Effect of the air oxidation stabilization of pitch on the microstructure and sodium storage of hard carbons
-
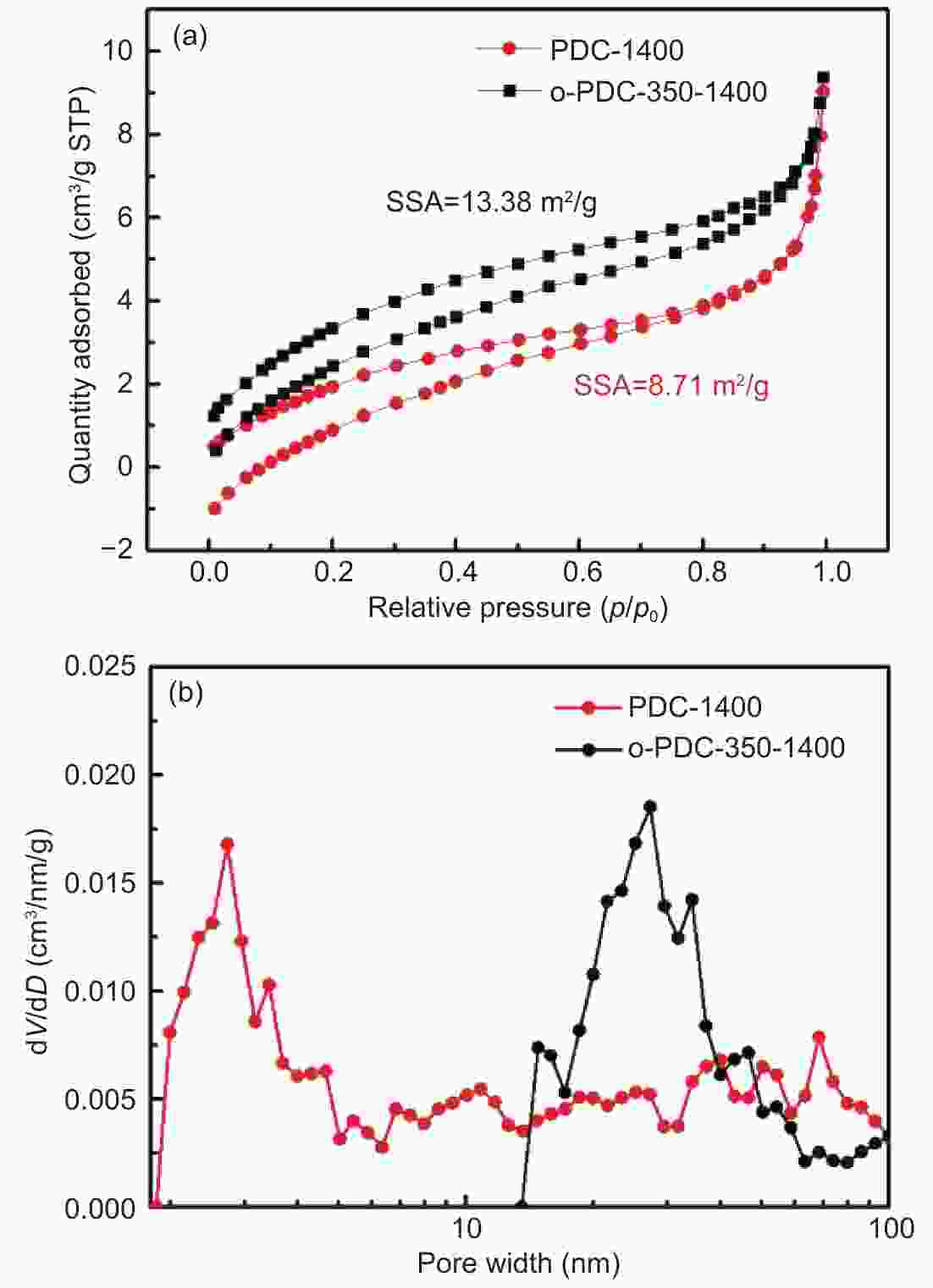
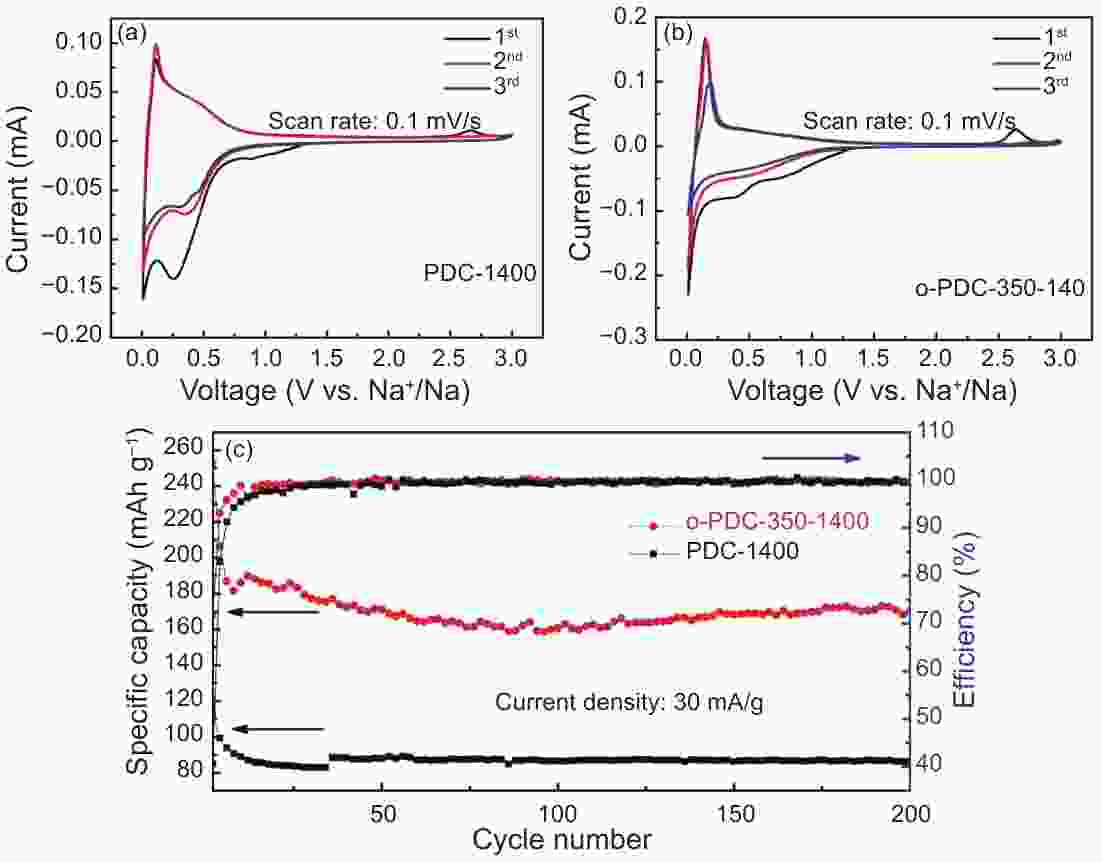
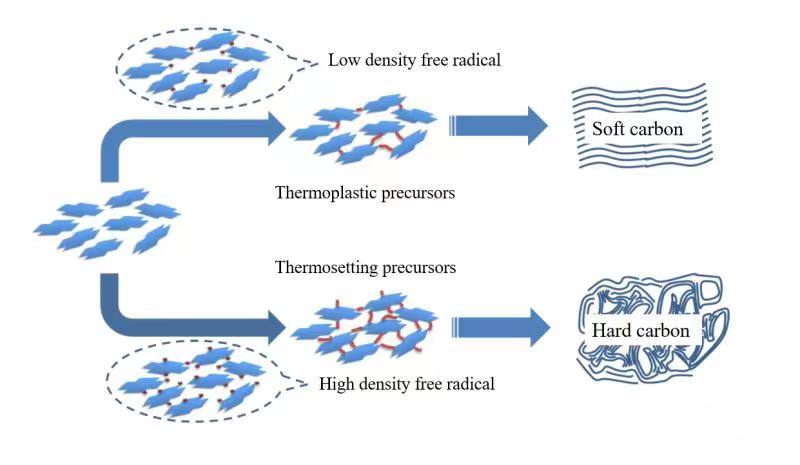
摘要: 以石油沥青为原料,通过空气氧化稳定化及炭化方法成功制备出钠离子电池用硬炭负极材料。研究了不同氧化稳定化温度下样品组成、结构的变化,及其对炭化样品形貌、结构和储钠性能的影响。结果表明,空气氧化处理可以引入大量的含氧官能团,诱导脱氢缩合和氧化交联反应的发生,使石油沥青发生由热塑性向热固性的转化。空气氧化稳定化处理有效地阻碍了沥青在高温炭化中固有的石墨化倾向,使碳层堆叠变得无序、同时产生更多的缺陷位。电化学测试结果表明,在100 mA g−1的电流密度下,与直接炭化样品PDC-1400相比,350 ℃氧化稳定化、1400 ℃炭化的硬炭样品o-PDC-350-1400的比容量提升约1.8倍(达到276.8 mAh g−1);首效提高22%(达到73.38%)。样品o-PDC-350-1400循环200圈后,充电比容量达170.2 mAh g−1,具有良好的循环稳定性。Abstract: Hard carbon anode materials for sodium ion batteries were prepared from petroleum pitch by air oxidation stabilization followed by carbonization. The effects of the oxidation stabilization temperature on the compositions and microstructures of the oxidized samples, as well as on the morphology, microstructure and sodium storage property of the carbonized samples were investigated. Results show that air oxidation introduces a large number of oxygen-containing functional groups, induces dehydrogenation condensation and oxidative crosslinking reactions, and transforms the petroleum asphalt from thermoplastic to thermosetting. The air oxidation stabilization treatment effectively hinders the inherent tendency of asphalt to graphitize during high temperature carbonization, resulting in carbons with randomly oriented carbon layers with more defects. Electrochemical tests show that o-PDC-350-1400 (oxidation stabilization at 350 °C, carbonization at 1400 °C) has a high charging specific capacity of 276.8 mAh g−1 at 100 mA g−1 and a high initial coulombic efficiency of 73.38%. Compared with sample PDC-1400 that was directly carbonized at 1400 °C, the charging specific capacity was increased by about 1.8 times and the initial coulombic efficiency was increased by 22%. The charging specific capacity of o-PDC-350-1400 after 200 cycles reached 170.2 mAh g−1, indicating good cycling stability.
-
Key words:
- Petroleum asphalt /
- Oxidation stabilization /
- Hard carbon /
- Sodium ion battery
-
Table 1. Element analysis, weight gain ratio and carbon yield of samples after oxidation stabilization.
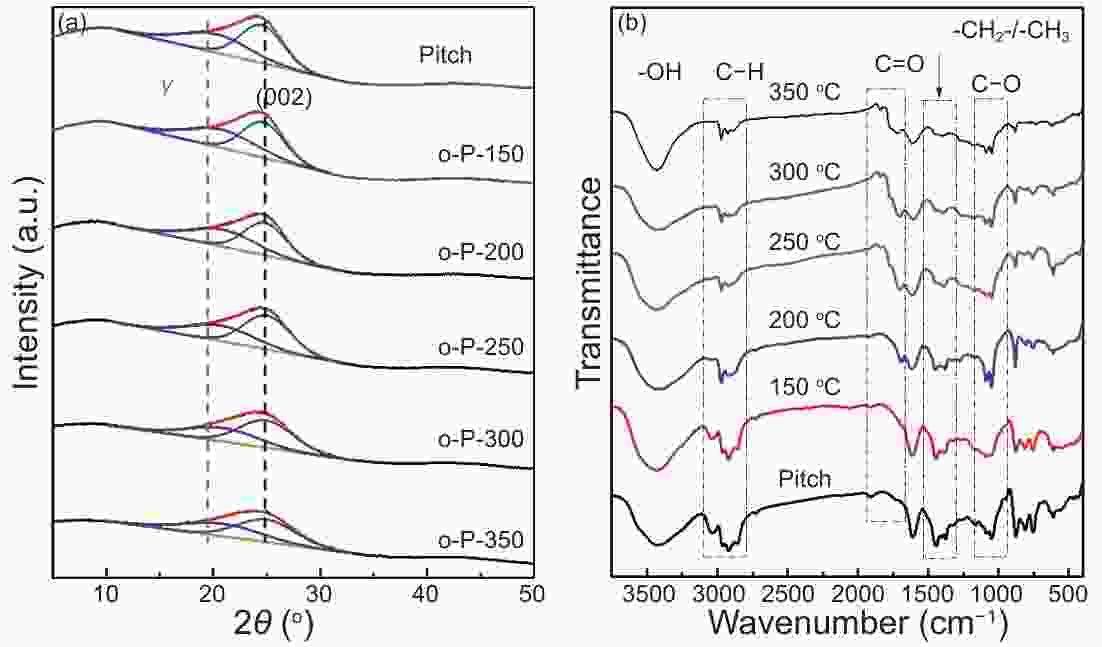
Samples Elemental composition(%) C/H Oxidation weight gain(%) Yield of carbon(%) N C H O* Pitch 0.13 94.34 5.02 0.51 1.57 - 53.3 o-P-150 0.25 91.41 5.08 3.26 1.50 0.4 51.8 o-P-200 0.25 88.18 4.7 6.87 1.56 3.3 66.4 o-P-250 0.23 81.32 3.24 15.21 2.09 9.4 72.3 o-P-300 0.25 74.58 2.31 22.86 2.69 6.4 67.0 o-P-350 0.35 70.91 1.32 27.42 4.48 −7.1 61.5 Note: * Calculated by subtraction method Table 2. Crystalline parameters of pristine petroleum asphalt and samples after oxidation stabilization.
Samples Fa Dm(nm) Dr(nm) La(nm) Lc(nm) Pitch 0.56 0.362 0.549 1.113 0.544 o-P-150 0.54 0.361 0.534 1.154 0.564 o-P-200 0.55 0.358 0.531 1.151 0.563 o-P-250 0.60 0.356 0.535 1.040 0.509 o-P-300 0.60 0.357 0.529 0.903 0.442 o-P-350 0.57 0.356 0.530 0.827 0.405 Table 3. Crystalline parameters and electrochemical performance of o-PDC-T-1400 series.
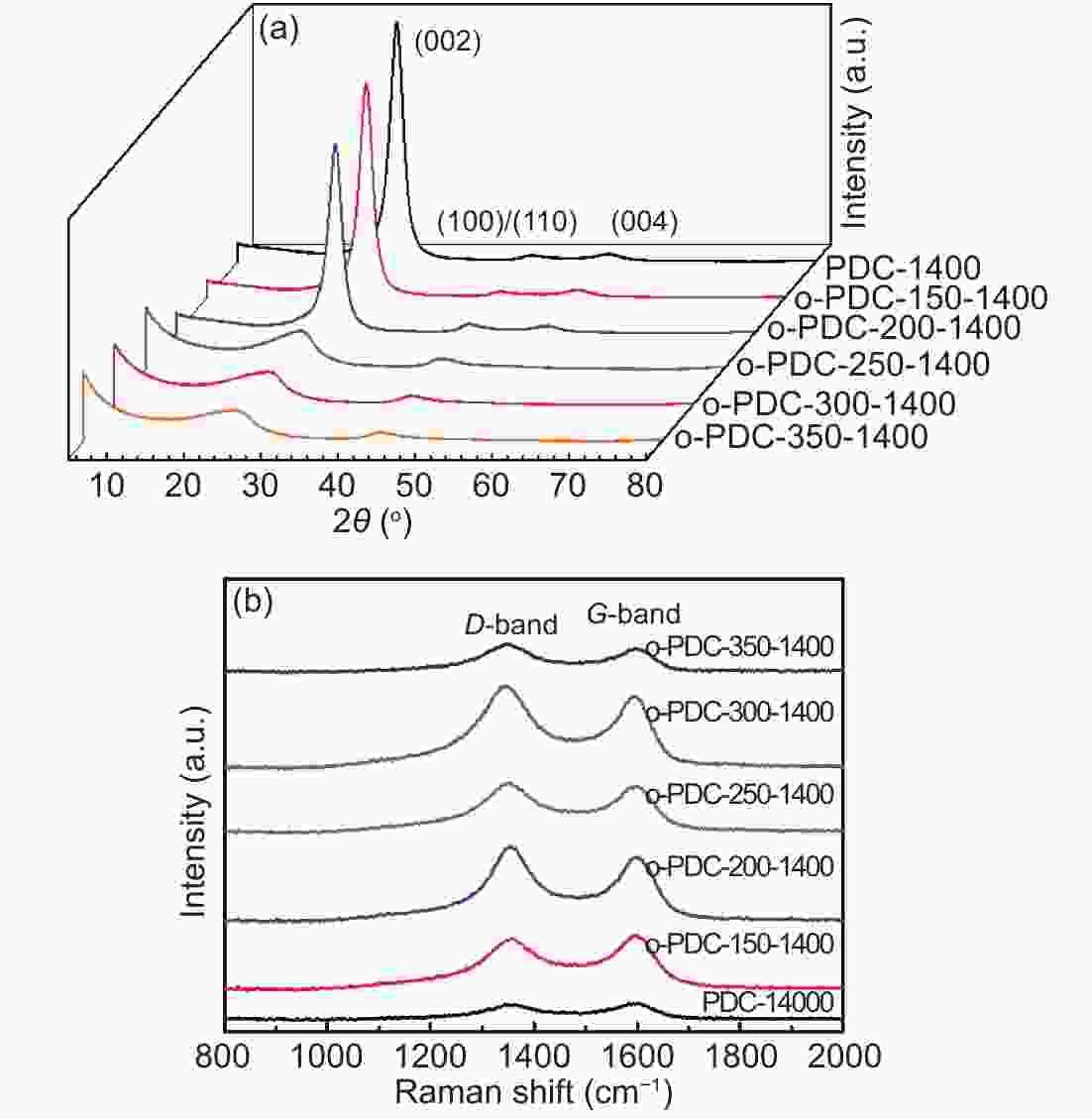
Samples Lc (nm) La (nm) d002(nm) ID/IG RC (mAh g−1 ) ICE (%) PDC-1400 3.59 4.739 0.347 0.95 99.6 51.45 o-PDC-150-1400 3.532 4.931 0.347 0.94 99.8 51.92 o-PDC-200-1400 3.484 5.292 0.347 0.95 93.1 48.70 o-PDC-250-1400 1.467 3.292 0.355 1.09 221.3 73.81 o-PDC-300-1400 1.382 3.399 0.356 1.14 230.3 73.02 o-PDC-350-1400 1.313 3.440 0.363 1.20 276.8 73.38 -
[1] Yamamoto H, Muratsubaki S, Kubota K, et al. Synthesizing higher-capacity hard-carbons from cellulose for Na- and K-ion batteries[J]. Journal of Materials Chemistry A,2018,6(35):16844-16848. doi: 10.1039/C8TA05203D [2] Xie F, Xu Z, Guo Z, et al. Hard carbons for sodium-ion batteries and beyond[J]. Prog Energy,2020,2:042002. [3] Pei L, Cao H, Yang L, et al. Hard carbon derived from waste tea biomass as high-performance anode material for sodium-ion batteries[J]. Ionics,2020,26:5535-5542. [4] Zhao G, Yu D, Zhang H, et al. Sulphur-doped carbon nanosheets derived from biomass as high-performance anode materials for sodium-ion batteries[J]. Nano Energy,2020,67:104219. [5] Yu Z, Lyu Y, Wang Y, et al. Hard carbon micro-nano tubes derived from kapok fiber as anode materials for sodium-ion batteries and the sodium-ion storage mechanism[J]. Chem Commun,2020,56:778-781. [6] Sun S, Yao J, Lin Y, et al. Triconstituent co-assembly to hierarchically porous carbons as high-performance anodes for sodium-ion batteries[J]. Journal of Alloys and Compounds,2019,771:140-146. doi: 10.1016/j.jallcom.2018.08.284 [7] Yang L. Modified Asphalt and its Emulsification Technology [M]. China Communications Press, 2004. [8] Li Y, Hu Y, Li H, et al. A superior low-cost amorphous carbon anode made from pitch and lignin for sodium-ion batteries[J]. Journal of Materials Chemistry A,2016,4(1):96-104. doi: 10.1039/C5TA08601A [9] Li Y, Mu L, Hu Y, et al. Pitch-derived amorphous carbon as high performance anode for sodium-ion batteries[J]. Energy Storage Materials,2016,2:139-145. doi: 10.1016/j.ensm.2015.10.003 [10] Miao Y, Zong J, Liu X. Phosphorus-doped pitch-derived soft carbon as an anode material for sodium ion batteries[J]. Materials Letters,2017,188:355-358. doi: 10.1016/j.matlet.2016.11.110 [11] Fernández A, Granda M, Bermejo J, et al. Air-blowing of anthracene oil for carbon precursors[J]. Carbon,2000,38(9):1315-1322. doi: 10.1016/S0008-6223(99)00264-X [12] Zhang D. Production and Application of Petroleum Asphalt [M]. China Petrochemical Press, 2001. [13] Zhang Y, Liang W, LIU C. Investigation of asphaltenes in Gudao and Shanjiasi asphalts by xrd[J]. Acta Petrolei Sinica(Petroleum Processing Section),1989(3):7-14. [14] Ebert L, Scanlon J, Mills D. X-ray diffraction of n-paraffins and stacked aromatic molecules: insights into the structure of petroleum asphaltenes[J]. Liquid Fuels Technology,1984,2(3):257-286. doi: 10.1080/07377268408915353 [15] Yen T, Erdman J, Pollack S. Investigation of the structure of petroleum asphaltenes by x-ray diffraction[J]. Analytical Chemistry,1961,33(11):1587-1594. doi: 10.1021/ac60179a039 [16] Drbohlav J, Stevenson W. The oxidative stabilization and carbonization of a synthetic mesophase pitch, part II: The carbonization process[J]. Carbon,1995,33(5):713-731. doi: 10.1016/0008-6223(95)00012-3 [17] Guan T, Zhang G, Zhao J, et al. Insight into the oxidative reactivity of pitch fractions for predicting and optimizing the oxidation stabilization of pitch[J]. Fuel,2019,242:184-194. doi: 10.1016/j.fuel.2019.01.034 [18] Qiu S, Xiao L, Sushko M, et al. Manipulating adsorption-insertion mechanisms in nanostructured carbon materials for high-efficiency sodium ion storage[J]. Advanced Energy Materials,2017,7(17):1700403. doi: 10.1002/aenm.201700403 [19] Bommier C, Surta T, Dolgos M, et al. New mechanistic insights on Na-ion storage in nongraphitizable carbon[J]. Nano Letters,2015,15(9):5888-5892. doi: 10.1021/acs.nanolett.5b01969 [20] Qi Y, Lu Y, Ding F, et al. Slope-dominated carbon anode with high specific capacity and superior rate capability for high safety Na-ion batteries[J]. Angewandte Chemie International Edition,2019,58(13):4361-4365. doi: 10.1002/anie.201900005 [21] Kim C, Verma D, Ho Nam D, et al. Conformal carbon layer coating on well-dispersed Si nanoparticles on graphene oxide and the enhanced electrochemical performance[J]. Journal of Industrial and Engineering Chemistry,2017,52:260-269. doi: 10.1016/j.jiec.2017.03.055 -





 下载:
下载: