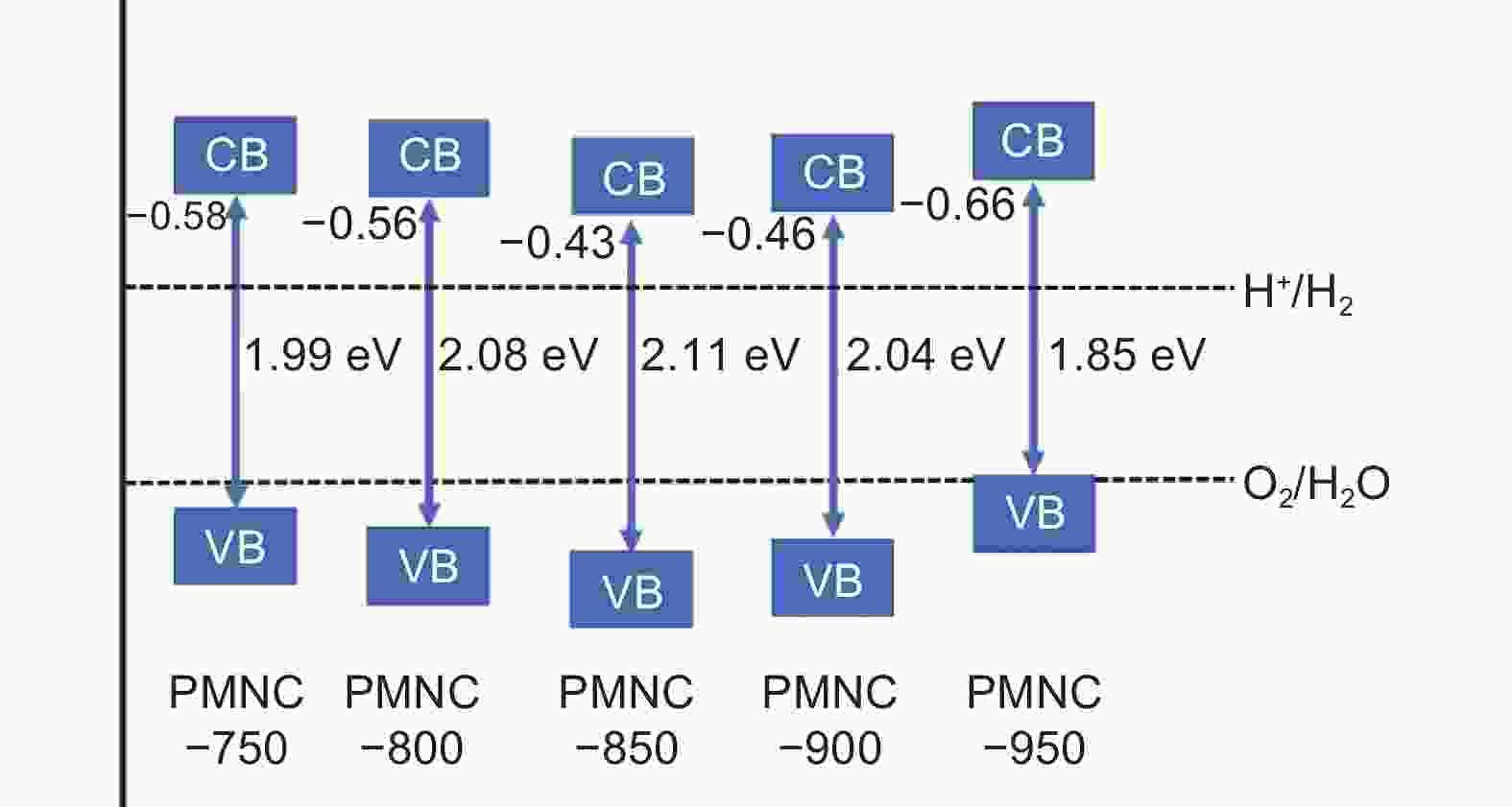
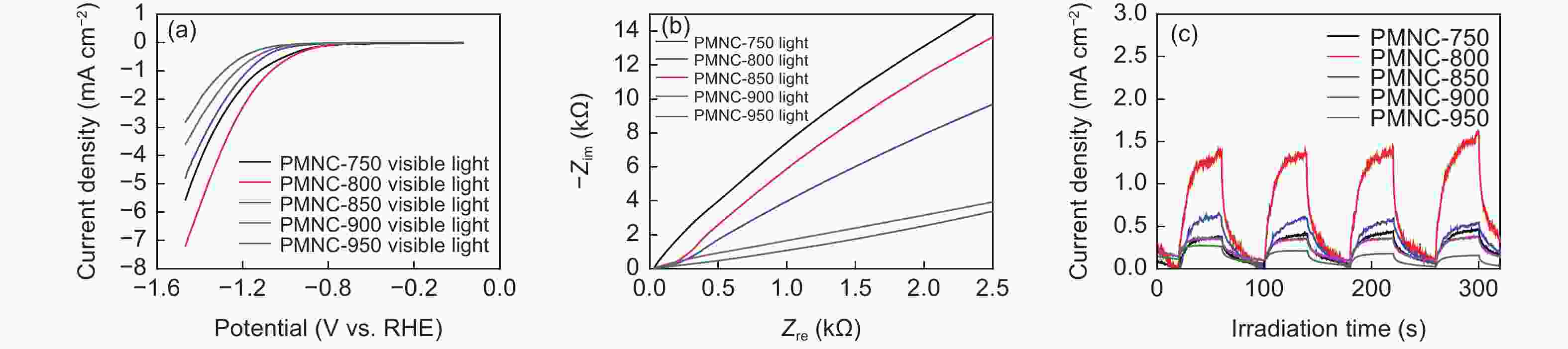
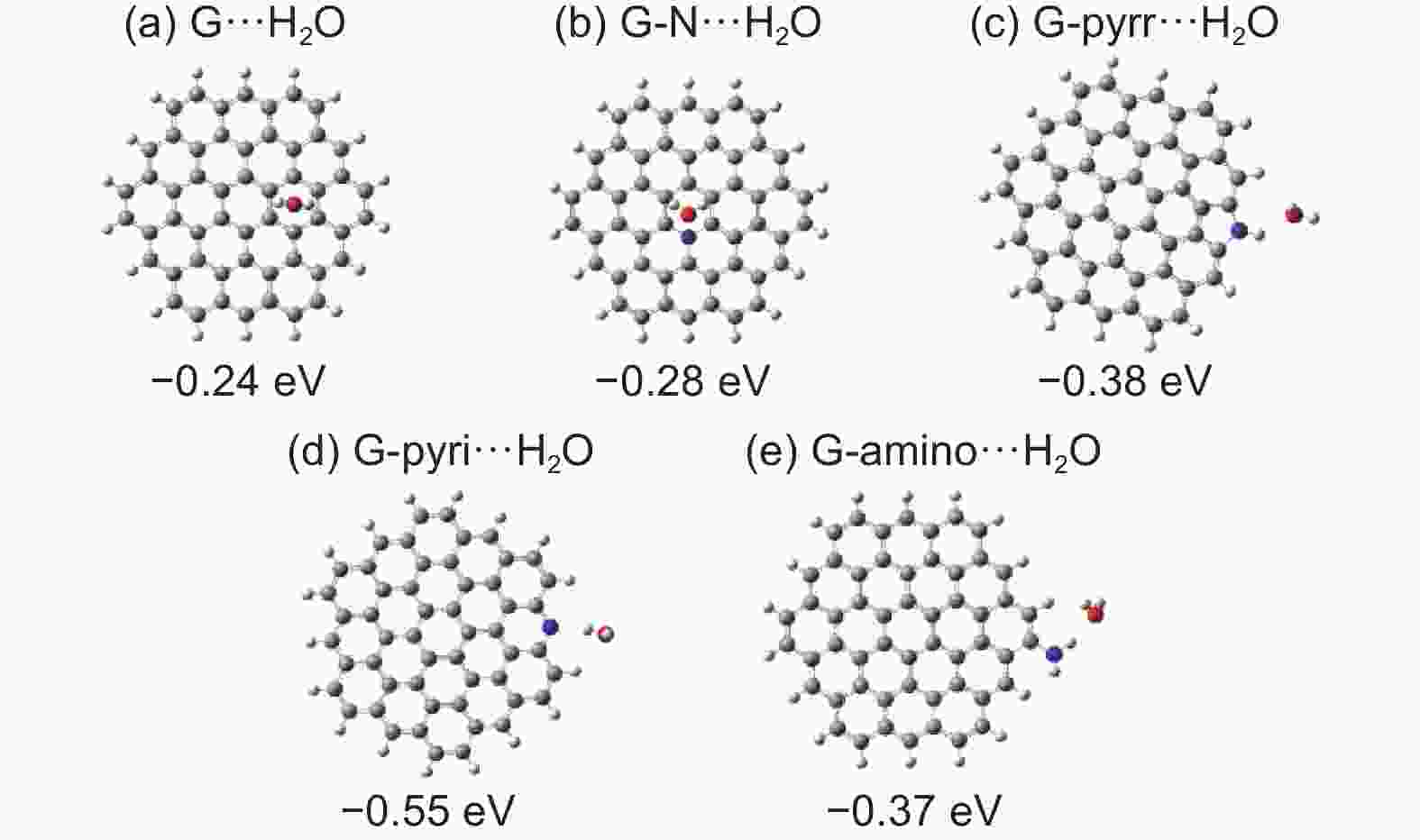
| [1] |
Kuang P Y, Sayed M, Fan J J, et al. 3D graphene‐based H2 production photocatalyst and electrocatalyst[J]. Adv Energy Mater,2020,10(14):1903802.
|
| [2] |
Nasir M S, Yang G R, Ayub I, et al. Recent development in graphitic carbon nitride based photocatalysis for hydrogen generation[J]. Appl. Catal. B Environ,2019,257:117855.
|
| [3] |
Du Z, Shen S L, Tang Z H, et al. Graphene quantum dots-based heterogeneous catalysts[J]. New Carbon Materials,2021,36(3):449-467.
|
| [4] |
Wu H, Tan H L, Toe C Y, et al. Photocatalytic and photoelectrochemical systems: Similarities and differences[J]. Adv Mater,2020,32(18):1904717.
|
| [5] |
Wang T T, Zhao Q D, Fu Y Y, et al. Carbon‐rich nonprecious metal single atom electrocatalysts for CO2 reduction and hydrogen evolution [J]. Small Methods. 2019, 3(10): 1900210.
|
| [6] |
Liao G F, Gong Y, Zhang L, et al. Semiconductor polymeric graphitic carbon nitride photocatalysts: The “holy grail” for the photocatalytic hydrogen evolution reaction under visible light[J]. Energy Environ Sci,2019,12(7):2080-2147.
|
| [7] |
Jiang Z F, Sun H L, Wang T Q, et al. Nature-based catalyst for visible-light-driven photocatalytic CO2 reduction[J]. Energy Environ Sci,2018,11(9):2382-2389.
|
| [8] |
Liu G Y, Li K K, Jia J, et al. Coal-based graphene as a promoter of TiO2 for photocatalytic degradation of organic dyes[J]. New Carbon Mater,2021,36(2):1-11.
|
| [9] |
Wang L, Xu X, Cheng Q F, et al. Near-infrared-driven photocatalysts: Design, construction, and applications[J]. Small,2021,17(9):e1904107.
|
| [10] |
Meng F P, Liu Y Z, Wang J, et al. Temperature dependent photocatalysis of g-C3N4, TiO2 and ZnO: Differences in photoactive mechanism[J]. J Colloid Interface Sci,2018,532:321-330.
|
| [11] |
Bie C B, Yu H G, Cheng B, et al. Design, fabrication, and mechanism of nitrogen-doped graphene-based photocatalyst[J]. Adv Mater,2021,33(9):e2003521.
|
| [12] |
Wang T Y, Huang H B, Li H L, et al. Carbon materials for solar-powered seawater desalination[J]. New Carbon Mater,2021,36(4):683-701.
|
| [13] |
Zhu C, Zhu M M, Sun Y, et al. Defects induced efficient overall water splitting on a carbon-based metal-free photocatalyst[J]. Appl Catal. B Environ,2018,237:166-174.
|
| [14] |
Wang Z, Li C, Domen K. Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting[J]. Chem Soc Rev,2019,48(7):2109-2125.
|
| [15] |
Afroz K, Moniruddin M, BakranKudaibergenov S, et al. A heterojunction strategy to improve the visible light sensitive water splitting performance of photocatalytic materials[J]. J Mater Chem A,2018,6(44):21696-21718.
|
| [16] |
Wang H L, Zhang L S, Chen Z G, et al. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances[J]. Chem Soc Rev,2014,43(15):5234-5244.
|
| [17] |
Takanabe K. Photocatalytic water splitting: Quantitative approaches toward photocatalyst by design[J]. ACS Catal,2017,7(11):8006-8022.
|
| [18] |
Zhang P, Lou X W. Design of heterostructured hollow photocatalysts for solar‐to‐chemical energy conversion[J]. Adv Mater,2019,31(29):1900281.
|
| [19] |
Kranz C, Wachtler M. Characterizing photocatalysts for water splitting: From atoms to bulk and from slow to ultrafast processes[J]. Chem Soc Rev,2021,50(2):1407-1437.
|
| [20] |
Ding J, Wang H L, Li Z, et al. Carbon nanosheet frameworks derived from peat moss as high performance sodium ion battery anodes[J]. ACS Nano,2013,7(12):11004-11015.
|
| [21] |
Frisch M J, Trucks H B S G W, Scuseria G E. Cheeseman. Gaussian 16, Revision A. 03[Z], Gaussian, Inc. Wallingford CT. 2016.
|
| [22] |
Becke A D. Density-functional thermochemistry[J]. J Chem. Phys,1993,98(7):5648-5652.
|
| [23] |
Grimme S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction[J]. J Comput Chem,2006,27(15):1787-1799.
|
| [24] |
Schwabe T, Grimme S. Double-hybrid density functionals with long-range dispersion corrections: Higher accuracy and extended applicability[J]. Phys Chem Chem Phys,2007,9(26):3397-3406.
|
| [25] |
Qiu B C, Zhu Q H, Du M M, et al. Efficient solar light harvesting CdS/Co9S8 hollow cubes for Z-Scheme photocatalytic water splitting[J]. Angew Chem Int Ed,2017,56(10):2684-2688.
|
| [26] |
Sun J H, Zhang J S, Zhang M W, et al. Bioinspired hollow semiconductor nanospheres as photosynthetic nanoparticles[J]. Nat Commun,2012,3(1):1139.
|
| [27] |
Tu W G, Zhou Y, Liu Q, et al. Robust hollow spheres consisting of alternating titania nanosheets and graphene nanosheets with high photocatalytic activity for CO2 conversion into renewable fuels[J]. Adv Funct Mater,2012,22(6):1215-1221.
|
| [28] |
Wang B, Jiang Z F, Yu J C. Treated rape pollen: A metal-free visible-light-driven photocatalyst from nature for efficient water disinfection[J]. J Mater Chem A,2019,7(15):9335-9344.
|
| [29] |
Chen L C, Teng C Y, Lin C Y, et al. Architecting nitrogen functionalities on graphene oxide photocatalysts for boosting hydrogen production in water decomposition process[J]. Adv Energy Mater,2016,6(22):1600719.
|
| [30] |
Sun J, Zheng G Y, Lee H W, et al. Formation of stable phosphorus-carbon bond for enhanced performance in black phosphorus nanoparticle-graphite composite battery anodes[J]. Nano Lett,2014,14(8):4573-4580.
|
| [31] |
Choudhary S, Mungse H P, Khatri O P. Dispersion of alkylated graphene in organic solvents and its potential for lubrication applications[J]. J Mater Chem,2012,22(39):21032.
|
| [32] |
Noel S, Liberelle B, Robitaille L, et al. Quantification of primary amine groups available for subsequent biofunctionalization of polymer surfaces[J]. Bioconjug Chem,2011,22(8):1690-1699.
|
| [33] |
Yeh T F, Chen S J, Teng H. Synergistic effect of oxygen and nitrogen functionalities for graphene-based quantum dots used in photocatalytic H2 production from water decomposition[J]. Nano Energy,2015,12:476-485.
|
| [34] |
Tossi C, Hällström L, Selin J, et al. Size- and density-controlled photodeposition of metallic platinum nanoparticles on titanium dioxide for photocatalytic applications[J]. J Mater Chem,2019,7(24):14519-14525.
|
| [35] |
Li X G, Bi W T, Zhang L, et al. Single-atom Pt as Co-catalyst for enhanced photocatalytic H2 evolution[J]. Adv Mater,2016,28(12):2427-2431.
|
| [36] |
Gao Z Q, Chen K Y, Wang L, et al. Aminated flower-like ZnIn2S4 coupled with benzoic acid modified g-C3N4 nanosheets via covalent bonds for ameliorated photocatalytic hydrogen generation[J]. Appl Catal B Environ,2020,268:118462.
|
| [37] |
Zhang L H, Jin Z Y, Huang S L, et al. Bio-inspired carbon doped graphitic carbon nitride with booming photocatalytic hydrogen evolution[J]. Appl Catal B Environ,2019,246:61-71.
|
| [38] |
Yang Y, Zhang C, Huang D L, et al. Boron nitride quantum dots decorated ultrathin porous g-C3N4: Intensified exciton dissociation and charge transfer for promoting visible-light-driven molecular oxygen activation[J]. Appl Catal B Environ,2019,245:87-99.
|
 20210216-Supporting information.pdf
20210216-Supporting information.pdf







 下载:
下载: