2014.11.164 [13] Boruah P K, Borthakur P, Darabdhara G, et al. Sunlight assisted degradation of dye molecules and reduction of toxic Cr(VI) in aqueous medium using magnetically recoverable Fe3O4/reduced graphene oxide nanocomposite[J]. RSC Adv,2016,6:11049-11063. doi: 10.1039/C5RA25035H [14] Vu H C, Dwivedi A D, Le T T, et al. Magnetite graphene oxide encapsulated in alginate beads for enhanced adsorption of Cr(VI) and As(V) from aqueous solutions: role of crosslinking metal cations in pH control[J]. Chem Eng J,2017,307:220-229. doi: 10.1016/j.cej.2016.08.058 [15] Madrakian T, Afkhami A, Ahmadi M. Adsorption and kinetic studies of seven different organic dyes onto magnetite nanoparticles loaded tea waste and removal of them from wastewater samples[J]. Spectrochim Acta A,2012,99:102-109. doi: 10.1016/j.saa.2012.09.025 [16] Ma J, Liu C H, Li R, et al. Properties and structural characterization of oxide starch/chitosan/graphene oxide biodegradable nanocomposites[J]. J Appl Polym Sci,2012,123:2933-2944. doi: 10.1002/app.34901 [17] Li X Y, Li F B, Jin Y, et al. The uptake of uranium by tea wastes investigated by batch, spectroscopic and modeling techniques[J]. J Mol Liq,2015,209:413-418. doi: 10.1016/j.molliq.2015.06.014 [18] Atarod M, Nasrollahzadeh M, Sajadi S M. Green synthesis of Pd/RGO/Fe3O4 nanocomposite using withania coagulans leaf extract and its application as magnetically separable and reusable catalyst for the reduction of 4-nitrophenol[J]. J Colloid Interf Sci,2016,465:249-258. doi: 10.1016/j.jcis.2015.11.060 [19] Madhuvilakku R, Alagar S, Mariappan R, et al. Green one-pot synthesis of flowers-like Fe3O4/rGO hybrid nanocomposites for effective electrochemical detection of riboflavin and low-cost supercapacitor applications[J]. Sensor Actuat B-Chem,2017,253:879-892. doi: 10.1016/j.snb.2017.06.126 [20] Helal A S, Mazario E, Mayoral A, et al. Highly efficient and selective extraction of uranium from aqueous solution using a magnetic device: Succinyl-β-cyclodextrin-APTES@maghemite nanoparticles[J]. Environ Sci: Nano,2018,5:158-168. doi: 10.1039/C7EN00902J [21] Campos A F C, Oliveira H A L, F N Silva, et al. Core-shell bimagnetic nanoadsorbents for hexavalent chromium removal from aqueous solutions[J]. J Hazard Mater,2019,362:82-91. doi: 10.1016/j.jhazmat.2018.09.008 -





 下载:
下载: