-
摘要: 随着近年来电动汽车的蓬勃发展,锂离子电池的使用量以及退役量都在逐年增长,随之而来的是废旧锂离子电池带来的环境污染以及资源浪费问题。目前商用锂离子电池多由过渡金属氧化物或磷酸盐基正极、石墨基负极、含有害锂盐的有机电解质、聚合物隔膜以及塑料或金属外壳组成,在电池退役后,其中的诸多贵金属以及石墨等都具有较高的回收价值。本文对锂离子电池工作原理及组成结构、废旧锂离子电池全组分回收等研究现状进行了综述,着重介绍废旧锂离子电池中正极材料、负极材料以及电解液回收的研究进展,从回收成本和二次污染等方面概述了不同方法所遇到的问题,最后对未来的发展提供了一些思路。Abstract: With the recent rapid development of electric vehicles, the use and decommissioning of Li-ion batteries have increased, causing environmental pollution and the waste of valuable materials in spent batteries. Commercial Li-ion batteries are mostly composed of transition metal oxide or phosphate-based cathodes, graphite-based anodes, organic electrolytes containing harmful lithium salts, polymer separators, and plastic or metal shells. After the battery is retired, many precious metals and graphite have a high recycling value. We review the current status of research on recovering these components with an emphasis on the leaching and separation of cathode and anode materials, and electrolytes in these batteries. The problems encountered in the different methods are outlined in terms of recycling cost and secondary pollution. Future research trends are outlined for the commercial full recovery of spent Li-ion batteries.
-
Key words:
- Spent Li-ion battery /
- Graphite /
- Full composition /
- Recycling
-
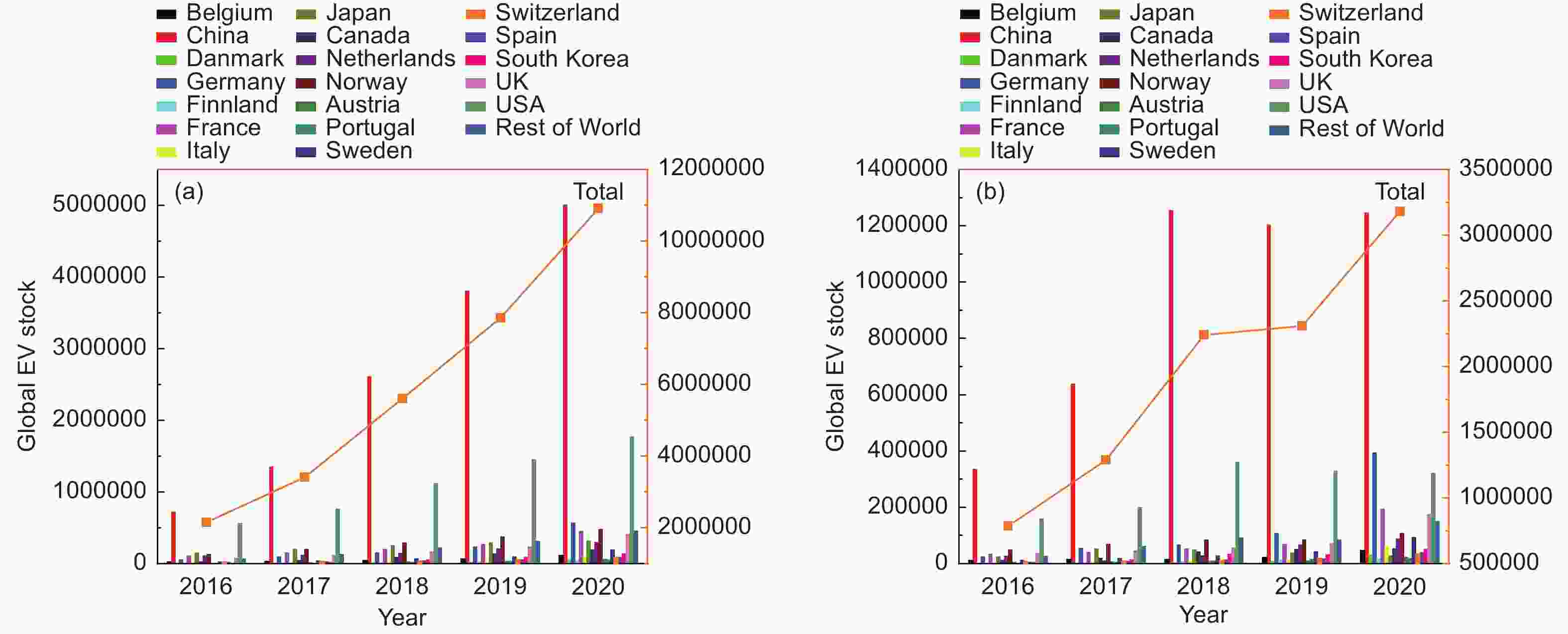
Figure 1. (a) Inventory of electric vehicles in various countries in recent years and (b) registered number of electric vehicles in various countries in recent years[7].
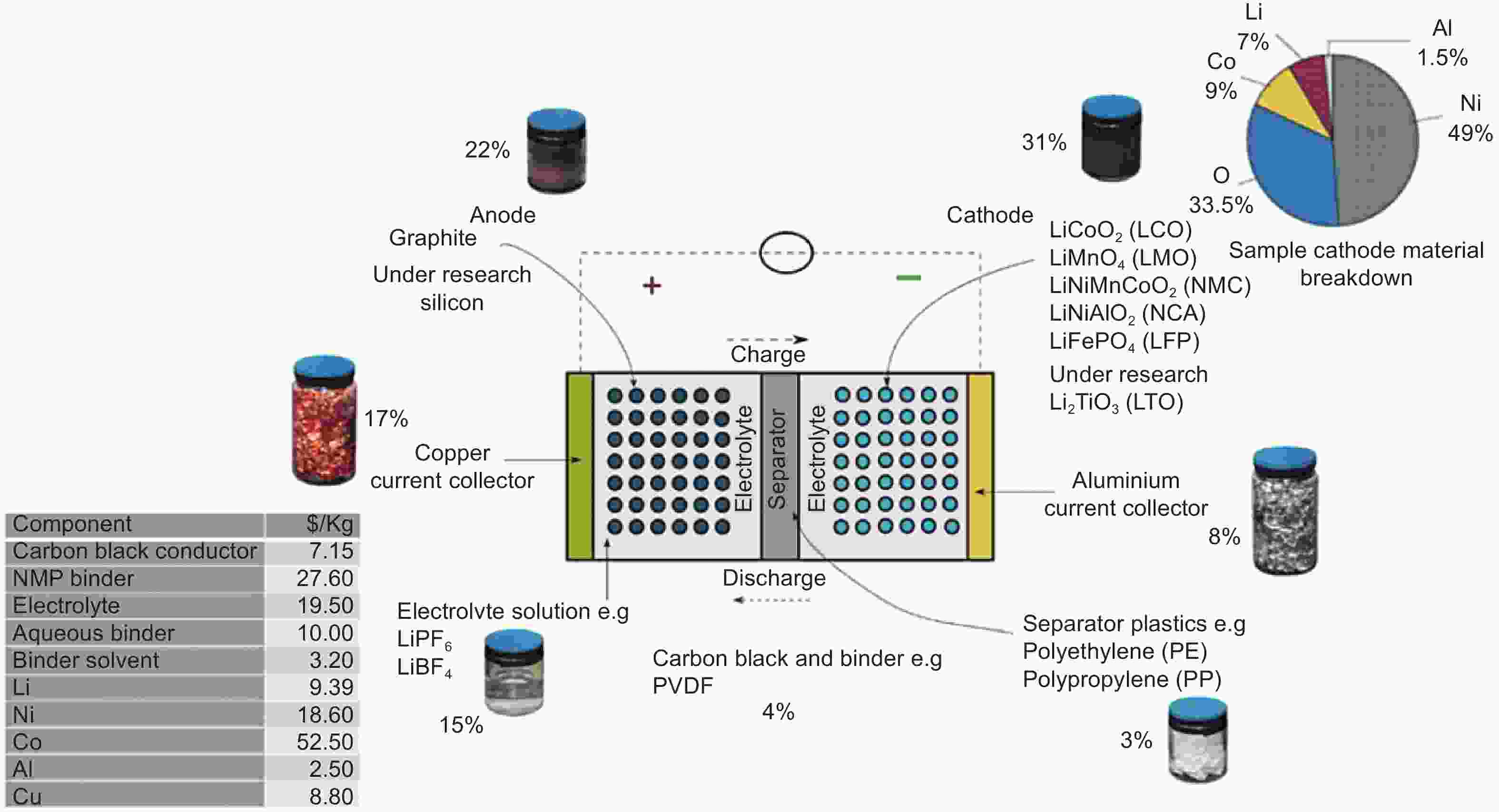
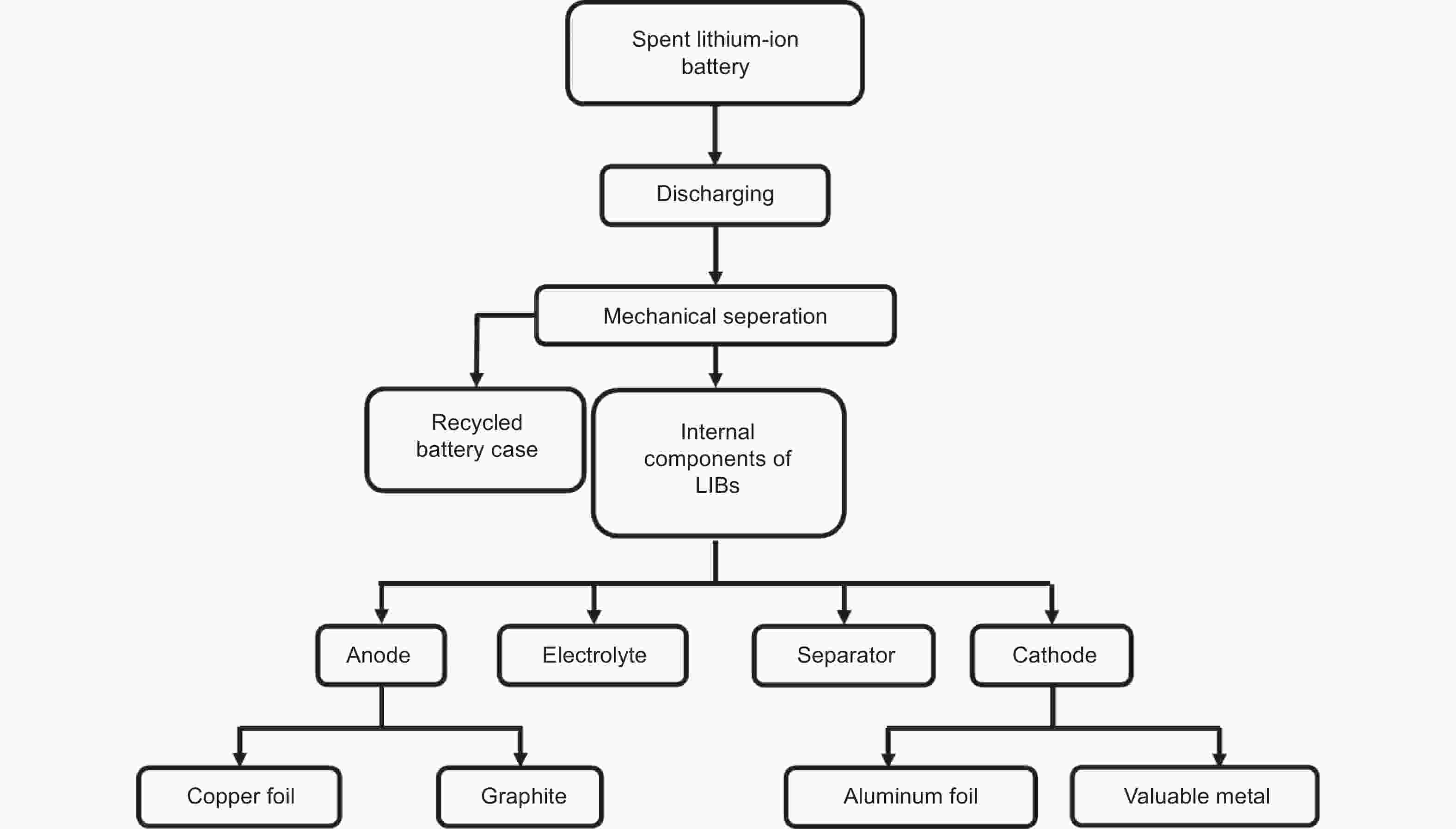
Figure 2. The components of LIBs and the weight distribution of each part[16] (Reprinted with permission).
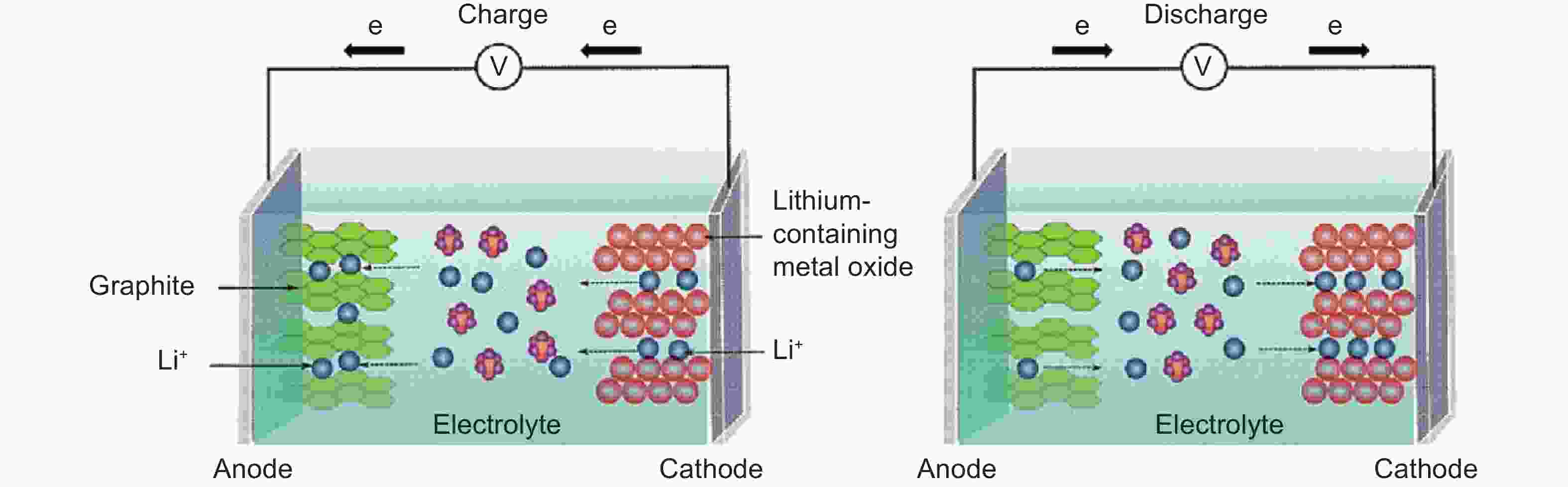
Figure 3. Charging and discharging working principle of LIBs[18] (Reprinted with permission).
Figure 6. (a) Working principle of the Falcon centrifugal separator[39] (Reprinted with permission). (b) TG-DSC curve of cathode active powder baked at different temperatures[51] (Reprinted with permission). (c) Process of separating positive electrode material and aluminum foil with deep eutectic solvent[56] (Reprinted with permission).
Figure 7. (a) Schematic diagram of the leaching mechanism of reducing agent LiFePO4[75] (Reprinted with permission). (b) Comparison of the concentration of inorganic acid and organic acid on the leaching rate of spent LIBs[78] (Reprinted with permission). (c) The influence of the oxalic acid concentration on the reaction efficiency of LiCoO2[49] (Reprinted with permission).
Figure 8. (a) Comparison of H2SO4 produced by pH adjustment and without measuring pH change in MS-MC biological leaching system[101] (Reprinted with permission). (b) Metal leaching rates of various strains[102]. (c) Glucosidase oxidation process (Reprinted with permission). (d) Final product leaching efficiencies of precious metals by gluconic acid[103] (Reprinted with permission).
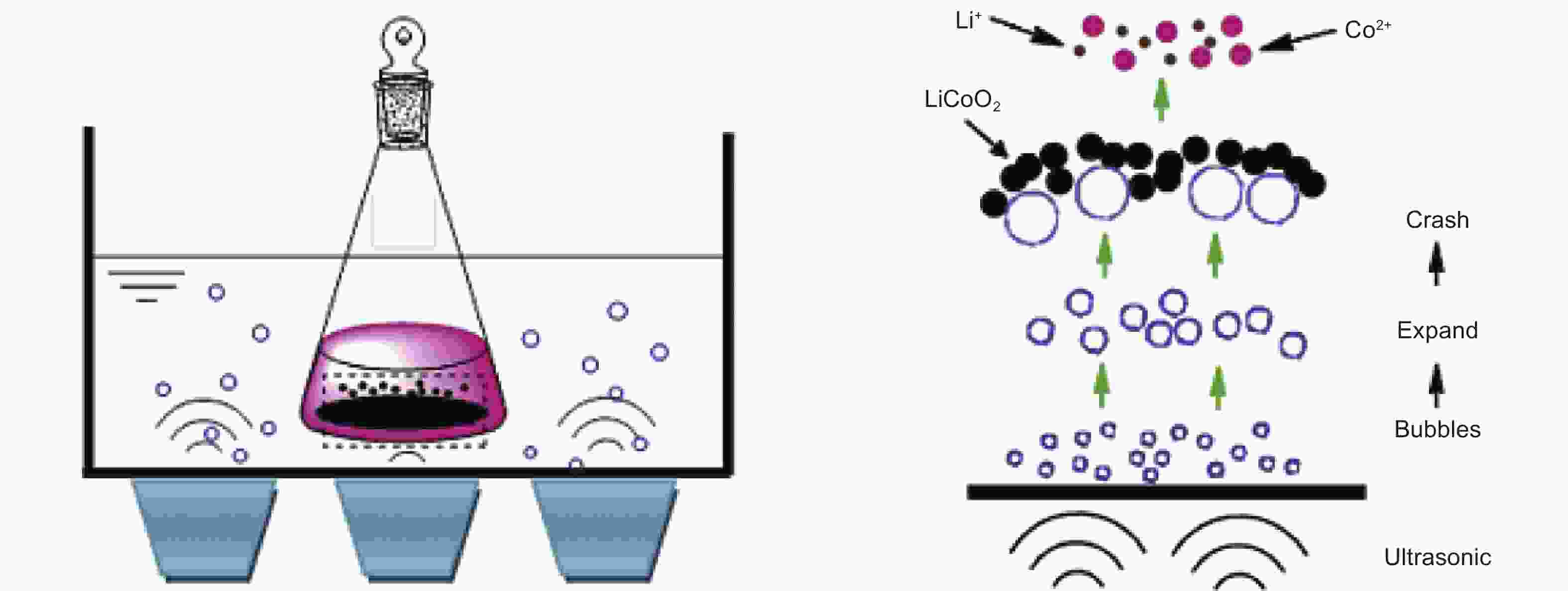
Figure 9. The mechanism of ultrasonic cavitation on the leaching process[53] (Reprinted with permission).
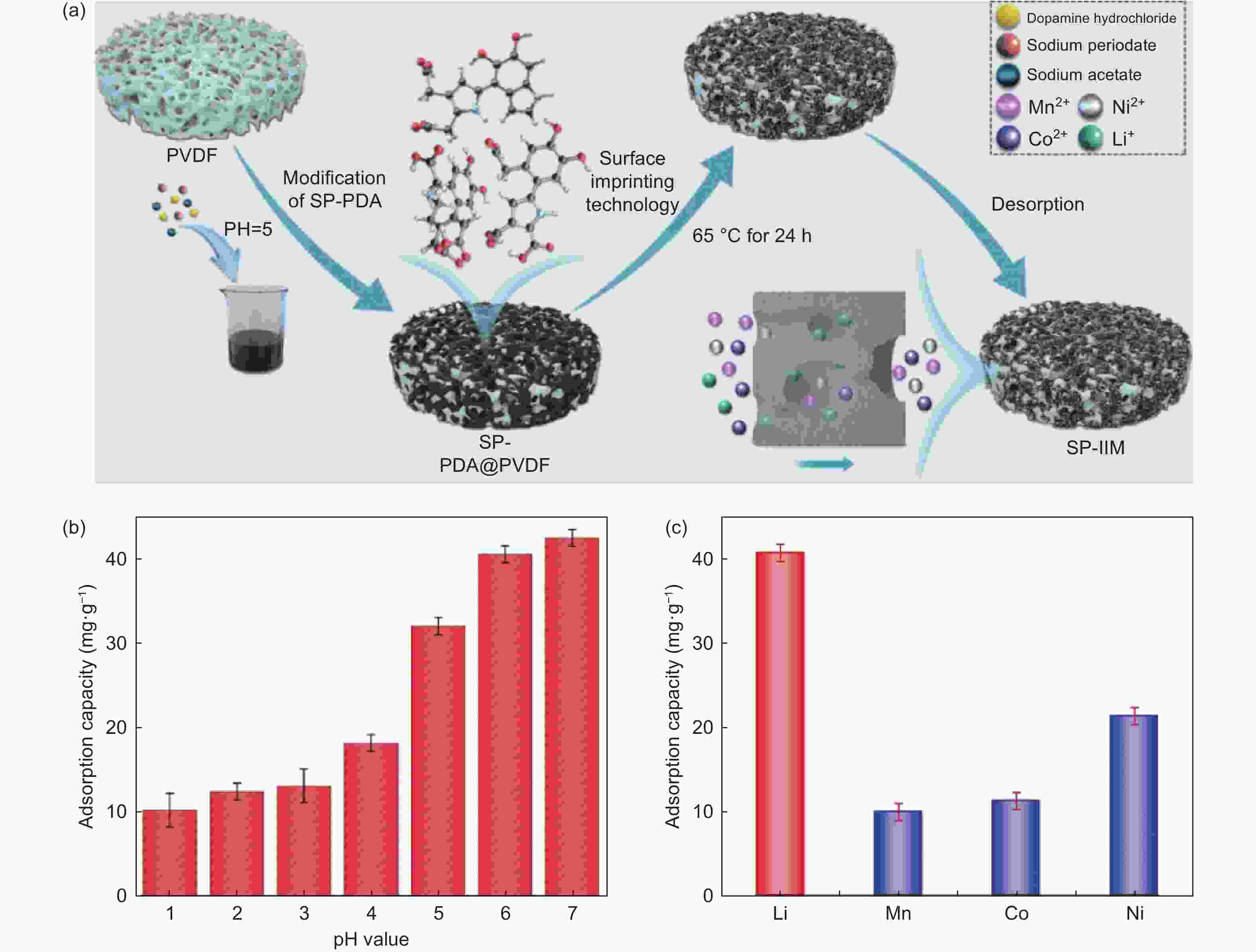
Figure 10. (a) Preparation process of ion imprinted membrane. (b) Adsorption capacity of ion imprinted membrane for lithium ions at different pH values. (c) Selective adsorption of Li+ by the imprinted membrane[125] (Reprinted with permission).
Table 1. Usage of various electric vehicle cathode and anode materials.
Vehicle Battery supplier Cathode Anode Renault Fluence Automotive Energy (Nissan NEC JV) LMO C Nissian Leaf EV Automotive Energy (Nissan NEC JV) LMO C Chevrolet Volt Compact Power (subsidiary of LG Chem) LMO C BYD E6 BYD LFP C Tesla Model S Panasonic Energy Nickel-type C Tesla Roadster Panasonic Energy NCA C Subaru G4e Subaru LVP C Honda Fit EV Toshiba Corporation NCM LTO Table 2. Harmful components in the main components of LIBs and their hazards.
Component Harmful ingredient Main hazard Cathode material Heavy metals such as Co and Ni Easily causing dermatitis, respiratory disorder, lung disease,
and gastrointestinal damage; carcinogenicAnode material Carbon materials such as graphite and acetylene black The combustion of the materials produces CO, CO2,
and other gases and solid dust to pollute the air.Lithium salt in electrolyte LiBF4, LiAsF6, LiPF6, etc. Strongly corrosive; it will decompose in water to produce highly
toxic HF, and produce P2O3 and other toxic substances during
combustion, which will pollute the environment.Electrolyte solvent EC, DMC, PC, DMSO, etc. The combustion of the materials produces CO, CO2, and other gases,
and aldehydes, ketones, and other organic pollutants.Separator Polypropylene (PP), polyethylene (PE) Naturally difficult to degrade, causing organic pollution Table 3. Summary of the research status of leaching spent LIB cathode materials with acids as the leaching agents.
Materials Leaching agent Leaching condition Reduction agent Solid-liquid ratio(g/L) Leaching rate Ref. LiCoO2 2 M H2SO4 80 °C + 1.5 h 0.11 M ascorbic acid 200 Li 95.7%, Co 93.8% [73] LiCoO2 2 M H2SO4 60 °C + 2 h 2% H2O2 (v/v) 33 Li 87.5%, Co 96.3% [82] LiCoO2 4 M H2SO4 85 °C + 2 h 10% H2O2 (v/v) 0.1 Li 96%, Co 95% [83] LNCM 2 M H2SO4 60 °C + 1 h 3% H2O2 (v/v) 50 Li: 99%; Co: 99%; Ni: 99%; Mn:99% [84] LNCM 1.5 M H2SO4 25 °C + 1 h 8% H2O2 (v/v) 30 Li: 80%; Co: 93%; Ni: 92%; Mn:90% [85] LNCM 0.5 M HNO3 70 °C +0.15 h 0.5 M ascorbic acid 20 Li, Co, Ni, and Mn~100% [86] LiFePO4 2.5 M H2SO4 60 °C +4 h N/A 100 Li: 97%; Fe: 98% [87] LiCoO2 3 M HCl 80 °C +40 min 3.5% H2O2 (v/v) 0.05 Li: 81%; Co: 79% [88] LiCoO2 0.7 M H3PO4 40 °C +1 h 4% H2O2 (v/v) 50 Co: 99.7%; Li: 99.9% [89] LiCoO2 1 M oxalic acid 80 °C +2 h N/A 50 Co, Li: >98% [49] LiCoO2 1.25 M ascorbic acid 70 °C +20 min N/A 25 Co: 94.8%; Li: 98.5% [90] LiCoO2 1.25 M citric acid 90 °C +0.5 h 1% H2O2 (v/v) 16.7 Co: 90%; Li: 98% [91] LiCoO2 1.5 M malic acid 90 °C +40 min 2% H2O2 (v/v) 20 Co: >90%; Li: ~100% [92] LiCoO2 0.5 M naphthalene disulfonic acid 60 °C +0.5 h 3% H2O2 (v/v) 25 Co: 97%; Li: 99% [93] LNCM 1.2 M DL-malic acid 90 °C +0.5 h 1.5% H2O2 (v/v) 40 Li: 98.9%; Co: 94.3%;
Ni: 95.1%; Mn: 96.4%[94] LNCM 2 M L-Tartaric acid 70 °C +0.5 h 4% H2O2 (v/v) 17 Li: 99%; Co: 99%;
Ni: 99%; Mn: 99%[40] LNCM 0.2 M H3PO4+0.4 M C6H8O7 90 °C +0.5 h N/A 20 Li: 100%; Co: 91.63%;
Ni: 93.38%; Mn: 92%[81] Note: M: mol L−1 -
[1] Yoshino A. The birth of the lithium-ion battery[J]. Angewandte Chemie International Edition,2012,51(24):5798-5800. doi: 10.1002/anie.201105006 [2] Bernardes A M, Espinosa D C R, Tenório J A S. Recycling of batteries: A review of current processes and technologies[J]. Journal of Power Sources,2004,130(1-2):291-298. doi: 10.1016/j.jpowsour.2003.12.026 [3] Joo S H, Shin D, Oh C, et al. Extraction of manganese by alkyl monocarboxylic acid in a mixed extractant from a leaching solution of spent lithium-ion battery ternary cathodic material[J]. Journal of Power Sources,2016,305:175-181. doi: 10.1016/j.jpowsour.2015.11.039 [4] Zhang K, Han X P, Hu Z, et al. Nanostructured Mn-based oxides for electrochemical energy storage and conversion[J]. Chemical Society Reviews,2015,44(3):699-728. doi: 10.1039/C4CS00218K [5] Granata G, Moscardini E, Pagnanelli F, et al. Product recovery from Li-ion battery wastes coming from an industrial pre-treatment plant: Lab scale tests and process simulations[J]. Journal of Power Sources,2012,206:393-401. doi: 10.1016/j.jpowsour.2012.01.115 [6] Li L, Zhang X X, Li M, et al. The recycling of spent lithium-ion batteries: A review of current processes and technologies[J]. Electrochemical Energy Reviews,2018,1(4):461-482. doi: 10.1007/s41918-018-0012-1 [7] ZSW data on stock as well as on new registrations of battery-electric vehicles [EB/OL]. [2020-12]. https://www.zsw-bw.de/en/media-center/data-service.html#c6700. [8] Bloomberg N. Electric vehicle outlook [R]. US: BloombergNEF, 2019. [9] Chu S, Cui Y, Liu N. The path towards sustainable energy[J]. Nature Materials,2016,16(1):16-22. [10] Harper G, Sommerville R, Kendrick E, et al. Recycling lithium-ion batteries from electric vehicles[J]. Nature,2019,575(7781):75-86. doi: 10.1038/s41586-019-1682-5 [11] Huang X Z. Research on short-process recycling and regeneration of ternary cathode materials for waste lithium-ion power batteries [D]. Beijing: Beijing General Research Institute of Nonferrous Metals, 2019. [12] Lv W G, Wang Z H, Cao H B, et al. A critical review and analysis on the recycling of spent lithium-ion batteries[J]. Acs Sustainable Chemistry & Engineering,2018,6(2):1504-1521. [13] Mrozik W, Rajaeifar M A, Heidrich O, et al. Environmental impacts, pollution sources and pathways of spent lithium-ion batteries[J]. Energy & Environmental Science,2021,14(12):6099-6121. [14] Winslow K M, Laux S J, Townsend T G. A review on the growing concern and potential management strategies of waste lithium-ion batteries[J]. Resources, Conservation and Recycling,2018,129:263-277. doi: 10.1016/j.resconrec.2017.11.001 [15] Roy J J, Rarotra S, Krikstolaityte V, et al. Green recycling methods to treat lithium-ion batteries e-waste: A circular approach to sustainability[J]. Advanced Materials,2021:2103346. doi: 10.1002/adma.202103346 [16] Wang M, Tang Y B. A review on the features and progress of dual‐ion batteries[J]. Advanced Energy Materials,2018,8(19):1703320. doi: 10.1002/aenm.201703320 [17] Whittingham M S. Lithium batteries and cathode materials[J]. Chemical Reviews,2004,35(50):4271-4301. [18] Yu D W, Huang Z, Makuza B, et al. Pretreatment options for the recycling of spent lithium-ion batteries: A comprehensive review[J]. Minerals Engineering,2021:173. [19] Chagnes A, Pospiech B. A brief review on hydrometallurgical technologies for recycling spent lithium-ion batteries[J]. Journal of Chemical Technology & Biotechnology,2013,88(7):1191-1199. [20] Lu L G, Han X B, Li J Q, et al. A review on the key issues for lithium-ion battery management in electric vehicles[J]. Journal of Power Sources,2013,226:272-288. doi: 10.1016/j.jpowsour.2012.10.060 [21] Lu M, Yin G P, Shi P F. Modifications of graphite material for negative electrode of Li-ion batteries [J]. Dianchi/Battery, 2001, 31(4). [22] Xu Z L, Liu X, Luo Y, et al. Nanosilicon anodes for high performance rechargeable batteries[J]. Progress in Materials Science,2017,90:1-44. doi: 10.1016/j.pmatsci.2017.07.003 [23] Ouardi K E, Dahbi M, Hakim C, et al. Facile synthesis of nanoparticles titanium oxide as high-capacity and high-capability electrode for lithium-ion batteries[J]. Journal of Applied Electrochemistry,2020,50(1):583-595. doi: 10.1007/s10800-020-01419-y [24] Park J, Margez C L, Greszler T A. Effect of particle size and electronic percolation on low-temperature performance in lithium titanate-based batteries[J]. ACS Omega,2019,4(25):21048-21053. doi: 10.1021/acsomega.9b02393 [25] Ali H, Khan H A, Pecht M G. Circular economy of Li batteries: Technologies and trends[J]. Journal of Energy Storage,2021, 40:102690. [26] Ordonez J, Gago E J, Girard A. Processes and technologies for the recycling and recovery of spent lithium-ion batteries[J]. Renewable & Sustainable Energy Reviews,2016,60:195-205. [27] Al-Fakih A, Mohammed B S, Liew M S, et al. Incorporation of waste materials in the manufacture of masonry bricks: An update review[J]. Journal of Building Engineering,2019,21:37-54. doi: 10.1016/j.jobe.2018.09.023 [28] Carolin C F, Kumar P S, Saravanan A, et al. Efficient techniques for the removal of toxic heavy metals from aquatic environment: A review[J]. Journal of Environmental Chemical Engineering,2017,5(3):2782-2799. doi: 10.1016/j.jece.2017.05.029 [29] Chowdhury S, Mazumder M A J, Al-Attas O, et al. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries[J]. Science of the Total Environment,2016,569:476-488. [30] et al. A future perspective on lithium-ion battery waste flows from electric vehicles[J]. Resources, Conservation and Recycling,2014,83:63-76. [31] USGS. Mineral commodity summaries [R]. Reston, VA: United States Geological Survey, 2019. [32] Olivetti E A, Ceder G, Gaustad G G, et al. Lithium-ion battery supply chain considerations: Analysis of potential bottlenecks in critical metals[J]. Joule,2017,1(2):229-243. doi: 10.1016/j.joule.2017.08.019 [33] Liu W Q, Liu W, Li X X, et al. Dynamic material flow analysis of critical metals for lithium-ion battery system in China from 2000-2018[J]. Resources Conservation and Recycling,2021:164. [34] Sun X, Hao H, Zhao F Q, et al. Tracing global lithium flow: A trade-linked material flow analysis[J]. Resources Conservation and Recycling,2017,124:50-61. doi: 10.1016/j.resconrec.2017.04.012 [35] Li L, Bian Y F, Zhang X X, et al. Economical recycling process for spent lithium-ion batteries and macro-and micro-scale mechanistic study[J]. Journal of Power Sources,2018,377:70-79. doi: 10.1016/j.jpowsour.2017.12.006 [36] Ku H, Jung Y, Jo M, et al. Recycling of spent lithium-ion battery cathode materials by ammoniacal leaching[J]. Journal of Hazardous Materials,2016,313:138-146. doi: 10.1016/j.jhazmat.2016.03.062 [37] Hageluken C. Recycling of electronic scrap at Umicore's integrated metals smelter and refinery[J]. World of Metallurgy- Erzmetall,2006,59(3):152-161. [38] Cardarelli F, Dube J. A method for recycling spent lithium metal polymer rechargeable batteries and related materials: CA, WO2002023651 A1[P]. 2012. [39] Zhang Y, He Y Q, Zhang T, et al. Application of falcon centrifuge in the recycling of electrode materials from spent lithium ion batteries[J]. Journal of Cleaner Production,2018,202:736-747. doi: 10.1016/j.jclepro.2018.08.133 [40] He L P, Sun S Y, Mu Y Y, et al. Recovery of lithium, nickel, cobalt, and manganese from spent lithium-ion batteries using L-tartaric acid as a leachant[J]. Acs Sustainable Chemistry & Engineering,2017,5(1):714-721. [41] Li J, Wang G X, Xu Z M. Generation and detection of metal ions and volatile organic compounds (VOCs) emissions from the pretreatment processes for recycling spent lithium-ion batteries[J]. Waste Management,2016,52:221-227. doi: 10.1016/j.wasman.2016.03.011 [42] Nie H, Xu L, Song D, et al. LiCoO2: Recycling from spent batteries and regeneration with solid state synthesis[J]. Green Chemistry,2015,17(2):1276-1280. doi: 10.1039/C4GC01951B [43] Granata G, Pagnanelli F, Moscardini E, et al. Simultaneous recycling of nickel metal hydride, lithium ion and primary lithium batteries: Accomplishment of European Guidelines by optimizing mechanical pre-treatment and solvent extraction operations[J]. Journal of Power Sources,2012,212:205-211. doi: 10.1016/j.jpowsour.2012.04.016 [44] Zhang T, He Y Q, Ge L H, et al. Characteristics of wet and dry crushing methods in the recycling process of spent lithium-ion batteries[J]. Journal of Power Sources,2013,240:766-771. doi: 10.1016/j.jpowsour.2013.05.009 [45] Song X L, Dai S Q, Xu Y S, et al. Experimental study on the discharge of the waste lithium ion battery[J]. Applied Chemical Industry,2015,44(4):594. [46] Zhang G W, He Y Q, Feng Y, et al. Enhancement in liberation of electrode materials derived from spent lithium-ion battery by pyrolysis[J]. Journal of Cleaner Production,2018,199:62-68. doi: 10.1016/j.jclepro.2018.07.143 [47] Bi H J, Zhu H B, Zu L, et al. A new model of trajectory in eddy current separation for recovering spent lithium iron phosphate batteries[J]. Waste Management,2019,100:1-9. doi: 10.1016/j.wasman.2019.08.041 [48] Yao L, Yao H S, Xi G X, et al. Recycling and synthesis of LiNi1/3Co1/3Mn1/3O2 from waste lithium ion batteries using D, L-malic acid[J]. Rsc Advances,2016,6(22):17947-17954. doi: 10.1039/C5RA25079J [49] Sun L, Qiu K Q. Organic oxalate as leachant and precipitant for the recovery of valuable metals from spent lithium-ion batteries[J]. Waste Management,2012,32(8):1575-1582. doi: 10.1016/j.wasman.2012.03.027 [50] Hanisch C, Haselrieder W, Kwade A. Recovery of active materials from spent lithium-ion electrodes and electrode production rejects [M]. Glocalized Solutions for Sustainability in Manufacturing. Springer. 2011: 85–89. [51] Chen Y M, Liu N N, Hu F, et al. Thermal treatment and ammoniacal leaching for the recovery of valuable metals from spent lithium-ion batteries[J]. Waste Management,2018,75:469-476. doi: 10.1016/j.wasman.2018.02.024 [52] Ahmed S, Nelson P A, Gallagher K G, et al. Energy impact of cathode drying and solvent recovery during lithium-ion battery manufacturing[J]. Journal of Power Sources,2016,322:169-178. doi: 10.1016/j.jpowsour.2016.04.102 [53] Li L, Zhai L Y, Zhang X X, et al. Recovery of valuable metals from spent lithium-ion batteries by ultrasonic-assisted leaching process[J]. Journal of Power Sources,2014,262:380-385. doi: 10.1016/j.jpowsour.2014.04.013 [54] Yang L, Xi G X, Liu J J. MnZn ferrite synthesized by sol-gel auto-combustion and microwave digestion routes using spent alkaline batteries[J]. Ceramics International,2015,41(3):3555-3560. doi: 10.1016/j.ceramint.2014.11.019 [55] Zeng X, Li J. Innovative application of ionic liquid to separate Al and cathode materials from spent high-power lithium-ion batteries[J]. Journal of Hazardous Materials,2014,271:50-56. doi: 10.1016/j.jhazmat.2014.02.001 [56] Wang M, Tan Q, Liu L, et al. A low-toxicity and high-efficiency deep eutectic solvent for the separation of aluminum foil and cathode materials from spent lithium-ion batteries[J]. Journal of Hazardous Materials,2019:380. [57] Yang S L, Yang K Y, Fan X P, et al. Research on sulfuric acid leaching and recovery of waste lithium-ion battery anode sheets[J]. Power Technology,2020,44(3):4. [58] Contestabile M, Panero S, Scrosati B. A laboratory-scale lithium-ion battery recycling process[J]. Journal of Power Sources,2001,92(1-2):65-69. doi: 10.1016/S0378-7753(00)00523-1 [59] Georgi-Maschler T, Friedrich B, Weyhe R, et al. Development of a recycling process for Li-ion batteries[J]. Journal of Power Sources,2012,207:173-182. doi: 10.1016/j.jpowsour.2012.01.152 [60] Zhang G W, Yuan X, He Y Q, et al. Recent advances in pretreating technology for recycling valuable metals from spent lithium-ion batteries[J]. Journal of Hazardous Materials,2021,406:124332. doi: 10.1016/j.jhazmat.2020.124332 [61] Hu J T, Zhang J L, Li H X, et al. A promising approach for the recovery of high value-added metals from spent lithium-ion batteries[J]. Journal of Power Sources,2017,351:192-199. doi: 10.1016/j.jpowsour.2017.03.093 [62] Li J, Wang G X, Xu Z M. Environmentally-friendly oxygen-free roasting/wet magnetic separation technology for in situ recycling cobalt, lithium carbonate and graphite from spent LiCoO2/graphite lithium batteries[J]. Journal of Hazardous Materials,2016,302:97-104. doi: 10.1016/j.jhazmat.2015.09.050 [63] Xiao J F, Li J, Xu Z M. Novel approach for in situ recovery of lithium carbonate from spent lithium ion batteries using vacuum metallurgy[J]. Environmental Science & Technology,2017,51(20):11960-11966. [64] Xiao J F, Li J, Xu Z M. Recycling metals from lithium ion battery by mechanical separation and vacuum metallurgy[J]. Journal of Hazardous Materials,2017,338:124-131. doi: 10.1016/j.jhazmat.2017.05.024 [65] He L P, Sun S Y, Song X F, et al. Leaching process for recovering valuable metals from the LiNi1/3Co1/3Mn1/3O2 cathode of lithium-ion batteries[J]. Waste Management,2017,64:171-181. doi: 10.1016/j.wasman.2017.02.011 [66] Guan J, Li Y G, Guo Y G, et al. Mechanochemical process enhanced cobalt and lithium recycling from wasted lithium-ion batteries[J]. Acs Sustainable Chemistry & Engineering,2017,5(1):1026-1032. [67] Tanong K, Coudert L, Mercier G, et al. Recovery of metals from a mixture of various spent batteries by a hydrometallurgical process[J]. Journal of Environmental Management,2016,181:95-107. [68] Gratz E, Sa Q, Apelian D, et al. A closed loop process for recycling spent lithium ion batteries[J]. Journal of Power Sources,2014,262:255-262. doi: 10.1016/j.jpowsour.2014.03.126 [69] Pagnanelli F, Moscardini E, Granata G, et al. Acid reducing leaching of cathodic powder from spent lithium ion batteries: Glucose oxidative pathways and particle area evolution[J]. Journal of Industrial and Engineering Chemistry,2014,20(5):3201-3207. doi: 10.1016/j.jiec.2013.11.066 [70] Meshram P, Pandey B D, Mankhand T R. Hydrometallurgical processing of spent lithium ion batteries (LIBs) in the presence of a reducing agent with emphasis on kinetics of leaching[J]. Chemical Engineering Journal,2015,281:418-427. doi: 10.1016/j.cej.2015.06.071 [71] Nayaka G, Pai K, Santhosh G, et al. Recovery of cobalt as cobalt oxalate from spent lithium ion batteries by using glycine as leaching agent[J]. Journal of environmental chemical engineering,2016,4(2):2378-2383. doi: 10.1016/j.jece.2016.04.016 [72] Poyraz A S, Huang J P, Cheng S B, et al. Effective recycling of manganese oxide cathodes for lithium based batteries[J]. Green Chemistry,2016,18(11):3414-3421. doi: 10.1039/C6GC00438E [73] Peng C, Hamuyuni J, Wilson B P, et al. Selective reductive leaching of cobalt and lithium from industrially crushed waste Li-ion batteries in sulfuric acid system[J]. Waste Management,2018,76:582-590. doi: 10.1016/j.wasman.2018.02.052 [74] Liu P C, Xiao L, Tang Y W, et al. Study on the reduction roasting of spent LiNixCoyMnzO2 lithium-ion battery cathode materials[J]. Journal of Thermal Analysis and Calorimetry,2019,136(3):1323-1332. doi: 10.1007/s10973-018-7732-7 [75] Liu Y, Lv W G, Zheng X H, et al. Near-to-stoichiometric acidic recovery of spent lithium-ion batteries through induced crystallization[J]. Acs Sustainable Chemistry & Engineering,2021,9(8):3183-3194. [76] Chen X P, Luo C B, Zhang J X, et al. Sustainable recovery of metals from spent lithium-ion batteries: A green process[J]. Acs Sustainable Chemistry & Engineering,2015,3(12):3104-3113. [77] Li L, Bian Y F, Zhang X X, et al. Process for recycling mixed-cathode materials from spent lithium-ion batteries and kinetics of leaching[J]. Waste Management,2018,71:362-371. doi: 10.1016/j.wasman.2017.10.028 [78] Fu Y P, He Y Q, Qu L L, et al. Enhancement in leaching process of lithium and cobalt from spent lithium-ion batteries using benzenesulfonic acid system[J]. Waste Management,2019,88:191-199. doi: 10.1016/j.wasman.2019.03.044 [79] Gao W F, Zhang X H, Zheng X H, et al. Lithium carbonate recovery from cathode scrap of spent lithium-ion battery: A closed-loop process[J]. Environmental Science & Technology,2017,51(3):1662-1669. [80] Yu M, Zhang Z H, Xue F, et al. A more simple and efficient process for recovery of cobalt and lithium from spent lithium-ion batteries with citric acid[J]. Separation and Purification Technology,2019,215:398-402. doi: 10.1016/j.seppur.2019.01.027 [81] Zhuang L Q, Sun C H, Zhou T, et al. Recovery of valuable metals from LiNi0.5Co0.2Mn0.3O2 cathode materials of spent Li-ion batteries using mild mixed acid as leachant[J]. Waste Management,2019,85:175-185. doi: 10.1016/j.wasman.2018.12.034 [82] Zhu S G, He W Z, Li G M, et al. Recovery of Co and Li from spent lithium-ion batteries by combination method of acid leaching and chemical precipitation[J]. Transactions of Nonferrous Metals Society of China,2012,22(9):2274-2281. doi: 10.1016/S1003-6326(11)61460-X [83] Chen L, Tang X C, Zhang Y, et al. Process for the recovery of cobalt oxalate from spent lithium-ion batteries[J]. Hydrometallurgy,2011,108(1-2):80-86. doi: 10.1016/j.hydromet.2011.02.010 [84] Liu T C, Chen J, Li H L, et al. An integrated process for the separation and recovery of valuable metals from the spent LiNi0.5Co0.2Mn0.3O2 cathode materials[J]. Separation and Purification Technology,2020, 245:116869. [85] Cheng Q, Chirdon W M, Lin M D, et al. Characterization, modeling, and optimization of a single-step process for leaching metallic ions from LiNi1/3Co1/3Mn1/3O2 cathodes for the recycling of spent lithium-ion batteries[J]. Hydrometallurgy,2019,185:1-11. doi: 10.1016/j.hydromet.2019.01.003 [86] Chen H, Gu S, Guo Y X, et al. Leaching of cathode materials from spent lithium-ion batteries by using a mixture of ascorbic acid and HNO3[J]. Hydrometallurgy,2021, 205:105746. [87] Zheng R J, Zhao L, Wang W H, et al. Optimized Li and Fe recovery from spent lithium-ion batteries via a solution-precipitation method[J]. Rsc Advances,2016,6(49):43613-43625. doi: 10.1039/C6RA05477C [88] Shuva M A H, Kurny A. Dissolution kinetics of cathode of spent lithium ion battery in hydrochloric acid solutions[J]. Journal of The Institution of Engineers (India):Series D,2013,94(1):13-16. doi: 10.1007/s40033-013-0018-0 [89] Chen X P, Ma H R, Luo C B, et al. Recovery of valuable metals from waste cathode materials of spent lithium-ion batteries using mild phosphoric acid[J]. Journal of Hazardous Materials,2017,326:77-86. doi: 10.1016/j.jhazmat.2016.12.021 [90] Li L, Lu J, Ren Y, et al. Ascorbic-acid-assisted recovery of cobalt and lithium from spent Li-ion batteries[J]. Journal of Power Sources,2012,218:21-27. doi: 10.1016/j.jpowsour.2012.06.068 [91] Fan B L, Chen X P, Zhou T, et al. A sustainable process for the recovery of valuable metals from spent lithium-ion batteries[J]. Waste Management & Research,2016,34(5):474-481. [92] Li L, Dunn J B, Zhang X X, et al. Recovery of metals from spent lithium-ion batteries with organic acids as leaching reagents and environmental assessment[J]. Journal of Power Sources,2013,233:180-189. doi: 10.1016/j.jpowsour.2012.12.089 [93] Qiu X J, Tian Y, Deng W T, et al. Coupling regeneration strategy of lithium-ion electrode materials turned with naphthalenedisulfonic acid[J]. Waste Management,2021,136:1-10. doi: 10.1016/j.wasman.2021.09.032 [94] Sun C H, Xu L P, Chen X P, et al. Sustainable recovery of valuable metals from spent lithium-ion batteries using DL-malic acid: Leaching and kinetics aspect[J]. Waste Management & Research,2018,36(2):113-120. [95] Hu C Y, Guo J, Wen J, et al. Preparation and electrochemical performance of nano-Co3O4 anode materials from spent Li-ion batteries for lithium-ion batteries[J]. Journal of Materials Science & Technology,2013,29(3):215-220. [96] Ferreira D A, Zimmer Prados L M, Majuste D, et al. Hydrometallurgical separation of aluminium, cobalt, copper and lithium from spent Li-ion batteries[J]. Journal of Power Sources,2009,187(1):238-246. doi: 10.1016/j.jpowsour.2008.10.077 [97] Wang W Q, Zhang Y C, Liu X G, et al. A simplified process for recovery of Li and Co from spent LiCoO2 cathode using Al foil as the in situ reductant[J]. Acs Sustainable Chemistry & Engineering,2019,7(14):12222-12230. [98] Zheng X H, Gao W F, Zhang X, et al. Spent lithium-ion battery recycling-reductive ammonia leaching of metals from cathode scrap by sodium sulphite[J]. Waste Management,2017,60:680-688. doi: 10.1016/j.wasman.2016.12.007 [99] Zhang J L, Liang G Q, Yang C, et al. A breakthrough method for the recycling of spent lithium-ion batteries without pre-sorting[J]. Green Chemistry,2021,23(21):8434-8440. doi: 10.1039/d1gc02132j [100] Mishra D, Kim D J, Ralph D E, et al. Bioleaching of metals from spent lithium ion secondary batteries using acidithiobacillus ferrooxidans[J]. Waste Management,2008,28(2):333-338. doi: 10.1016/j.wasman.2007.01.010 [101] Xin Y Y, Guo X M, Chen S, et al. Bioleaching of valuable metals Li, Co, Ni and Mn from spent electric vehicle Li-ion batteries for the purpose of recovery[J]. Journal of Cleaner Production,2016,116:249-258. doi: 10.1016/j.jclepro.2016.01.001 [102] Biswal B K, Jadhav U U, Madhaiyan M, et al. Biological leaching and chemical precipitation methods for recovery of Co and Li from spent lithium-ion batteries[J]. Acs Sustainable Chemistry & Engineering,2018,6(9):12343-12352. [103] Fan E S, Shi P C, Zhang X X, et al. Glucose oxidase-based biocatalytic acid-leaching process for recovering valuable metals from spent lithium-ion batteries[J]. Waste Management,2020,114:166-173. doi: 10.1016/j.wasman.2020.06.047 [104] Zhang K H, Li B, Wu Y F, et al. Recycling of indium from waste LCD: A promising non-crushing leaching with the aid of ultrasonic wave[J]. Waste Management,2017,64:236-243. doi: 10.1016/j.wasman.2017.03.031 [105] Zhang L B, Guo W Q, Peng J H, et al. Comparison of ultrasonic-assisted and regular leaching of germanium from by-product of zinc metallurgy[J]. Ultrasonics Sonochemistry,2016,31:143-149. doi: 10.1016/j.ultsonch.2015.12.006 [106] Liu K, Zhang F S. Innovative leaching of cobalt and lithium from spent lithium-ion batteries and simultaneous dechlorination of polyvinyl chloride in subcritical water[J]. Journal of Hazardous Materials,2016,316:19-25. doi: 10.1016/j.jhazmat.2016.04.080 [107] Li J S, Yang X Y, Yin Z L. Recovery of manganese from sulfuric acid leaching liquor of spent lithium-ion batteries and synthesis of lithium ion-sieve[J]. Journal of Environmental Chemical Engineering,2018,6(5):6407-6413. doi: 10.1016/j.jece.2018.09.044 [108] Tanong K, Tran L H, Mercier G, et al. Recovery of Zn (II), Mn (II), Cd (II) and Ni (II) from the unsorted spent batteries using solvent extraction, electrodeposition and precipitation methods[J]. Journal of Cleaner Production,2017,148:233-244. doi: 10.1016/j.jclepro.2017.01.158 [109] Virolainen S, Fini M F, Laitinen A, et al. Solvent extraction fractionation of Li-ion battery leachate containing Li, Ni, and Co[J]. Separation and Purification Technology,2017,179:274-82. doi: 10.1016/j.seppur.2017.02.010 [110] Chen X P, Kang D Z, Cao L, et al. Separation and recovery of valuable metals from spent lithium ion batteries: Simultaneous recovery of Li and Co in a single step[J]. Separation and Purification Technology,2019,210:690-697. doi: 10.1016/j.seppur.2018.08.072 [111] Chen X P, Chen Y B, Zhou T, et al. Hydrometallurgical recovery of metal values from sulfuric acid leaching liquor of spent lithium-ion batteries[J]. Waste Management,2015,38:349-356. doi: 10.1016/j.wasman.2014.12.023 [112] Wang F, Sun R, Xu J, et al. Recovery of cobalt from spent lithium ion batteries using sulphuric acid leaching followed by solid-liquid separation and solvent extraction[J]. Rsc Advances,2016,6(88):85303-85311. doi: 10.1039/C6RA16801A [113] Hong H S, Kim D W, Choi H L, et al. Solvent extraction of Co, Ni and Mn from NCM sulfate leaching solution of Li (NCM) O2 secondary battery scraps[J]. Archives of Metallurgy and Materials,2017, 62(2):1011-1014. [114] Contestabile M, Panero S, Scrosati B. A laboratory-scale lithium battery recycling process[J]. Journal of Power Sources,1999,83(1-2):75-78. doi: 10.1016/S0378-7753(99)00261-X [115] Zhang P, Yokoyama T, Itabashi O, et al. Hydrometallurgical process for recovery of metal values from spent lithium-ion secondary batteries[J]. Hydrometallurgy,1998,47(2-3):259-271. doi: 10.1016/S0304-386X(97)00050-9 [116] Freitas M B J G, Garcia E M. Electrochemical recycling of cobalt from cathodes of spent lithium-ion batteries[J]. Journal of Power Sources,2007,171(2):953-959. doi: 10.1016/j.jpowsour.2007.07.002 [117] Lupi C, Pasquali M. Electrolytic nickel recovery from lithium-ion batteries[J]. Minerals Engineering,2003,16(6):537-542. doi: 10.1016/S0892-6875(03)00080-3 [118] Nishide H, Deguchi J, Tsuchida E. Selective adsorption of metal ions on crosslinked poly (vinylpyridine) resin prepared with a metal ion as a template[J]. Chemistry letters,1976,5(2):169-174. doi: 10.1246/cl.1976.169 [119] Yu C, Lu J, Dai J W, et al. Bio-inspired fabrication of Ester-functionalized imprinted composite membrane for rapid and high-efficient recovery of lithium ion from seawater[J]. Journal of colloid and interface science,2020,572:340-353. doi: 10.1016/j.jcis.2020.03.091 [120] Sun D S, Meng M J, Qiao Y, et al. Synthesis of ion imprinted nanocomposite membranes for selective adsorption of lithium[J]. Separation and Purification Technology,2018,194:64-72. doi: 10.1016/j.seppur.2017.10.052 [121] Donato L, Drioli E. Imprinted membranes for sustainable separation processes[J]. Frontiers of Chemical Science and Engineering,2021,15(4):775-792. doi: 10.1007/s11705-020-1991-0 [122] Sun D S, Zhu Y Z, Meng M J, et al. Fabrication of highly selective ion imprinted macroporous membranes with crown ether for targeted separation of lithium ion[J]. Separation and Purification Technology,2017,175:19-26. doi: 10.1016/j.seppur.2016.11.029 [123] Cui J Y, Zhou Z P, Xie A T, et al. Facile synthesis of degradable CA/CS imprinted membrane by hydrolysis polymerization for effective separation and recovery of Li+[J]. Carbohydrate Polymers,2019,205:492-499. doi: 10.1016/j.carbpol.2018.10.094 [124] Lu J, Qin Y Y, Zhang Q, et al. Multilayered ion-imprinted membranes with high selectivity towards Li+ based on the synergistic effect of 12-crown-4 and polyether sulfone[J]. Applied Surface Science,2018,427:931-941. doi: 10.1016/j.apsusc.2017.08.016 [125] Li Z, He G Z, Zhao G C, et al. Preparation of a novel ion-imprinted membrane using sodium periodate-oxidized polydopamine as the interface adhesion layer for the direction separation of Li+ from spent lithium-ion battery leaching solution[J]. Separation and Purification Technology,2021, 277:119519. [126] Wang H Y, Huang K, Zhang Y, et al. Recovery of lithium, nickel, and cobalt from spent lithium-ion battery powders by selective ammonia leaching and an adsorption separation system[J]. Acs Sustainable Chemistry & Engineering,2017,5(12):11489-11495. [127] Yang Y, Song S L, Lei S Y, et al. A process for combination of recycling lithium and regenerating graphite from spent lithium-ion battery[J]. Waste Management,2019,85:529-537. doi: 10.1016/j.wasman.2019.01.008 [128] Ma X T, Chen M Y, Chen B, et al. High-performance graphite recovered from spent lithium-ion batteries[J]. Acs Sustainable Chemistry & Engineering,2019,7(24):19732-19738. [129] Guo Y, Li F, Zhu H C, et al. Leaching lithium from the anode electrode materials of spent lithium-ion batteries by hydrochloric acid (HCl)[J]. Waste Management,2016,51:227-233. doi: 10.1016/j.wasman.2015.11.036 [130] Liu Z P, Guo Y, He W Z, et al. Study on the leaching of lithium from anode active materials of waste lithium batteries[J]. Environmental Science and Technology,2015(S2):93-95. [131] Mclaughlin J W. Li reclamation process: 5888463A[P]. 1999-03-30. [132] Qiu Z Z, Yi X W, Liang Q S, et al. A recycling process of waste lithium-ion battery electrolyte: China, CNl08281729A[P]. 2018-07-13. [133] Steven E. System and method for removing all electrolyte from an energy storage and/or conversion device using a supercrifieal fluid: W02003061056[P]. 2003-07-24. [134] Mu D, Liu Y, Li R, et al. Transcritical CO2 extraction of electrolytes for lithium-ion batteries: optimization of the recycling process and quality–quantity variation[J]. New Journal of Chemistry,2017,41(15):7177-7185. doi: 10.1039/C7NJ00771J [135] Liu Y, Mu D, Dai Y, et al. Analysis on extraction behaviour of lithium-ion battery electrolyte solvents in supercritical CO2 by gas chromatography[J]. International Journal of Electrochemical Science,2016,11(9):7594-7604. [136] Cao H, Wen L, Guo Z Q, et al. Application analysis and prospect of carbon materials in low temperature lithium iron phosphate materials[J]. New Carbon Materials,2022,37(1):46-58. doi: 10.1016/S1872-5805(22)60584-5 [137] Jin C B, Shi P, Zhang X Q, et al. Advances in carbon materials for stable lithium metal batteries[J]. New Carbon Materials,2022,37(1):1-24. doi: 10.1016/S1872-5805(22)60573-0 -






 下载:
下载: