The oxidation reaction mechanism and its kinetics for a carbonaceous precursor prepared from ethylene tar for use as an anode material for lithium-ion batteries
-
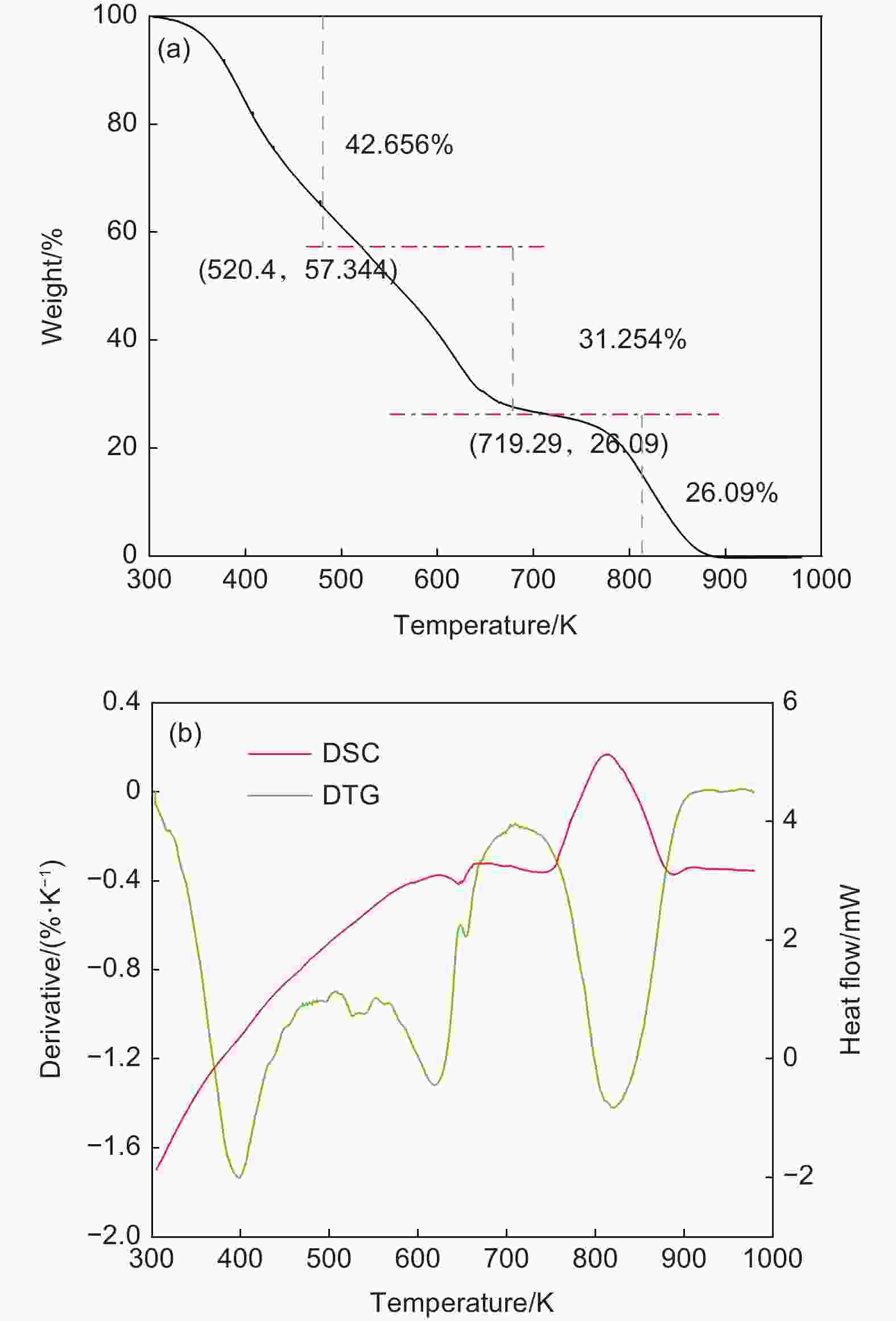
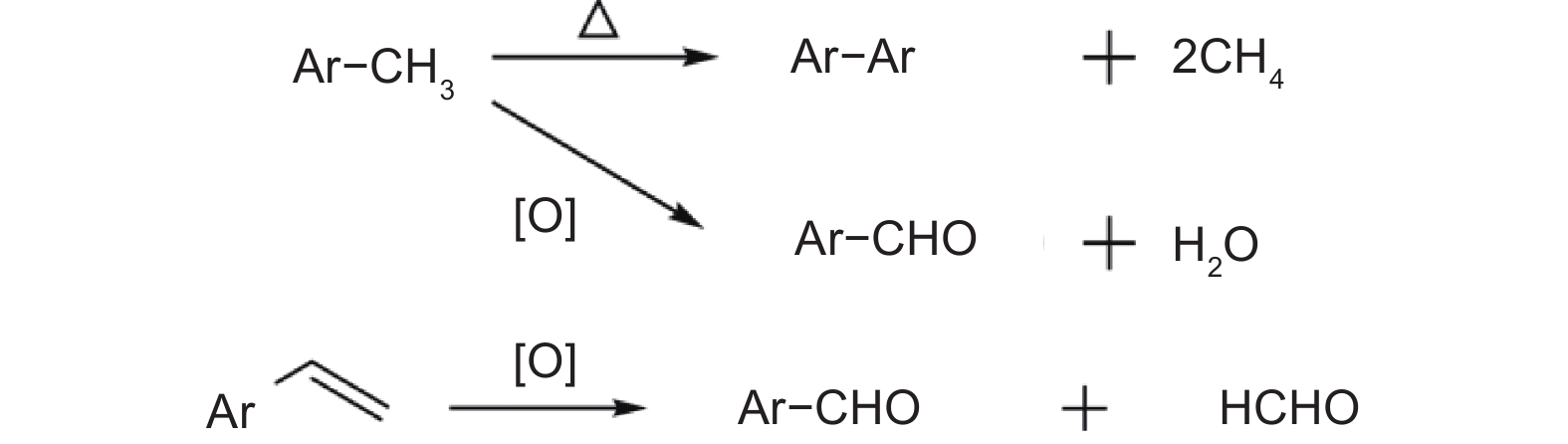
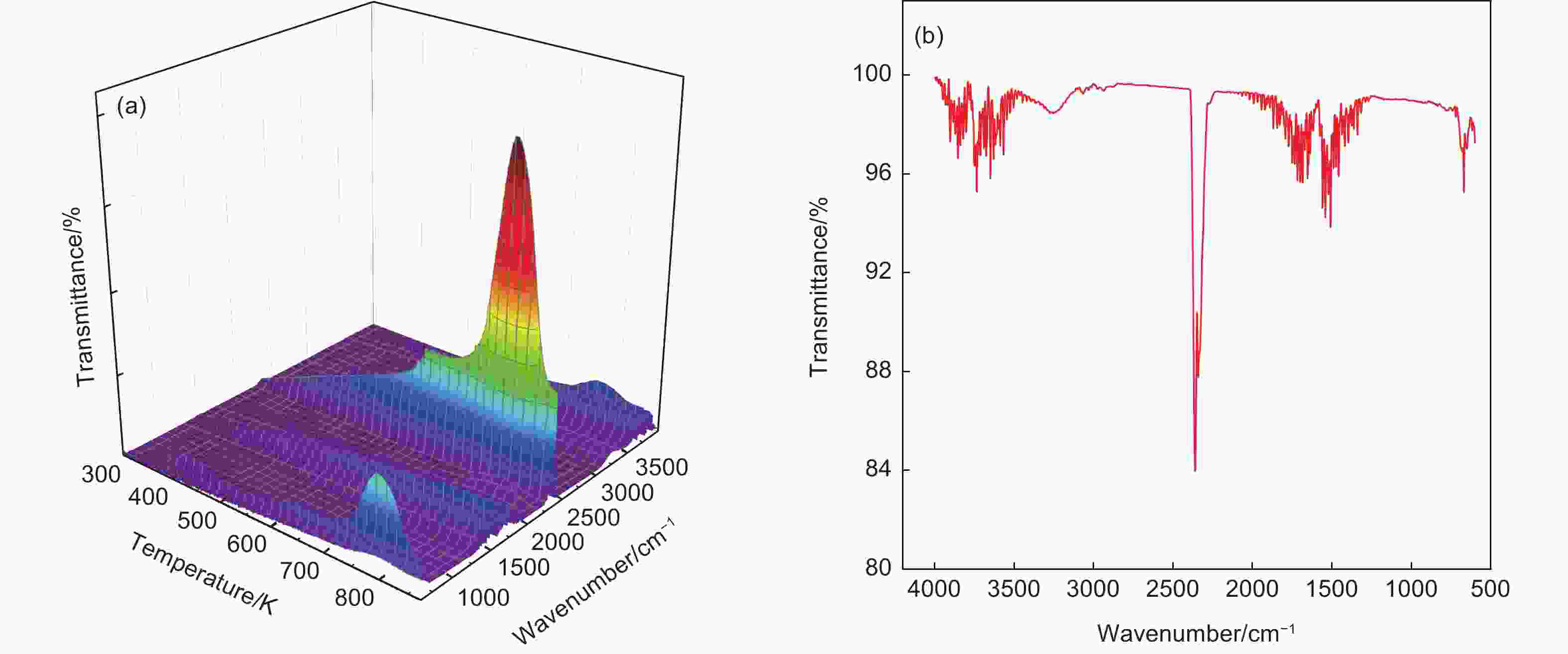
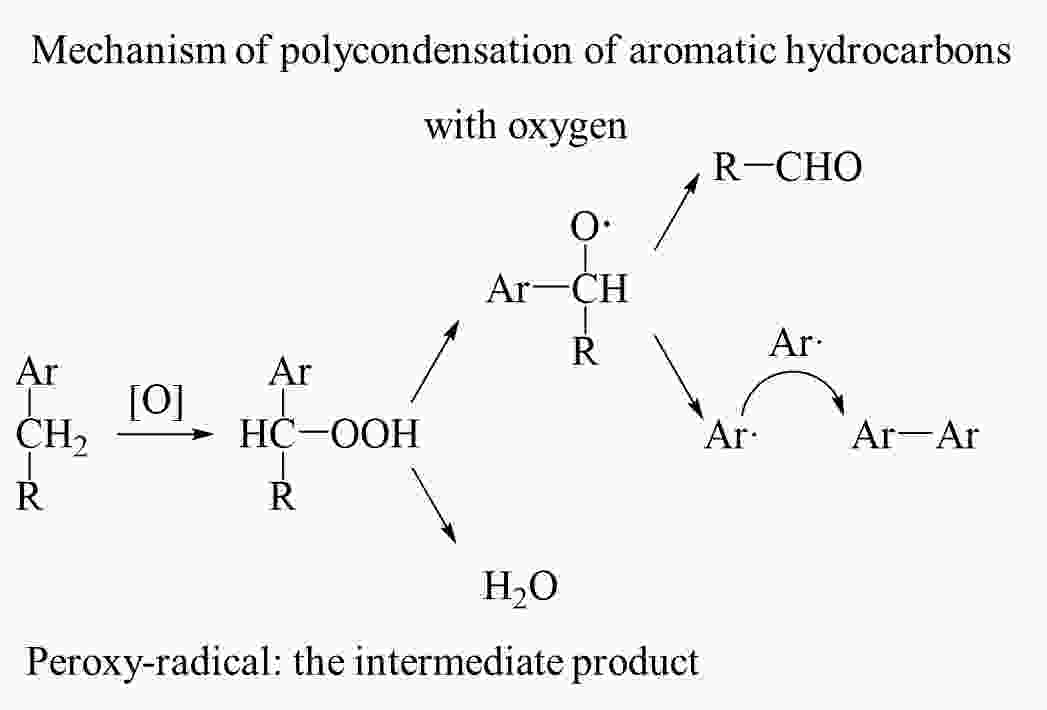
摘要: 为了得到优质的碳质前驱体,研究了乙烯焦油在空气中的氧化反应机理及其反应动力学,并制备出高软化点沥青应用于锂离子电池负极石墨材料的包覆改性。根据热重曲线将乙烯焦油的氧化过程分成350−550、550−700和700−900 K三个阶段,并采用质谱和红外技术对不同反应温度下的尾气成份进行在线分析以揭示乙烯焦油在空气中的氧化反应机理。根据不同反应温度下乙烯焦油与氧气的热失重曲线,整个反应过程被分为4个阶段,进一步利用Coats-Redfern等转化率法分析17种常用反应动力学模型与实验数据的拟合度,筛选出最适宜表达乙烯焦油与氧气的反应动力学模型。结果表明:(1)在乙烯焦油的氧化过程中,芳香化合物的支链先与氧气反应生成醇类、醛类小分子化合物和含有过氧自由基的芳香化合物,然后含有过氧自由基的芳香化合物进行热缩聚反应形成分子量更大的芳香族化合物;(2)可采用四级反应模型描述乙烯焦油的前3阶段反应动力学,活化能分别为47.33、18.69和9.00 kJ·mol−1;可采用三维扩散模型描述第4阶段的反应动力学,其活化能为88.37 kJ·mol−1。(3)经所制沥青包覆改性后,石墨负极循环300圈后的容量保持率由51.54%增长为79.07%。Abstract: The oxidation reaction mechanism and its kinetics for ethylene tar were investigated in order to obtain a suitable anode material for Li-ion batteries. The oxidation of ethylene tar was divided into 3 stages (350–550, 550–700 and 700–900 K) according to the thermogravimetric curve. To reveal the oxidation reaction mechanism, the components of the gases evolved at different stages were analyzed by mass spectrometry and infrared technology. Based on these results the reaction was divided into 4 stages (323–400, 400–605, 605–750 and 750–860 K) to perform simulation calculations of the kinetics. Using the iso-conversion method (Coats-Redfern) to analyze the linear regression rates (R2) between 17 common reaction kinetics models and experimental data, an optimum reaction kinetics model for expressing the oxidation of ethylene tar was determined and the results were as follows. (1) During oxidation, the side chains of aromatic compounds first react with oxygen to form alcohols and aldehydes, leaving peroxy-radicals on aromatic rings. Subsequently, the aromatic compounds with peroxy-radicals undergo polymerization/condensation reactions to form larger molecules. (2) A fourth-order reaction model was used to describe the first 3 stages in the oxidation process, and the activation energies are 47.33, 18.69 and 9.00 kJ·mol−1 at 323–400, 400–605, 605–750 K, respectively. A three-dimensional diffusion model was applied to the fourth stage of the oxidation process, and the activation energy is 88.37 kJ·mol−1 at 750–860 K. A high softening point pitch was also produced for use as a coating of the graphite anode, and after it had been applied the capacity retention after 300 cycles increased from 51.54% to 79.07%.
-
Mechanisms Symbol $ f\left(\alpha \right) $ $ G\left(\alpha \right) $ Order of reaction First order $ {\mathrm{F}}_{1} $ $ 1-\alpha $ $ -\mathrm{l}\mathrm{n}\left(1-\alpha \right) $ Second order $ {\mathrm{F}}_{2} $ $ {\left(1-\alpha \right)}^{2} $ $ {\left(1-\alpha \right)}^{-1}-1 $ Third order $ {\mathrm{F}}_{3} $ $ {\left(1-\alpha \right)}^{3} $ $ \left[{\left(1-\alpha \right)}^{-2}-1\right]/2 $ Fourth order $ {\mathrm{F}}_{4} $ $ {\left(1-\alpha \right)}^{4} $ $ \left[{\left(1-\alpha \right)}^{-3}-1\right]/3 $ Diffusion One-way transport $ {\mathrm{D}}_{1} $ $ 0.5\alpha $ $ {\alpha }^{2} $ Two-way transport $ {\mathrm{D}}_{2} $ $ {\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{-1} $ $ \alpha +\left(1-\alpha \right)\mathrm{l}\mathrm{n}\left(1-\alpha \right) $ Three-way transport $ {\mathrm{D}}_{3} $ $ 1.5{\left(1-\alpha \right)}^{2/3}{\left[1-{\left(1-\alpha \right)}^{1/3}\right]}^{-1} $ $ {\left[1-{\left(1-\alpha \right)}^{1/3}\right]}^{2} $ Contracting geometry Contracting cylinder $ {\mathrm{R}}_{2} $ $ 2{\left(1-\alpha \right)}^{1/2} $ $ 1-{\left(1-\alpha \right)}^{1/2} $ Contracting sphere $ {\mathrm{R}}_{3} $ $ 3{\left(1-\alpha \right)}^{2/3} $ $ 1-{\left(1-\alpha \right)}^{1/3} $ Random nucleation and nuclei growth Avrami-Erofeev $ {\mathrm{A}}_{3/2} $ $ 1.5\left(1-\alpha \right){\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{1/3} $ $ {\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{3/2} $ Avrami-Erofeev $ {\mathrm{A}}_{2} $ $ 2\left(1-\alpha \right){\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{1/2} $ $ {\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{1/2} $ Avrami-Erofeev $ {\mathrm{A}}_{3} $ $ 3\left(1-\alpha \right){\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{2/3} $ $ {\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{1/3} $ Avrami-Erofeev $ {\mathrm{A}}_{4} $ $ 4\left(1-\alpha \right){\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{3/4} $ $ {\left[-\mathrm{l}\mathrm{n}\left(1-\alpha \right)\right]}^{1/4} $ Exponential nucleation Power law $ {\mathrm{P}}_{3/2} $ $ 2/3{\alpha }^{-1/2} $ $ {\alpha }^{2/3} $ Power law $ {\mathrm{P}}_{2} $ $ 2{\alpha }^{1/2} $ $ {\alpha }^{1/2} $ Power law $ {\mathrm{P}}_{3} $ $ 3{\alpha }^{2/3} $ $ {\alpha }^{1/3} $ Power law $ {\mathrm{P}}_{4} $ $ 4{\alpha }^{3/4} $ $ {\alpha }^{1/4} $ Table 2. The fundamental properties of ethylene tar and ET-HR
Elemental analysis/% 2C/H 3TS/% Four components 8SO/°C 9CV/% C H N S 1O 4S 5Ar 6R 7As ET 92.280 7.324 0 0.027 0.369 1.050 100 5.633 90.501 3.423 0.443 <25 7.740 ET-HR 92.820 6.869 0 0.022 0.289 1.130 100 0 91.911 7.162 0.927 <25 14.10 Note: 1SO: By difference. 2C/H: Atomic ratio. 3TS: Toluene soluble components. 4S: Saturated fraction. 5Ar: Aromatic fraction. 6R: Resin. 7As: Asphaltene. 8SP: Softening point. 9CV: Coking value. Table 3. Possible compounds distinguished by GC-MS in ET-HR
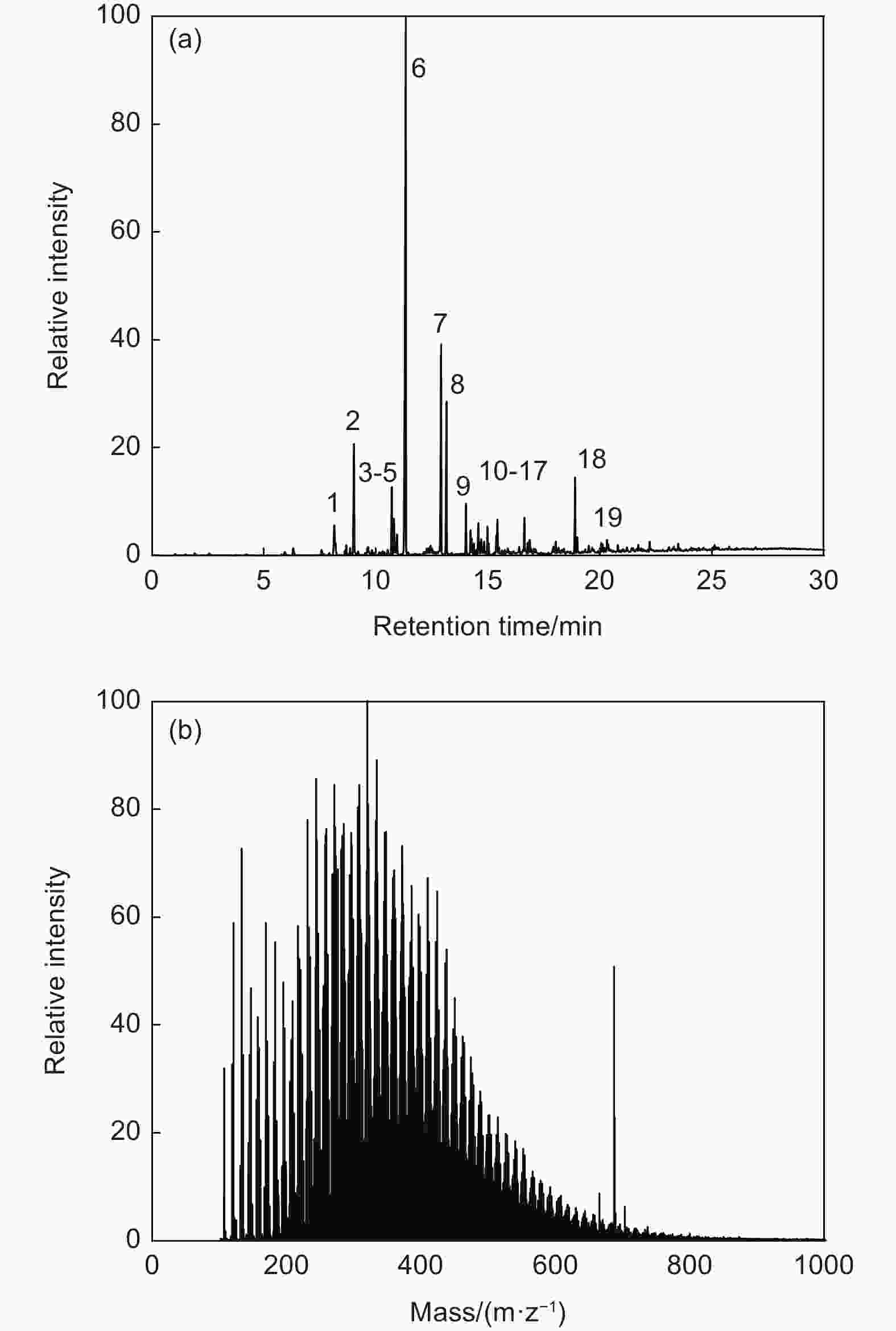
Peak No. Names or types Retention time/min Area percentage/% 1 (Z)-1-Phenylpropene 8.140 2.689 2 Indene 9.015 6.919 3 Naphthalene,1,2-dihydro- 10.708 3.874 4 Cycloprop[a]indene,1,1a,6,6a-tetrahydro- 10.798 3.095 5 Benzene, (1-methylene-2-propenyl)- 10.940 1.143 6 Naphthalene 11.332 42.252 7 Benzocycloheptatriene 12.909 12.971 8 Benzocycloheptatriene 13.154 8.688 9 Naphthalene,2-ethenyl- 14.023 2.430 10 Naphthalene,2-ethyl- 14.222 1.167 11 Naphthalene, 1-ethyl- 14.274 0.844 12 Naphthalene,1,6-dimethyl- 14.570 1.651 13 Naphthalene,2-ethenyl- 14.699 1.066 14 Biphenylene 14.982 1.395 15 1,1’-Biphenyl,4-methyl- 15.374 0.970 16 Naphthalene,2-ethenyl- 15.426 1.780 17 Fluorene 16.630 1.987 18 Phenanthrene 18.895 4.355 19 Anthracene 18.985 0.724 Table 4. Evolved gases analysis
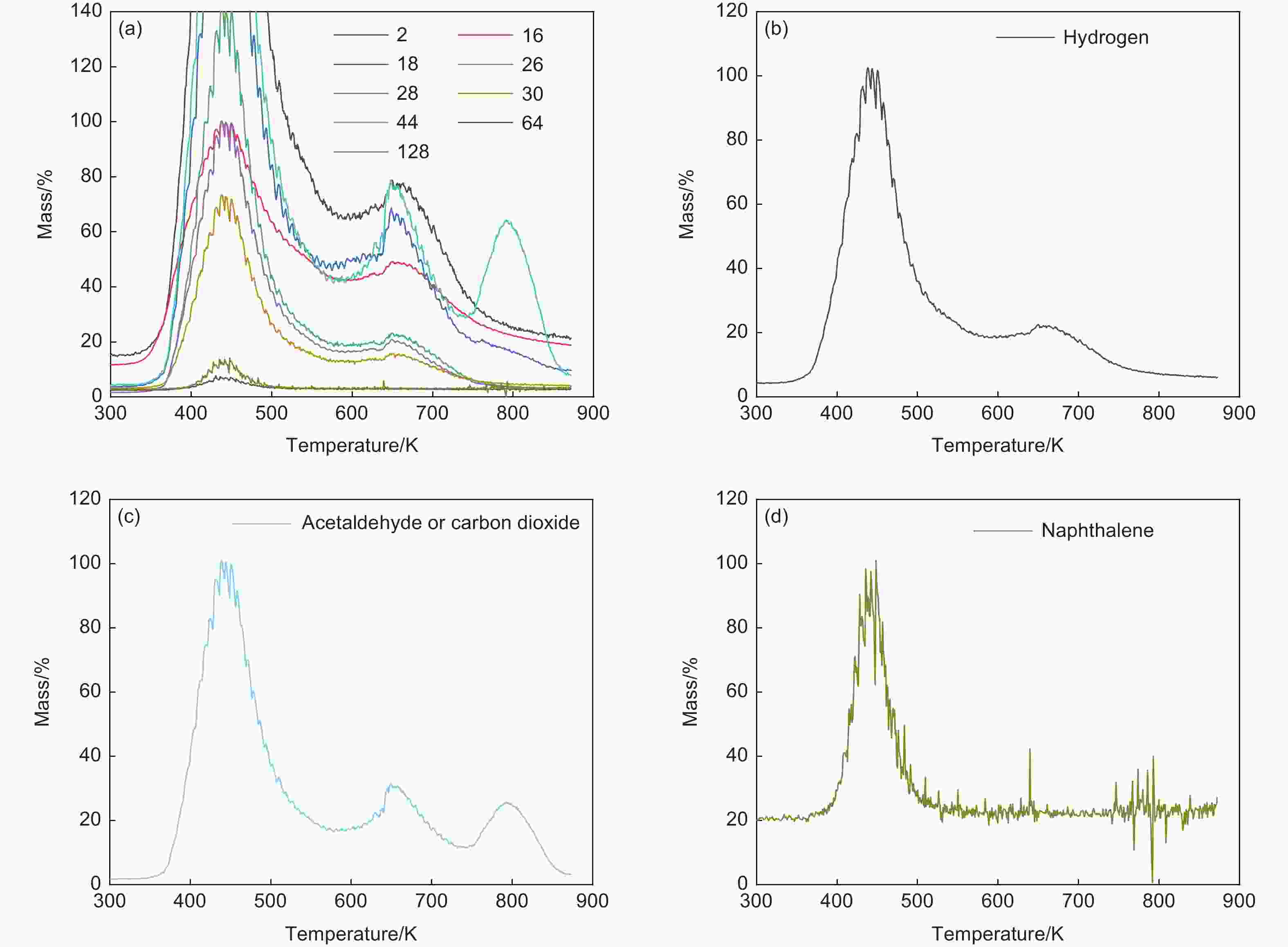
Fragment Molecular formula Molecule 2 H2 Hydrogen 16 CH4 Methane 18 H2O Water 26 C2H2 Acetylene 28 C2H4 Ethylene 30 HCHO Formaldehyde 44 CH3CHO/CO2 Acetaldehyde/carbon dioxide 64 SO2 Sulfur dioxide 128 C10H8 Naphthalene Table 5. Linear regression rates (R2) of different models of 4 parts in the process of oxidation
Model ${ {R}^{2} }_{{\rm{stage}}1}$(323-400 K) ${ {R}^{2} }_{{\rm{stage}}2}$(400-605 K) ${ {R}^{2} }_{{\rm{stage}}3}$(605-750 K) ${ {R}^{2} }_{{\rm{stage}}4}$(750-860 K) $ {\mathrm{F}}_{1} $ 0.99195 0.92258 NA 0.96740 $ {\mathrm{F}}_{2} $ 0.99372 0.97629 NA 0.91237 $ {\mathrm{F}}_{3} $ 0.99525 0.98949 0.77979 0.89775 $ {\mathrm{F}}_{4} $ 0.99653 0.99102 0.92887 0.89578 $ {\mathrm{D}}_{1} $ 0.99152 0.9389 NA 0.94713 $ {\mathrm{D}}_{2} $ 0.99211 0.95419 NA 0.97933 $ {\mathrm{D}}_{3} $ 0.99268 0.9667 NA 0.98793 $ {\mathrm{R}}_{2} $ 0.99097 0.85181 NA 0.98390 $ {\mathrm{R}}_{3} $ 0.99131 0.88105 NA 0.98385 $ {\mathrm{A}}_{3/2} $ 0.99283 0.96561 NA 0.97099 $ {\mathrm{A}}_{2} $ 0.98835 NA NA 0.95101 $ {\mathrm{A}}_{3} $ 0.98236 NA NA 0.91699 $ {\mathrm{A}}_{4} $ 0.97154 NA NA 0.82846 $ {\mathrm{P}}_{3/2} $ 0.99103 0.90477 NA 0.92539 $ {\mathrm{P}}_{2} $ 0.99103 0.90477 NA 0.92539 $ {\mathrm{P}}_{3} $ 0.97785 NA NA NA $ {\mathrm{P}}_{4} $ 0.96399 NA NA NA Table 6. Oxidation kinetics parameters of ET-HR
Reaction section Model $ k $ $ b $ ${E}_{ {\rm{a} } }({\rm{kJ} } \cdot { {\rm{mol} } }^{-1})$ $A({ {\rm{min} } }^{-1})$ $ {R}^{2} $ ET-HRpart 1 $ {\mathrm{F}}_{4} $ −5692.77928 0.84939 47329.76693 66554.85476 0.99653 ET-HRpart 2 $ {\mathrm{F}}_{4} $ −2247.94165 −7.74028 18689.38688 4.888708542 0.99102 ET-HRpart 3 $ {\mathrm{F}}_{4} $ −1083.02397 −9.46354 9004.261287 0.420382735 0.92887 ET-HRpart 4 $ {\mathrm{D}}_{3} $ −10628.96641 −1.51215 88369.22673 11715.00931 0.98793 -
[1] Kamran U, Heo Y J, Lee J W, et al. Functionalized carbon materials for electronic devices: A review[J]. Micromachines,2019,10(4):234. doi: 10.3390/mi10040234 [2] Liu D, Kong Q Q, Jia H, et al. Dual-functional 3D multi-wall carbon nanotubes/graphene/silicone rubber elastomer: Thermal management and electromagnetic interference shielding[J]. Carbon,2021,138(1):216-224. [3] Wu M G, Liao J Q, Yu L X, et al. 2020 Roadmap on carbon materials for energy storage and conversion[J]. Chemistry-An Asian Journal,2020,15(7):995-1013. doi: 10.1002/asia.201901802 [4] Faisal M, Pamungkas A Z, Krisnandi Y K. Study of amine functionalized mesoporous carbon as CO2 storage materials[J]. Processes,2021,9(456):456. [5] Newman J W. Petroleum pitch-a consistent carbon precursor[J]. Carbon,1973,11(6):674-674. [6] Jo Y J, Lee J D. Effect of petroleum pitch coating on electrochemical performance of graphite as anode materials[J]. The Korean Journal of Chemical Engineering,2019,36(10):1724-1731. doi: 10.1007/s11814-019-0354-3 [7] Mora E, Blanco C, Granda M, et al. A novel method to obtain a petroleum-derived mesophase pitch suitable as carbon fibre precursor[J]. Carbon,2003,41(3):445-452. doi: 10.1016/S0008-6223(02)00354-8 [8] Paolo, Davini. Desulphurization properties of active carbons obtained from petroleum pitch pyrolysis[J]. Carbon,1999,37(9):1363-1371. doi: 10.1016/S0008-6223(98)00331-5 [9] Mochida I, Sone Y, Korai Y. Reflux modification of ethylene tar pitch into mesophase pitch precursor using less aluminum chloride[J]. Journal of Materials Science Letters,1985,4(10):1237-1240. doi: 10.1007/BF00723468 [10] Shi K, Yang J X, Ye C, et al. A comparison of ethylene-tar-derived isotropic pitches prepared by air blowing and nitrogen distillation methods and their carbon fibers[J]. Materials,2019,12(2):305. doi: 10.3390/ma12020305 [11] Shu M Z, Maeda T, Mondori J, et al. Preparation of isotropic pitch precursors for general purpose carbon fibers (GPCF) by air blowing-III. Air blowing of isotropic naphthalene and hydrogenated coal tar pitches with addition of 1, 8-dinitronaphthalene[J]. Carbon,1993,31(3):421-426. doi: 10.1016/0008-6223(93)90129-X [12] Shu M Z, Maeda T, Tokumitsu K, et al. Preparation of isotropic pitch precursors for general purpose carbon fibers (GPCF) by air blowing-II. Air blowing of coal tar, hydrogenated coal tar, and petroleum pitches[J]. Carbon,1993,31(3):413-419. doi: 10.1016/0008-6223(93)90128-W [13] Fernández J, Figueiras A, Granda M, et al. Modification of coal-tar pitch by air-blowing-I. Variation of pitch composition and properties[J]. Carbon,1995,33(3):295-307. doi: 10.1016/0008-6223(94)00130-R [14] Prada V, Granda M, Bermejo J, et al. Preparation of novel pitches by tar air-blowing [J]. Carbon: An International Journal Sponsored by the American Carbon Society. 1999, 37(1): 97-106. [15] Maeda T, Shu M Z, Tokumitsu K, et al. Preparation of isotropic pitch precursors for general purpose carbon fibers (GPCF) by air blowing-I. Preparation of spinnable isotropic pitch precursor from coal tar by air blowing[J]. Carbon,1993,31(3):407-412. doi: 10.1016/0008-6223(93)90127-V [16] Alcañiz-Monge J, Cazorla-Amorós D, Linares-Solano A, et al. Preparation of general purpose carbon fibers from coal tar pitches with low softening point[J]. Carbon: An International Journal Sponsored by the American Carbon Society,1997,35(8):1079-1087. [17] Dunham M G, Edie D. Model of stabilization for pan-based carbon fiber precursor bundles[J]. Carbon,1992,30(3):435-450. doi: 10.1016/0008-6223(92)90042-U [18] Miura K, Nakagawa, H, Hashimoto K, et al. Examination of the oxidative stabilization reaction of the pitch-based carbon fiber through continuous measurement of oxygen chemisorption and gas formation rate[J]. Carbon,1995,33(3):275-282. doi: 10.1016/0008-6223(94)00133-K [19] Barr J B, Lewis I C. Chemical changes during the mild air oxidation of pitch[J]. Carbon,1978,16(6):439-444. doi: 10.1016/0008-6223(78)90090-8 [20] SugioŌtani. Mechanism of the carbonization of MP carbon fiber at the low temperature range[J]. Carbon,1967,5(3):219-220. doi: 10.1016/0008-6223(67)90003-6 [21] Metzinger T, Hüttinger K J. Investigations on the crosslinking of binder pitch matrix of carbon bodies with molecular oxygen-part I. Chemistry of reactions between pitch and oxygen[J]. Carbon,1997,35(7):885-892. doi: 10.1016/S0008-6223(97)00038-9 [22] Lewis I C, Singer L S. Electron spin resonance study of the reaction of aromatic hydrocarbons with oxygen[J]. The Journal of Physical Chemistry,1981,85(4):354-360. doi: 10.1021/j150604a011 [23] Matsumoto T, Mochida I. A structural study on oxidative stabilization of mesophase pitch fibers derived from coal tar[J]. Carbon,1992,30(7):1041-1046. doi: 10.1016/0008-6223(92)90134-I [24] Yang C Q, Simms J R. Infrared spectroscopy studies of the petroleum pitch carbon fiber-I: The raw materials, the stabilization, and carbonization processes[J]. Carbon,1993,31(3):451-459. doi: 10.1016/0008-6223(93)90133-U [25] Mochida I, Inaba T, Korai Y, et al. Carbonization properties of carbonaceous substances oxidized by air blowing-I: Carbonization behaviors and chemical structure of residual oils oxidized by air blowing[J]. Carbon,1983,21(6):543-552. doi: 10.1016/0008-6223(83)90237-3 [26] Fernández A L, Granda M, Bermejo J, et al. Air-blowing of anthracene oil for carbon precursors[J]. Carbon,2000,38(9):1315-1322. doi: 10.1016/S0008-6223(99)00264-X [27] Lavin J G. Chemical reactions in the stabilization of mesophase pitch-based carbon fiber[J]. Carbon,1992,30(3):351-357. doi: 10.1016/0008-6223(92)90030-Z [28] Yamaguchi C, Mondori J, Matsumoto A, et al. Air-blowing reactions of pitch: I. Oxidation of aromatic hydrocarbons[J]. Carbon,1995,33(2):193-201. doi: 10.1016/0008-6223(94)00127-L [29] Ge C Z, Sun Z L, Yang H X, et al. Preparation and characterization of high softening point and homogeneous isotropic pitches produced from distilled ethylene tar by a novel bromination method[J]. New Carbon Materials,2018,33(1):71-81. doi: 10.1016/S1872-5805(18)60327-0 [30] Yang P Y, He X C, Zhang W J, et al. Study on thermal degradation of cattlehide collagen fibers by simultaneous TG–MS–FTIR[J]. Journal of Thermal Analysis & Calorimetry,2017,127(3):1-8. [31] Aboulkas A, Harfi K E, Bouadili A E. Kinetic and mechanism of Tarfaya (Morocco) oil shale and LDPE mixture pyrolysis[J]. Journal of Materials Processing Technology,2008,206(1-3):16-24. doi: 10.1016/j.jmatprotec.2007.11.282 [32] Gai C, Zhang Y, Chen W T, et al. Thermogravimetric and kinetic analysis of thermal decomposition characteristics of low-lipid microalgae[J]. Bioresource Technology,2013,150:139-148. [33] Yi C, Jia D, Luo Y H. Investigation of agricultural residues pyrolysis behavior under inert and oxidative conditions[J]. Journal of Analytical & Applied Pyrolysis,2008,83(2):165-174. [34] White J E, Catallo W J, Legendre B L. Biomass pyrolysis kinetics: A comparative critical review with relevant agricultural residue case studies[J]. Journal of Analytical and Applied Pyrolysis,2011,91(1):1-33. doi: 10.1016/j.jaap.2011.01.004 [35] Wang X N, Ma Y H, Niu R X, et al. Pyrolysis behavior and kinetic of coal tar pitch modified with paraformaldehyde[J]. Waste and Biomass Valorization,2017,8(1):209-216. doi: 10.1007/s12649-016-9568-3 [36] Duan S Z, Wu X W, Wang Y F, et al. Recent progress in the research and development of natural graphite for use in thermal management, battery electrodes and the nuclear industry[J]. New Carbon Materials,2023,38(1):73-95. doi: 10.1016/S1872-5805(23)60717-6 [37] Kim B H, Kim J H, Kim J G, et al. Electrochemical and structural properties of lithium battery anode materials by using a molecular weight controlled pitch derived from petroleum residue[J]. Journal of industrial and engineering chemistry,2016,41:1-9. [38] Zhan C Z, Zeng X J, LV R T, et al. Preparation of porous graphitic carbon and its dual-ion capacitance energy storage mechanism[J]. New Carbon Materials,2023,38(3):576-582. doi: 10.1016/S1872-5805(23)60727-9 -





 下载:
下载: