-
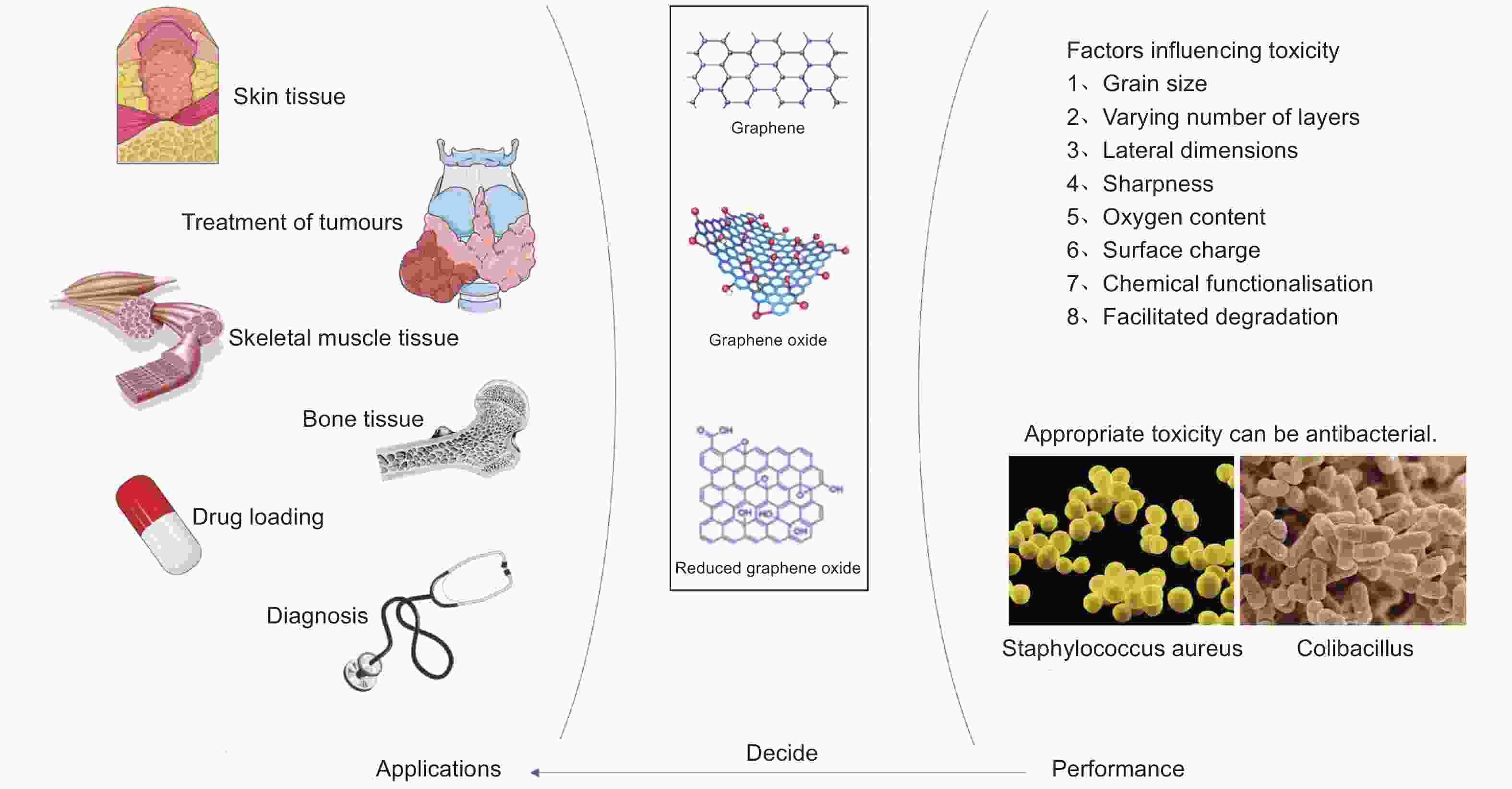
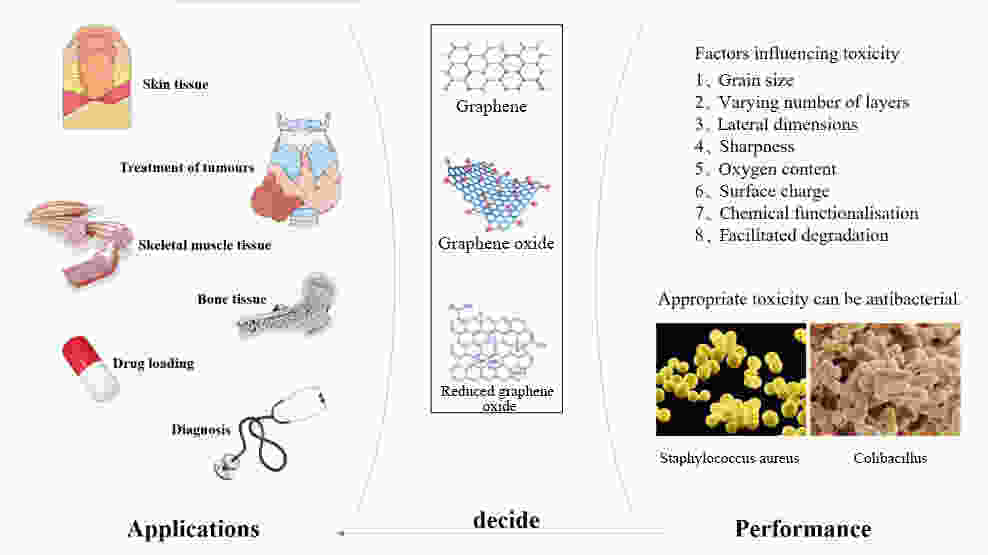
摘要: 石墨烯是紧密堆积在二维蜂窝状晶格中一层扁平的碳原子单层,具有良好的光学性、导电性、力学性、低毒性、抗菌性、生物相容性和稳定性。氧化石墨烯、还原氧化石墨烯是石墨烯的衍生物,具备石墨烯类似的性质,石墨烯及其衍生物的研究进展十分迅速,在生物医药方向拥有较大的应用潜力。本文从石墨烯及其衍生物的毒性、抗菌性的角度出发,综述了石墨烯及其衍生物在治疗皮肤创伤、治疗肿瘤、促进骨骼肌和骨再生、载药、诊断疾病等方面的应用,并提出了目前所存在的问题和解决对策,展望了未来石墨烯基材料的研究发展前景。Abstract: Graphene (Gr) is a monolayer of carbon atoms in a two-dimensional honeycomb lattice, and has derivatives of graphene oxide and reduced graphene oxide. Gr is widely used in various fields for its good optics, conductivity, mechanical properties, low toxicity, antibacterial properties, biocompatibility and stability. Graphene oxide and reduced graphene oxide have similar properties to Gr, and all three materials have advantages and disadvantages and are often not used alone but are composited with other materials to improve their properties for a particular application. From the perspectives of the toxicity and antibacterial properties of Gr and its derivatives, this paper reviews their uses in treating skin wounds and tumours, promoting the regeneration of skeletal muscle and bone, and facilitating drug loading and diagnosis. The problems associated with these applications are analyzed and solutions are suggested. Future research and development prospects for Gr-based materials are presented.
-
Key words:
- Graphene /
- Antibacterial /
- Biomedicine /
- Biological dressing /
- Tumour treatment
-
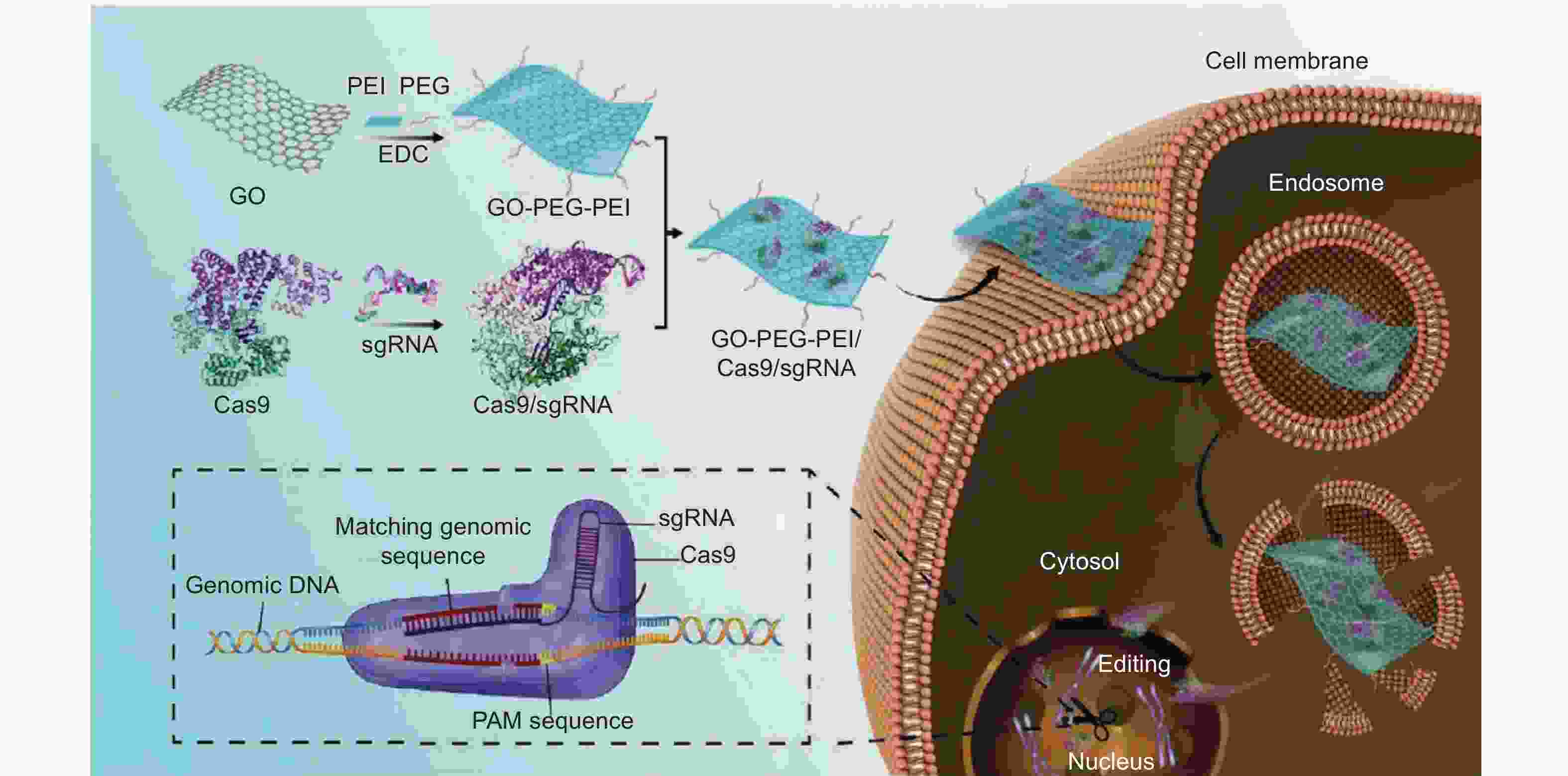
Figure 1. Schematic diagram of the GO−PEG−PEI-based Cas9/sgRNA delivery system ( Reprinted with permission)[11].
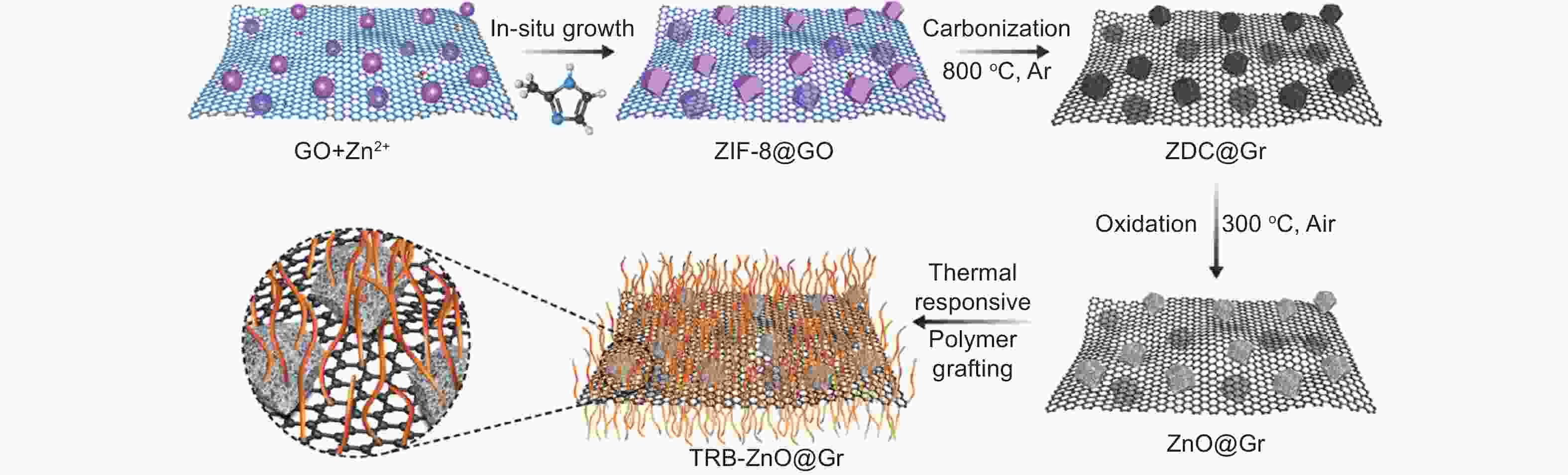
Figure 2. Schematic illustrations for the preparation of 2D TRB-ZnO@Gr nanosheets (Reprinted with permission)[20].
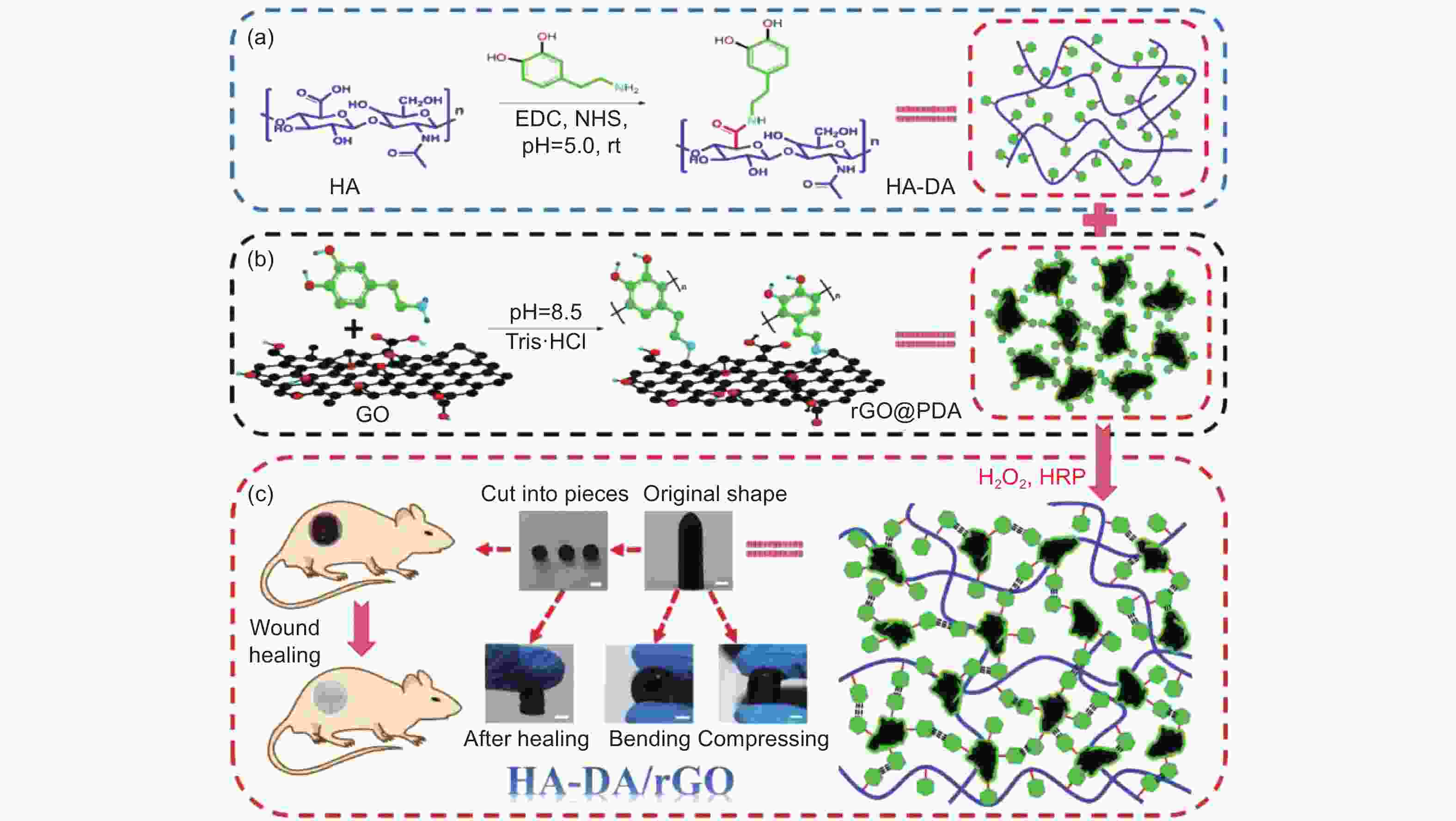
Figure 3. Diagrammatic sketch of HA-DA/rGO hydrogel preparation: (a) preparation scheme of HA-DA polymer, (b) rGO@PDA and (c) scheme of HA-DA/rGO hydrogel and the original, bending, compressing, self-healing representation and the application in wound healing. Scale bar: 5 mm. (DA and PDA are abbreviated forms of dopamine and polydopamine, respectively) (Reprinted with permission)[50].
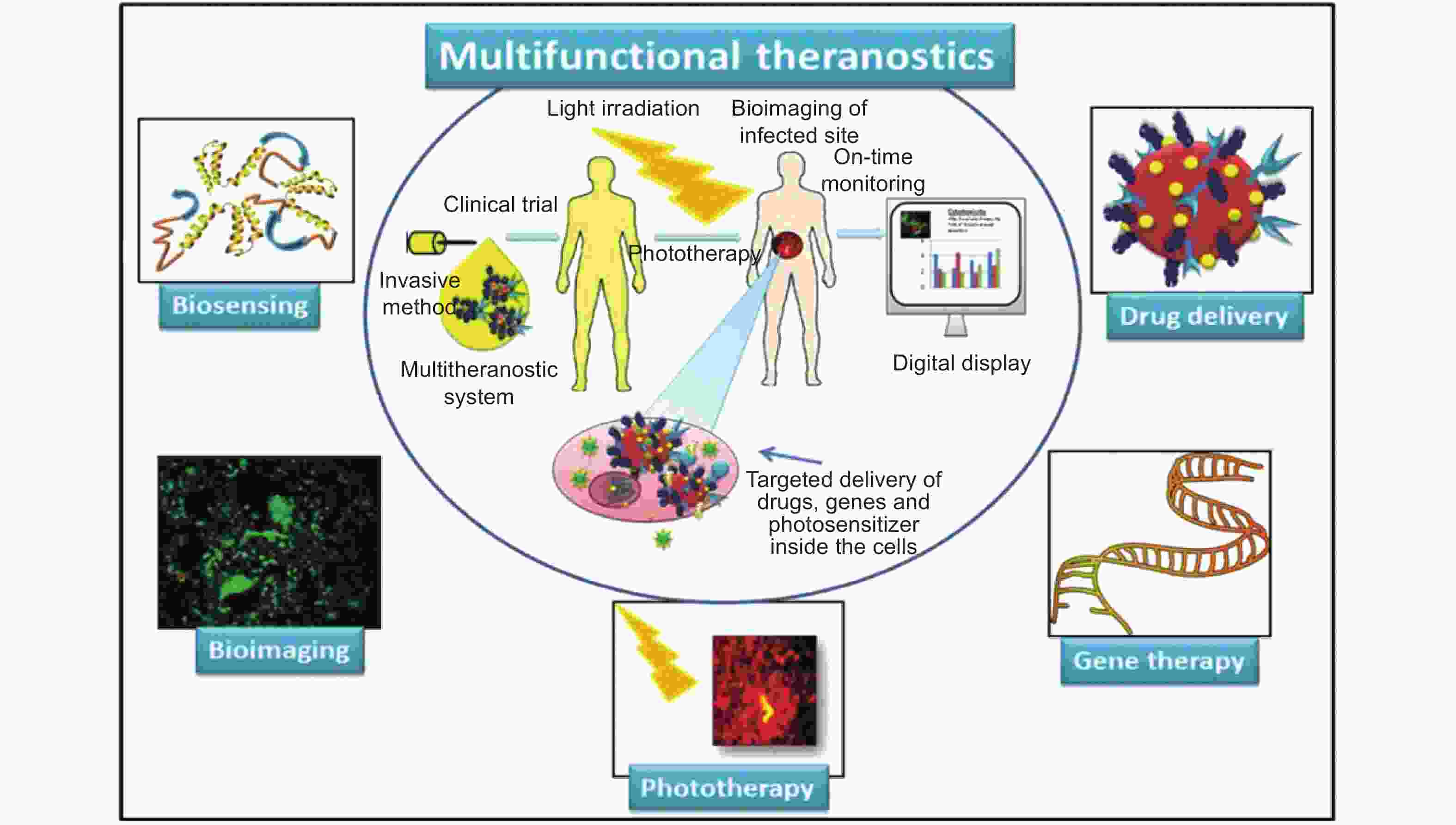
Figure 4. Schematic diagram describing different roles of graphene in cancer theranostics (Reprinted with permission)[93].
-
[1] Volkov Y, Mcintyre J, Prina-Mello A. Graphene toxicity as a double-edged sword of risks and exploitable opportunities: a critical analysis of the most recent trends and developments[J]. 2D Materials,2017,4(2):022001. doi: 10.1088/2053-1583/aa5476 [2] Wang Beidi. Study on the functionalization and preliminary application of graphene oxide[D]. Master Degree, Fudan University, 2012. [3] Chen Jianli. Chemical functionalization of graphene oxide and graphene-based derivatives/nanocomposites: preparations and properties[D]. Doctoral Degree, Jilin University, 2013. [4] Liao K H, Lin Y S, Macosko C W, et al. Cytotoxicity of graphene oxide and graphene in human erythrocytes and skin fibroblasts[J]. ACS Applied Materials & Interfaces,2011,3(7):2607-2615. [5] Maity S, Pakhira B, Ghosh S, et al. Microcarbon-based facial creams activate aerial oxygen under light to reactive oxygen species damaging cell[J]. Applied Nanoscience,2017,7(8):607-616. doi: 10.1007/s13204-017-0604-9 [6] Bengtson S, Kling K, Madsen A M, et al. No cytotoxicity or genotoxicity of graphene and graphene oxide in murine lung epithelial FE1 cells in vitro[J]. Environmental and Molecular Mutagenesis,2016,57(6):469-482. doi: 10.1002/em.22017 [7] De Luna La V, De Moraes A C M, Consonni S R, et al. Comparative in vitro toxicity of a graphene oxide-silver nanocomposite and the pristine counterparts toward macrophages[J]. Journal of Nanobiotechnology,2016,14:12. doi: 10.1186/s12951-016-0165-1 [8] Ionita M, Crica L E, Voicu S I, et al. Synergistic effect of carbon nanotubes and graphene for high performance cellulose acetate membranes in biomedical applications[J]. Carbohydrate Polymers,2018,183:50-61. doi: 10.1016/j.carbpol.2017.10.095 [9] Pena-Bahamonde J, San Miguel V, Nguyen H N, et al. Functionalization of reduced graphene oxide with polysulfone brushes enhance antibacterial properties and reduce human cytotoxicity[J]. Carbon,2017,111:258-268. doi: 10.1016/j.carbon.2016.10.005 [10] Yan D, Zhao H Y, Pei J Y, et al. The rational designed graphene oxide-Fe2O3 composites with low cytotoxicity[J]. Materials Science & Engineering C-Materials for Biological Applications,2017,72:659-666. [11] Ganguly P, Breen A, Pillai S C. Toxicity of nanomaterials: exposure, pathways, assessment, and recent advances[J]. Acs Biomaterials Science & Engineering,2018,4(7):2237-2275. [12] Siew Q Y, Tham S Y, Loh H S, et al. One-step green hydrothermal synthesis of biocompatible graphene/TiO2 nanocomposites for non-enzymatic H2O2 detection and their cytotoxicity effects on human keratinocyte and lung fibroblast cells[J]. Journal of Materials Chemistry B,2018,6(8):1195-1206. doi: 10.1039/C7TB02891A [13] Khatamian M, Divband B, Farahmand-Zahed F. Synthesis and characterization of zinc (II)-loaded zeolite/graphene oxide nanocomposite as a new drug carrier[J]. Materials Science & Engineering C-Materials for Biological Applications,2016,66:251-258. [14] Carpio I E M, Santos C M, Wei X,et al. Toxicity of a polymer-graphene oxide composite against bacterial planktonic cells, biofilms, and mammalian cells[J]. Nanoscale,2012,4(15):4746-4756. doi: 10.1039/c2nr30774j [15] Cho Y C, Pak P J, Joo Y H, et al. In vitro and in vivo comparison of the immunotoxicity of single- and multi-layered graphene oxides with or without pluronic F-127[J]. Scientific Reports,2016,6(1):38884. doi: 10.1038/srep38884 [16] Pulingam T, Thong K L, Appaturi J N, et al. Synergistic antibacterial actions of graphene oxide and antibiotics towards bacteria and the toxicological effects of graphene oxide on human epidermal keratinocytes[J]. European Journal of Pharmaceutical Sciences,2020,142:105087. doi: 10.1016/j.ejps.2019.105087 [17] Wu S, Liu Y, Zhang H, et al. Nano-graphene oxide improved the antibacterial property of antisense yycG RNA on staphylococcus aureus[J]. Journal of Orthopaedic Surgery and Research,2019,14(1):305. doi: 10.1186/s13018-019-1356-x [18] Khalil W A, Sherif H H A, Hemdan B A, et al. Biocompatibility enhancement of graphene oxide-silver nanocomposite by functionalisation with polyvinylpyrrolidone[J]. IET Nanobiotechnol,2019,13(8):816-823. doi: 10.1049/iet-nbt.2018.5321 [19] Stan M S, Nica I C, Popa M, et al. Reduced graphene oxide/TiO2 nanocomposites coating of cotton fabrics with antibacterial and self-cleaning properties[J]. Journal of Industrial Textiles,2018,49(3):277-293. [20] Fan X, Yang F, Huang J, et al. Metal-organic-framework-derived 2D carbon nanosheets for localized multiple bacterial eradication and augmented anti-infective therapy[J]. Nano Letters,2019,19(9):5885-5896. doi: 10.1021/acs.nanolett.9b01400 [21] Liang Y, Chen B, Li M, et al. Injectable antimicrobial conductive hydrogels for wound disinfection and infectious wound healing[J]. Biomacromolecules,2020,21(5):1841-1852. doi: 10.1021/acs.biomac.9b01732 [22] Huang S, Liu H, Liao K, et al. Functionalized GO nanovehicles with nitric oxide release and photothermal activity-based hydrogels for bacteria-infected wound healing[J]. ACS Applied Materials & Interfaces,2020,12(26):28952-28964. [23] Jian Z, Wang H, Liu M, et al. Polyurethane-modified graphene oxide composite bilayer wound dressing with long-lasting antibacterial effect[J]. Materials Science & Engineering C-Materials for Biological Applications,2020,111:110833. [24] Jang J, Lee J M, Oh S B, et al. Development of antibiofilm nanocomposites: Ag/Cu bimetallic nanoparticles synthesized on the surface of graphene oxide nanosheets[J]. ACS Applied Materials & Interfaces,2020,12(32):35826-35834. [25] Zhang Y, Ruan H, Guo C, et al. Thin-film nanocomposite reverse osmosis membranes with enhanced antibacterial resistance by incorporating p-aminophenol-modified graphene oxide[J]. Separation and Purification Technology,2020,234:116017-116017. doi: 10.1016/j.seppur.2019.116017 [26] Oves M, Rauf M A, Ansari M O, et al. Graphene decorated zinc oxide and curcumin to disinfect the methicillin-resistant staphylococcus aureus[J]. Nanomaterials,2020,10(5):1004. doi: 10.3390/nano10051004 [27] Xu W P, Zhang L C, Li J P, et al. Facile synthesis of silver@graphene oxide nanocomposites and their enhanced antibacterial properties[J]. Journal of Materials Chemistry,2011,21(12):4593-4597. doi: 10.1039/c0jm03376f [28] Shoeb M, Mobin M, Rauf M A, et al. In vitro and in vivo antimicrobial evaluation of graphene-polyindole (Gr@PIn) nanocomposite against methicillin-resistant staphylococcus aureus pathogen[J]. ACS Omega,2018,3(8):9431-9440. doi: 10.1021/acsomega.8b00326 [29] Cui J, Liu Y. Preparation of graphene oxide with silver nanowires to enhance antibacterial properties and cell compatibility[J]. RSC Advances,2015,5(104):85748-85755. doi: 10.1039/C5RA16371D [30] Li C, Ye R, Bouckaert J, et al. Flexible nanoholey patches for antibiotic-free treatments of skin infections[J]. ACS Applied Materials & Interfaces,2017,9(42):36665-36674. [31] Yan X, Fang W W, Xue J, et al. Thermoresponsive in situ forming hydrogel with sol-gel irreversibility for effective methicillin-resistant staphylococcus aureus infected wound healing[J]. ACS Nano,2019,13(9):10074-10084. doi: 10.1021/acsnano.9b02845 [32] Tong C, Li L, Xiao F, et al. Daptomycin and AgNP co-loaded rGO nanocomposites for specific treatment of Gram-positive bacterial infection in vitro and in vivo[J]. Biomaterials Science,2019,7(12):5097-5111. doi: 10.1039/C9BM01229J [33] Sadeghianmaryan A, Karimi Y, Naghieh S, et al. Electrospinning of scaffolds from the polycaprolactone/polyurethane composite with graphene oxide for skin tissue engineering[J]. Applied Biochemistry and Biotechnology,2020,191(2):567-578. doi: 10.1007/s12010-019-03192-x [34] Pourjavadi A, Mazaheri Tehrani Z, Salami H, et al. Both tough and soft double network hydrogel nanocomposite based on O‐carboxymethyl chitosan/poly(vinyl alcohol) and graphene oxide: a promising alternative for tissue engineering[J]. Polymer Engineering & Science,2020,60(5):889-899. [35] Mahmoudi N, Eslahi N, Mehdipour A, et al. Temporary skin grafts based on hybrid graphene oxide-natural biopolymer nanofibers as effective wound healing substitutes: pre-clinical and pathological studies in animal models[J]. Journal of Materials Science: Materials in Medicine,2017,28(5):73. doi: 10.1007/s10856-017-5874-y [36] Li Longjian. Study on the preparation of aloin—loaded graphene oxide gel and its effect on wound healing[D]. Master Degree, Zhejiang University, 2017. [37] Li J, Zhou C, Luo C,et al. N-acetyl cysteine-loaded graphene oxide-collagen hybrid membrane for scarless wound healing[J]. Theranostics,2019,9(20):5839-5853. doi: 10.7150/thno.34480 [38] Tang P, Han L, Li P, et al. Mussel-inspired electroactive and antioxidative scaffolds with incorporation of polydopamine-reduced graphene oxide for enhancing skin wound healing[J]. ACS Applied Materials & Interfaces,2019,11(8):7703-7714. [39] Safina I, Bourdo S E, Algazali K M, et al. Graphene-based 2D constructs for enhanced fibroblast support[J]. PLoS One,2020,15(5):e0232670. doi: 10.1371/journal.pone.0232670 [40] Jafarkhani M, Salehi Z, Bagheri Z, et al. Graphene functionalized decellularized scaffold promotes skin cell proliferation[J]. The Canadian Journal of Chemical Engineering,2019,98(1):62-68. [41] Kumar A, Zo S M, Kim J H, et al. Enhanced physical, mechanical, and cytocompatibility behavior of polyelectrolyte complex hydrogels by reinforcing halloysite nanotubes and graphene oxide[J]. Composites Science and Technology,2019,175:35-45. doi: 10.1016/j.compscitech.2019.03.008 [42] Lasocka I, Jastrzebska E, Szulc-Dabrowska L, et al. The effects of graphene and mesenchymal stem cells in cutaneous wound healing and their putative action mechanism[J]. International Journal of Nanomedicine,2019,14:2281-2299. doi: 10.2147/IJN.S190928 [43] Li Z, Wang H, Yang B, et al. Three-dimensional graphene foams loaded with bone marrow derived mesenchymal stem cells promote skin wound healing with reduced scarring[J]. Materials Science & Engineering C-Materials for Biological Applications,2015,57:181-188. [44] Haghshenas M, Hoveizi E, Mohammadi T,et al. Use of embryonic fibroblasts associated with graphene quantum dots for burn wound healing in Wistar rats[J]. In Vitro Cellular & Developmental Biology - Animal,2019,55(4):312-322. [45] Zhang B, He J, Shi M, et al. Injectable self-healing supramolecular hydrogels with conductivity and photo-thermal antibacterial activity to enhance complete skin regeneration[J]. Chemical Engineering Journal,2020,400:125994. doi: 10.1016/j.cej.2020.125994 [46] Yu Xunzhou. Preparing a photothermal-induced targeted graphene nano-antibacterial materials for treatment of skin infection[D]. Master Degree, Army Medical University, 2019. [47] Qian Wei. Preparation of novel graphene-based composite materials and experimental study on using them to promote skin wound repair[D]. Doctoral Degree, Army Medical University, 2019. [48] Azarniya A, Eslahi N, Mahmoudi N, et al. Effect of graphene oxide nanosheets on the physico-mechanical properties of chitosan/bacterial cellulose nanofibrous composites[J]. Composites Part A: Applied Science and Manufacturing,2016,85:113-122. doi: 10.1016/j.compositesa.2016.03.011 [49] Qian W, Hu X, He W, et al. Polydimethylsiloxane incorporated with reduced graphene oxide (rGO) sheets for wound dressing application: preparation and characterization[J]. Colloids and Surfaces B: Biointerfaces,2018,166:61-71. doi: 10.1016/j.colsurfb.2018.03.008 [50] Liang Y, Zhao X, Hu T, et al. Adhesive hemostatic conducting injectable composite hydrogels with sustained drug release and photothermal antibacterial activity to promote full-thickness skin regeneration during wound healing[J]. Small,2019,15(12):1900046. doi: 10.1002/smll.201900046 [51] Zhang Q, Du Q, Zhao Y, et al. Graphene oxide-modified electrospun polyvinyl alcohol nanofibrous scaffolds with potential as skin wound dressings[J]. RSC Advances,2017,7(46):28826-28836. doi: 10.1039/C7RA03997B [52] Di Luca M, Vittorio O, Cirillo G, et al. Electro-responsive graphene oxide hydrogels for skin bandages: the outcome of gelatin and trypsin immobilization[J]. International Journal of Pharmaceutics,2018,546(1-2):50-60. doi: 10.1016/j.ijpharm.2018.05.027 [53] Li Zhonghua. The research on three-dimensional graphene foams loaded with bone marrow derived mesenchymal stem cells promoting skin wound healing[D]. Doctoral Degree, Shandong University, 2017. [54] Fusco L, Pelin M, Sosa S, et al. In vitro pro-inflammatory effects of graphene and graphene oxide on human skin keratinocytes and monocytes[J]. Basic & Clinical Pharmacology & Toxicology,2018,123:80-80. [55] Fusco L, Pelin M, Mukherjee S, et al. Keratinocytes are capable of selectively sensing low amounts of graphene-based materials:implications for cutaneous applications[J]. Carbon,2020,159:598-610. doi: 10.1016/j.carbon.2019.12.064 [56] Li Q, Li F, Qi X, et al. Pluronic® F127 stabilized reduced graphene oxide hydrogel for the treatment of psoriasis: in vitro and in vivo studies[J]. Colloids and Surfaces B: Biointerfaces,2020,195:111246. doi: 10.1016/j.colsurfb.2020.111246 [57] Chou H Y, Wang H D, Kuo C H, et al. Antioxidant graphene oxide nanoribbon as a novel whitening agent inhibits microphthalmia-associated transcription factor-related melanogenesis mechanism[J]. ACS Omega,2020,5(12):6588-6597. doi: 10.1021/acsomega.9b04316 [58] Sarkar S, Gurjarpadhye A A, Rylander C G, et al. Optical properties of breast tumor phantoms containing carbon nanotubes and nanohorns[J]. Journal of Biomedical Optics,2011,16(5):051304. doi: 10.1117/1.3574762 [59] Turcheniuk K, Dumych T, Bilyy R, et al. Plasmonic photothermal cancer therapy with gold nanorods/reduced graphene oxide core/shell nanocomposites[J]. RSC Advances,2016,6:1600-1610. doi: 10.1039/C5RA24662H [60] Matai I, Kaur G, Soni S, et al. Near-infrared stimulated hydrogel patch for photothermal therapeutics and thermoresponsive drug delivery[J]. Journal of Photochemistry & Photobiology, B: Biology,2020,210:111960. [61] Karbalaei A, Mohammadalipour Z, Rahmati M, et al. GO/TiO2 hybrid nanoparticles as a new photosensitizer in photodynamic therapy of A375 melanoma cancer cells[J]. Journal of Skin and Stem Cell,2016,4(2):e63984. [62] Ahirwar S, Mallick S, Bahadur D. Photodynamic therapy using graphene quantum dot derivatives[J]. Journal of Solid State Chemistry,2020,282:121107. doi: 10.1016/j.jssc.2019.121107 [63] Han R, Tang K, Hou Y, et al. Ultralow-intensity near infrared light synchronously activated collaborative chemo/photothermal/photodynamic therapy[J]. Biomaterials Science,2020,8(2):607-618. doi: 10.1039/C9BM01607D [64] Liu J, Saul D, Boker K O, et al. Current methods for skeletal muscle tissue repair and regeneration[J]. Biomed Research International,2018,2018:1-11. [65] Tedesco F S, Cossu G. Stem cell therapies for muscle disorders[J]. Current Opinion in Neurology,2012,25(5):597-603. doi: 10.1097/WCO.0b013e328357f288 [66] Charge S B P, Rudnicki M A. Cellular and molecular regulation of muscle regeneration[J]. Physiological reviews,2004,84(1):209-238. doi: 10.1152/physrev.00019.2003 [67] Jenkins T L, Little D. Synthetic scaffolds for musculoskeletal tissue engineering: cellular responses to fiber parameters[J]. Npj Regenerative Medicine,2019,4:15. doi: 10.1038/s41536-019-0076-5 [68] Palmieri V, Sciandra F, Bozzi M, et al. 3D graphene scaffolds for skeletal muscle regeneration: future perspectives[J]. Frontiers in Bioengineering and Biotechnology,2020,8:383. doi: 10.3389/fbioe.2020.00383 [69] Dai Z H, Wang Y L, Liu L Q, et al. Hierarchical graphene-based films with dynamic self-siffening for biomimetic artificial muscle[J]. Advanced Functional Materials,2016,26(38):7003-7010. doi: 10.1002/adfm.201503917 [70] Krueger E, Chang A N, Brown D, et al. Graphene foam as a three-dimensional platform for myotube growth[J]. Acs Biomaterials Science & Engineering,2016,2(8):1234-1241. [71] Papi M, Palmieri V, Bugli F, et al. Biomimetic antimicrobial cloak by graphene-oxide agar hydrogel.[J]. Scientific Reports,2017,7(1):12. doi: 10.1038/s41598-017-00047-5 [72] Park J, Choi J H, Kim S, et al. Micropatterned conductive hydrogels as multifunctional muscle-mimicking biomaterials: graphene-incorporated hydrogels directly patterned with femtosecond laser ablation[J]. Acta Biomaterialia,2019,97:141-153. doi: 10.1016/j.actbio.2019.07.044 [73] Grasman J M, Zayas M J, Page R L, et al. Biomimetic scaffolds for regeneration of volumetric muscle loss in skeletal muscle injuries[J]. Acta Biomaterialia,2015,25:2-15. doi: 10.1016/j.actbio.2015.07.038 [74] Ciriza J, Del Burgo L S, Virumbrales-Munoz M, et al. Graphene oxide increases the viability of C2C12 myoblasts microencapsulated in alginate[J]. International Journal of Pharmaceutics,2015,493(1-2):260-270. doi: 10.1016/j.ijpharm.2015.07.062 [75] Ciriza J, Saenz Del Burgo L, Gurruchaga H,et al. Graphene oxide enhances alginate encapsulated cells viability and functionalty while not affecting the foreign body response[J]. Drug Delivery,2018,25(1):1147-1160. doi: 10.1080/10717544.2018.1474966 [76] Choe G, Kim S W, Park J, et al. Anti-oxidant activity reinforced reduced graphene oxide/alginate microgels: mesenchymal stem cell encapsulation and regeneration of infarcted hearts[J]. Biomaterials,2019,225:119513. doi: 10.1016/j.biomaterials.2019.119513 [77] Shin Y C, Kang S H, Lee J H, et al. Three-dimensional graphene oxide-coated polyurethane foams beneficial to myogenesis[J]. Journal of Biomaterials Science-Polymer Edition,2018,29(7-9):762-774. doi: 10.1080/09205063.2017.1348738 [78] Jo S B, Erdenebileg U, Dashnyam K, et al. Nano-graphene oxide/polyurethane nanofibers: mechanically flexible and myogenic stimulating matrix for skeletal tissue engineering[J]. Journal of Tissue Engineering,2020,11:2041731419900424. [79] Azizi M, Navidbakhsh M, Hosseinzadeh S, et al. Cardiac cell differentiation of muscle satellite cells on aligned composite electrospun polyurethane with reduced graphene oxide[J]. Journal of Polymer Research,2019,26(11):258. doi: 10.1007/s10965-019-1936-9 [80] Shin Y C, Lee J H, Jin L H, et al. Stimulated myoblast differentiation on graphene oxide-impregnated PLGA-collagen hybrid fibre matrices[J]. Journal of Nanobiotechnology,2015,13:21. doi: 10.1186/s12951-015-0081-9 [81] Shin Y C, Kim C, Song S-J, et al. Ternary aligned nanofibers of RGD peptide-displaying M13 bacteriophage/PLGA/graphene oxide for facilitated myogenesis[J]. Nanotheranostics,2018,2(2):144-156. doi: 10.7150/ntno.22433 [82] Nakayama K H, Shayan M, Huang N F. Engineering biomimetic materials for skeletal muscle repair and regeneration[J]. Advanced Healthcare Materials,2019,8(5):1801168. [83] Ge Zhen, Zou Gang, Liu Yi, et al. Tissue-engineering scaffolds: characteristics and applications in tissue engineering[J]. Chinese Journal of Tissue Engineering Research,2018,22(26):4222-4228. [84] Zhang S, Yang Q, Zhao W, et al. In vitro and in vivo biocompatibility and osteogenesis of graphene-reinforced nanohydroxyapatite polyamide66 ternary biocomposite as orthopedic implant material[J]. International Journal of Nanomedicine,2016,11:3179-3189. doi: 10.2147/IJN.S105794 [85] Rethinam S, Wilson Aruni A, Vijayan S,et al. Enhanced bone regeneration using an electrospun nanofibrous membrane – a novel approach[J]. Journal of Drug Delivery Science and Technology,2019,53:101163-101163. doi: 10.1016/j.jddst.2019.101163 [86] Talukdar Y, Rashkow J T, Patel S, et al. Nanofilm generated non-pharmacological anabolic bone stimulus[J]. Journal of Biomedical Materials Research Part A,2020,108(1):178-186. doi: 10.1002/jbm.a.36807 [87] Kurantowicz N, Strojny B, Sawosz E, et al. Biodistribution of a high dose of diamond, graphite, and graphene oxide nanoparticles after multiple intraperitoneal injections in rats[J]. Nanoscale Research Letters,2015,10(1):398. doi: 10.1186/s11671-015-1107-9 [88] Richard J. Chitosan-graphene nanocomposite microneedle arrays for transdermal drug delivery[D]. Doctoral Degree, University of Sheffield, 2015. [89] Yadav I, Nayak S K, Rathnam V S S, et al. Reinforcing effect of graphene oxide reinforcement on the properties of poly (vinyl alcohol) and carboxymethyl tamarind gum based phase-separated film[J]. Journal of the Mechanical Behavior of Biomedical Materials,2018,81:61-71. doi: 10.1016/j.jmbbm.2018.02.021 [90] Ganguly S, Das P, Maity P P, et al. Green reduced graphene oxide toughened semi-IPN monolith hydrogel as dual responsive drug release system: rheological, physicomechanical, and electrical evaluations[J]. The Journal of Physical Chemistry Part B,2018,122(29):7201-7218. doi: 10.1021/acs.jpcb.8b02919 [91] Altinbasak I, Jijie R, Barras A, et al. Reduced graphene-oxide-embedded polymeric nanofiber mats: an "on-demand" photothermally triggered antibiotic release platform[J]. ACS Applied Materials & Interfaces,2018,10(48):41098-41106. [92] Pagneux Q, Ye R, Chengnan L, et al. Electrothermal patches driving the transdermal delivery of insulin[J]. Nanoscale Horizons,2020,5(4):663-670. doi: 10.1039/C9NH00576E [93] Geetha Bai R, Ninan N, Muthoosamy K, et al. Graphene: a versatile platform for nanotheranostics and tissue engineering[J]. Progress in Materials Science,2018,91:24-69. doi: 10.1016/j.pmatsci.2017.08.004 [94] Wang M, Wiraja C, Wee M, et al. Hairpin-structured probe conjugated nano-graphene oxide for the cellular detection of connective tissue growth factor mRNA[J]. Analytica Chimica Acta,2018,1038:140-147. doi: 10.1016/j.aca.2018.07.016 [95] Qi X, Liu H, Guo W, et al. New opportunities: second harmonic generation of boron-doped graphene quantum dots for stem cells imaging and ultraprecise tracking in wound healing[J]. Advanced Functional Materials,2019,29(37):1902235.1-1902235.10. [96] Guo J, Zhong Z, Li Y, et al. "Three-in-one" SERS adhesive tape for rapid sampling, release, and detection of wound infectious pathogens[J]. ACS Applied Materials & Interfaces,2019,11(40):36399-36408. [97] You X, Yang S, Li J, et al. Green and mild oxidation: an efficient strategy toward water-dispersible graphene[J]. ACS Applied Materials & Interfaces,2017,9(3):2856-2866. [98] Ramadas M, Bharath G, Ponpandian N, et al. Investigation on biophysical properties of hydroxyapatite/graphene oxide (HAp/GO) based binary nanocomposite for biomedical applications[J]. Materials Chemistry and Physics,2017,199:179-184. doi: 10.1016/j.matchemphys.2017.07.001 [99] Hussein K H, Abdelhamid H N, Zou X, et al. Ultrasonicated graphene oxide enhances bone and skin wound regeneration[J]. Materials Science & Engineering C-Materials for Biological Applications,2019,94:484-492. [100] Xue J, Wang X, Wang E, et al. Bioinspired multifunctional biomaterials with hierarchical microstructure for wound dressing[J]. Acta Biomaterialia,2019,100:270-279. doi: 10.1016/j.actbio.2019.10.012 [101] Narayanan K B, Kim H D, Han S S. Biocompatibility and hemocompatibility of hydrothermally derived reduced graphene oxide using soluble starch as a reducing agent[J]. Colloids and Surfaces B: Biointerfaces,2020,185:110579. doi: 10.1016/j.colsurfb.2019.110579 [102] Castilho C J, Li D, Liu M, et al. Mosquito bite prevention through graphene barrier layers[J]. Proceedings of The National Academy of Sciences of The United States of America,2019,116(37):18304-18309. doi: 10.1073/pnas.1906612116 [103] Zhang G, Hu J, Ren T, et al. Microstructural and tribological properties of a dopamine hydrochloride and graphene oxide coating applied to multifilament surgical sutures[J]. Polymers (Basel),2020,12(8):1630. doi: 10.3390/polym12081630 [104] Heo C, Lee S Y, Jo A, et al. Flexible, transparent, and noncytotoxic graphene electric field stimulator for effective cerebral blood volume enhancement[J]. ACS Nano,2013,7(6):4869-4878. doi: 10.1021/nn305884w -





 下载:
下载: