-
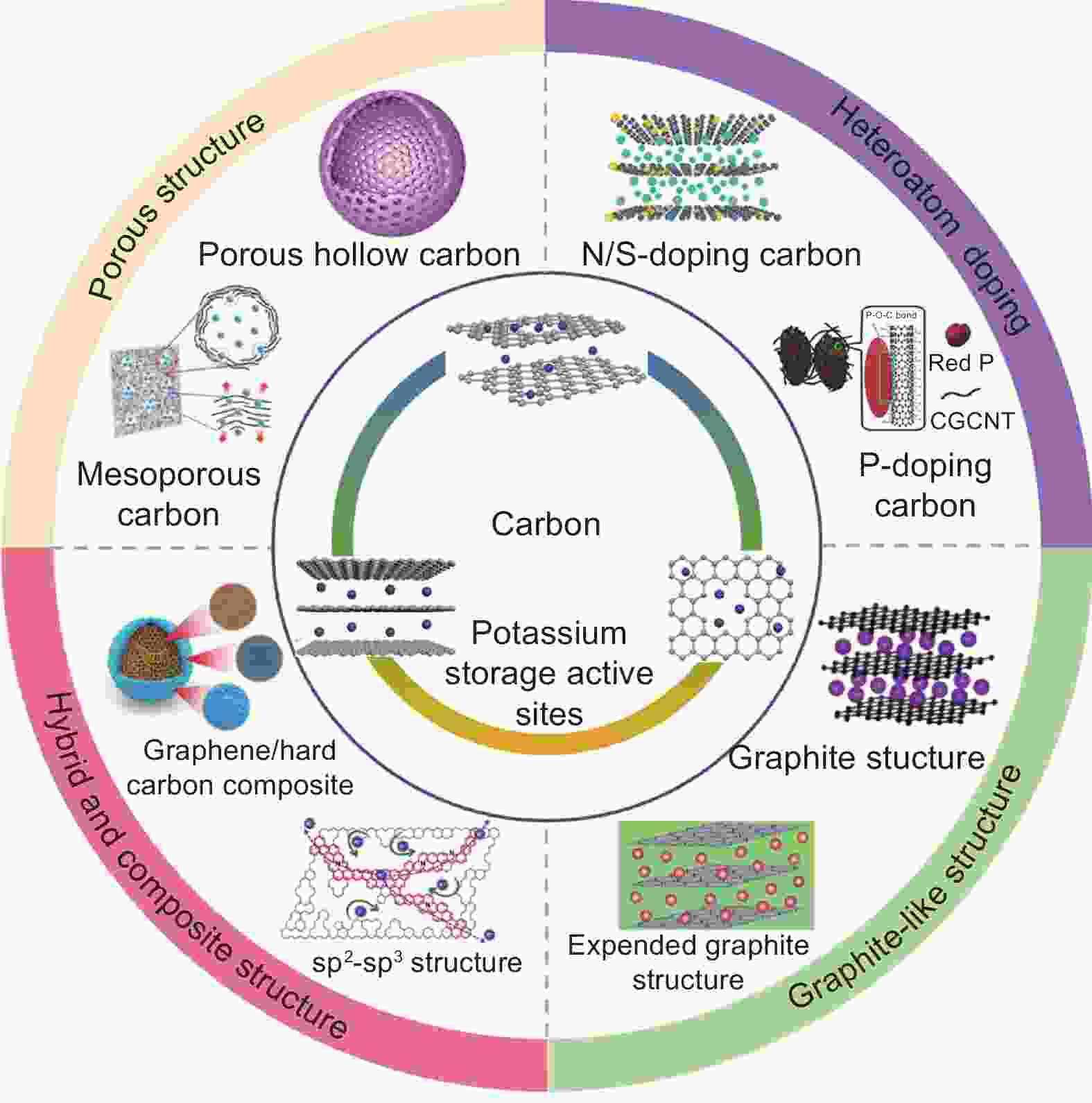
摘要: 炭材料因低成本、无毒性和微观结构可调等优点被认为是最具应用潜力的钾离子电池负极材料,其电化学储钾行为与活性位点的类型密切相关。近年来,为了满足不同电化学储钾器件的应用需求,针对炭结构设计及其活性位点调控已取得大量研究进展。本文讨论了层间插层和离子吸附两种储钾机制的差异,以指导炭结构的合理设计。在此基础上,从库仑效率、容量、电位、倍率和稳定性等方面,综述了不同炭材料的活性位点演变规律及其对储钾性能的影响。同时,总结了炭材料用作钾离子全电池和钾离子电容器负极材料时的结构设计原则和储钾机制差异。并指出了炭材料储钾在活性位点设计方面需要解决的问题及今后研究和改进方向。Abstract: Carbon materials have attracted considerable attention as anodes for potassium ion batteries owing to their low-cost, nontoxicity, and controllable structures. The potassium storage behavior of carbon materials is highly associated with their active sites. In recent years, significant advances have been made in designing the active sites of carbon materials to meet the requirements of different potassium-based storage devices. Here, potassium storage mechanisms (intercalation and adsorption) for guiding the rational design of carbon materials are discussed. Based on these mechanisms, the review provides fundamental insight into the relationship between the structures and potassium storage performance of different carbon materials, including graphite, soft carbon, hard carbon, porous carbon, heteroatom-doped carbon, hybridized carbon and composited carbon. The structural design principles of carbon anode materials for potassium-ion full cell and potassium-ion capacitors are summarized based on the initial coulombic efficiency, capacity, potential plateau, rate performance, and cyclic stability. Finally, the problems and future research directions for the design of active sites in carbon materials for electrochemical potassium storage are considered.
-
Key words:
- Carbon materials /
- Active site /
- Potassium-ion battery /
- Anode materials
-
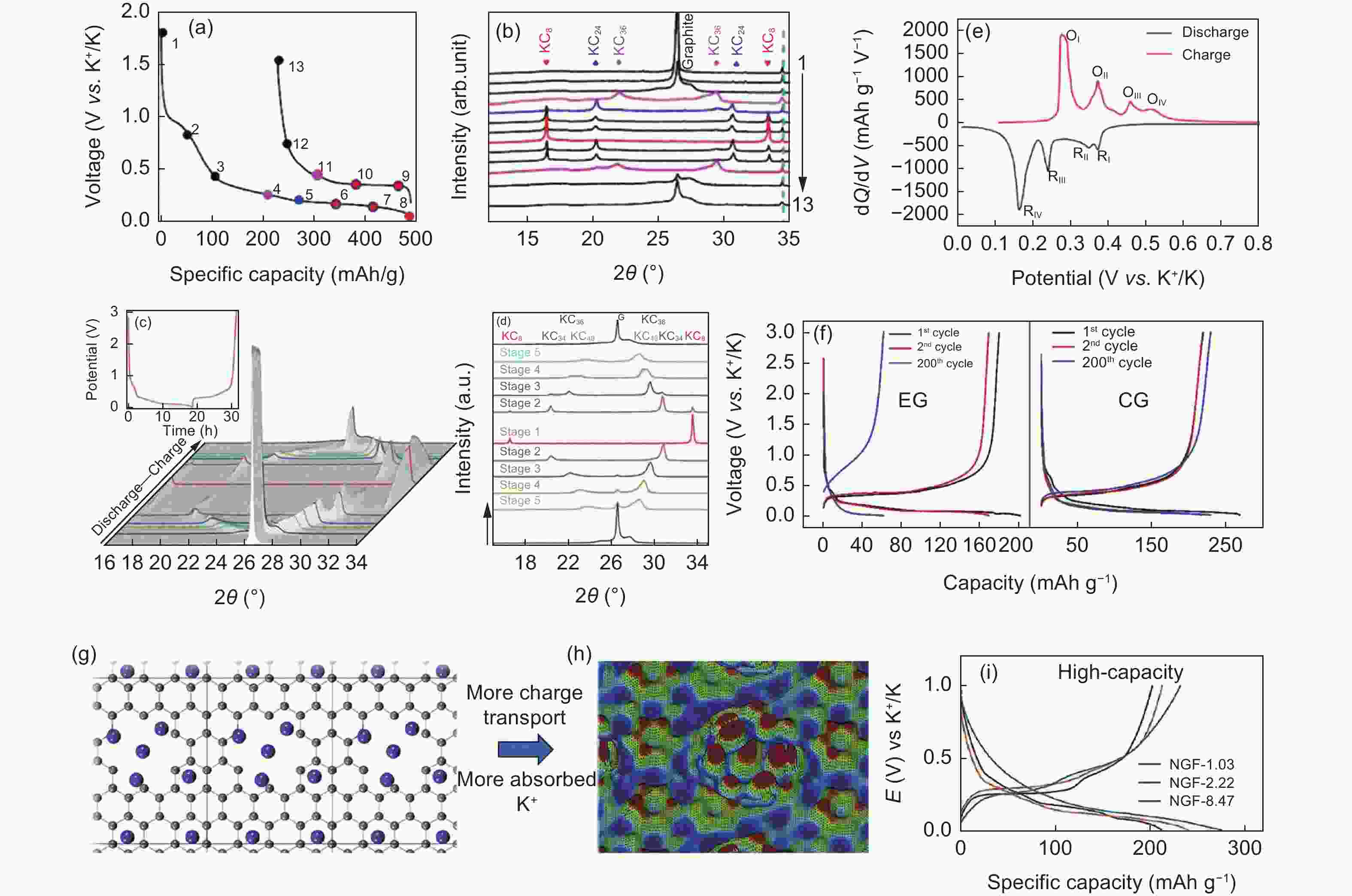
Figure 4. (a) The initial discharge/charge profile of a graphite electrode at C/10 (1 C = 279 mA g-1) and (b) XRD patterns of electrodes corresponding to the marked stage compounds of charge in panel (a)[45]. (c) Waterfall representation XRD patterns at the first cycle and (d) the selected XRD patterns during the initial discharge/charge with different stages. (e) The dQ/dV profiles[69]and (f) galvanostatic charge/discharge voltage profiles at 50 mA g-1 of CG and EG[26]. (g) The structure of high-density K adsorbed around pores in graphene. (h) Electron charge density distribution differences and (i) the galvanostatic charge/discharge profiles of the NGF electrode[70](Reproduced with permission).
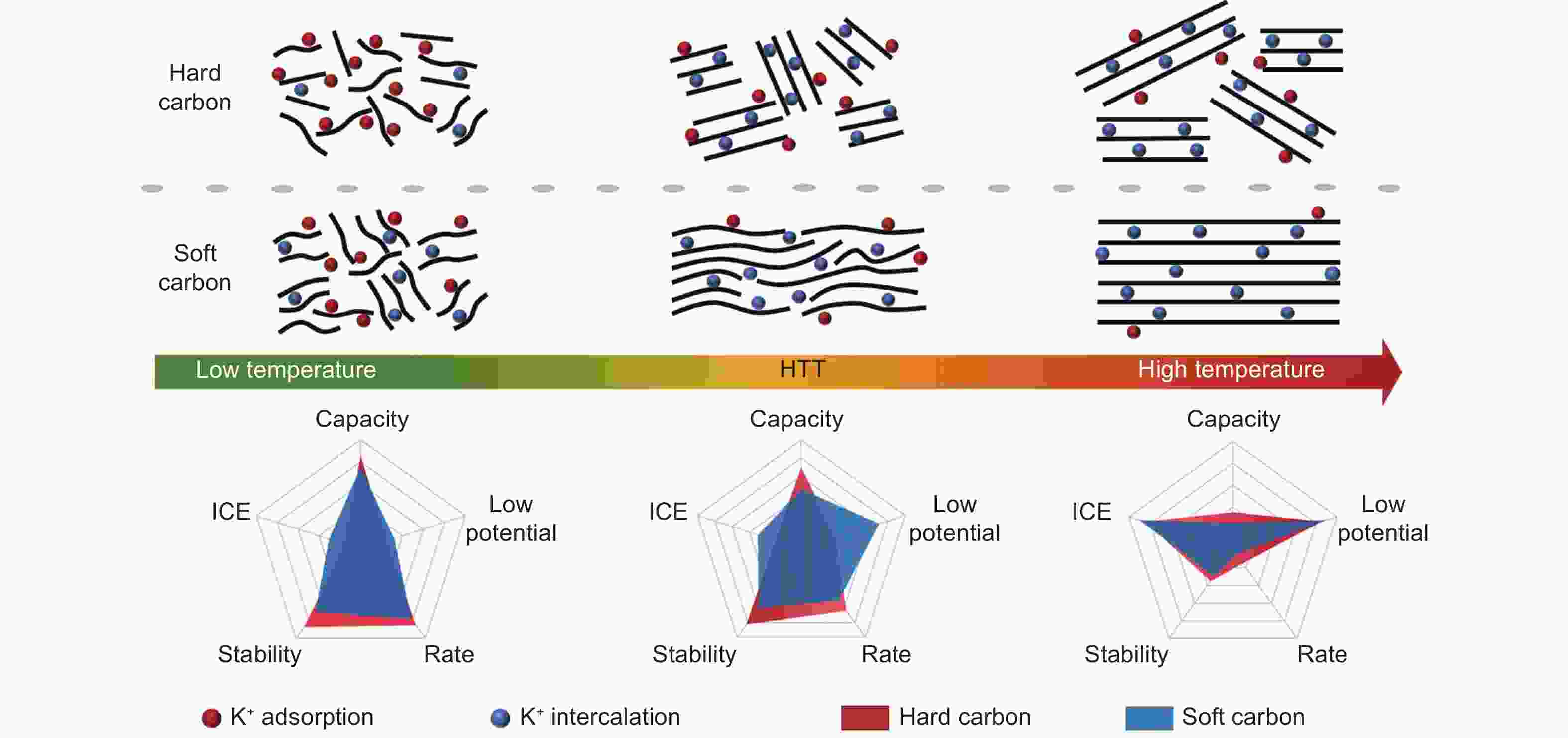
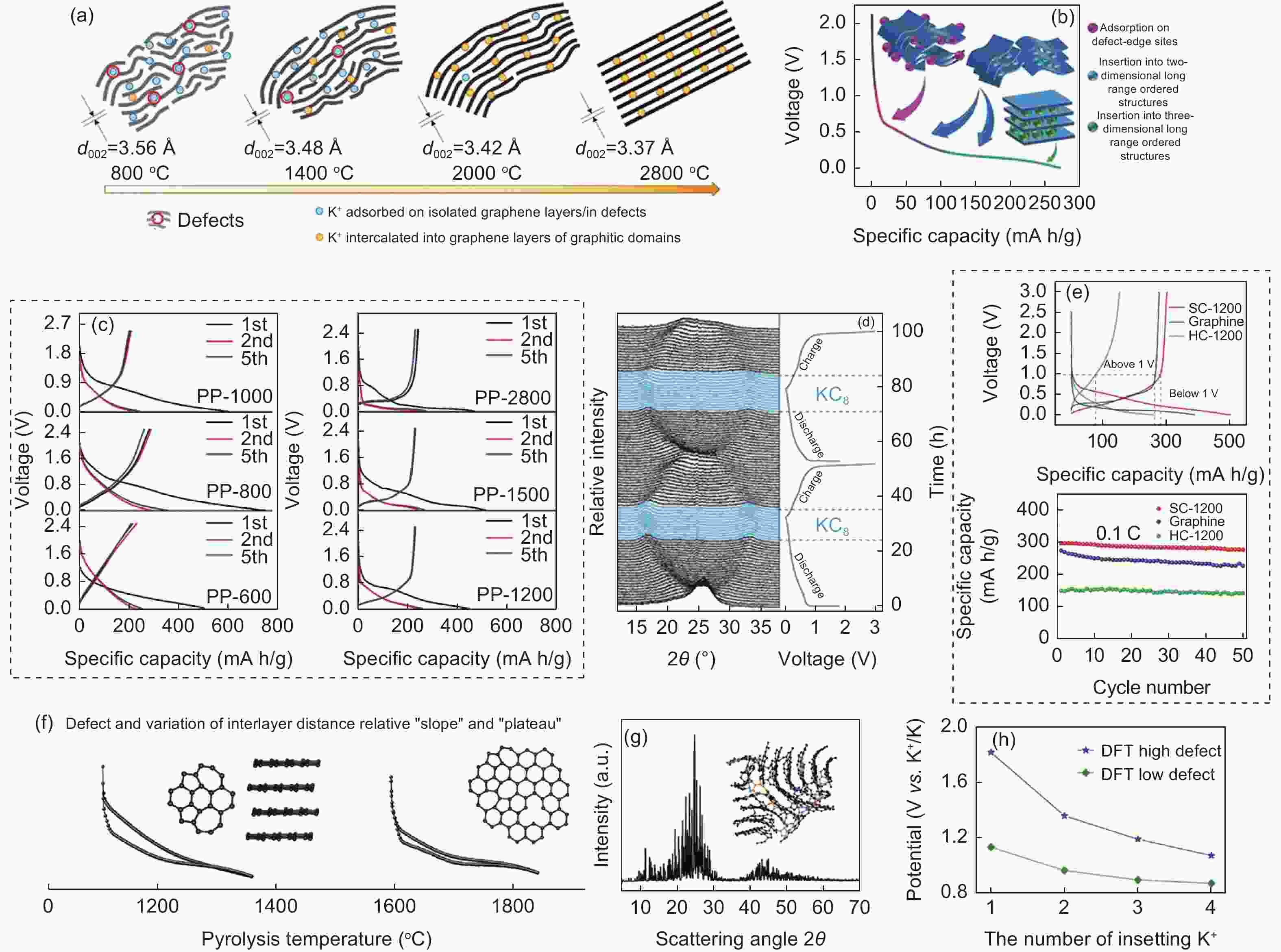
Figure 5. (a) Correlation between the microstructure of soft carbon and HTT[53]. (b) The potassium storage mechanism of soft carbon at different stages during discharge. (c) Discharge/charge curves of soft carbon at different HTTs for cycles 1, 2 and 5[19]. (d) The in-situ XRD spectra of SC-1200. (e) The discharge/charge profiles in the 1st cycle and cyclability test at 0.1 C of SC-1200, graphite, and HC-1200[52]. (f) Schematic diagram of the major determinants affecting potential profiles of potassium storage. (g) Typical structure fragment and the calculated WAXS pattern based on RMC modeling. (h) Effect of the defect concentration on potential revealed by DFT calculation[72](Reproduced with permission).
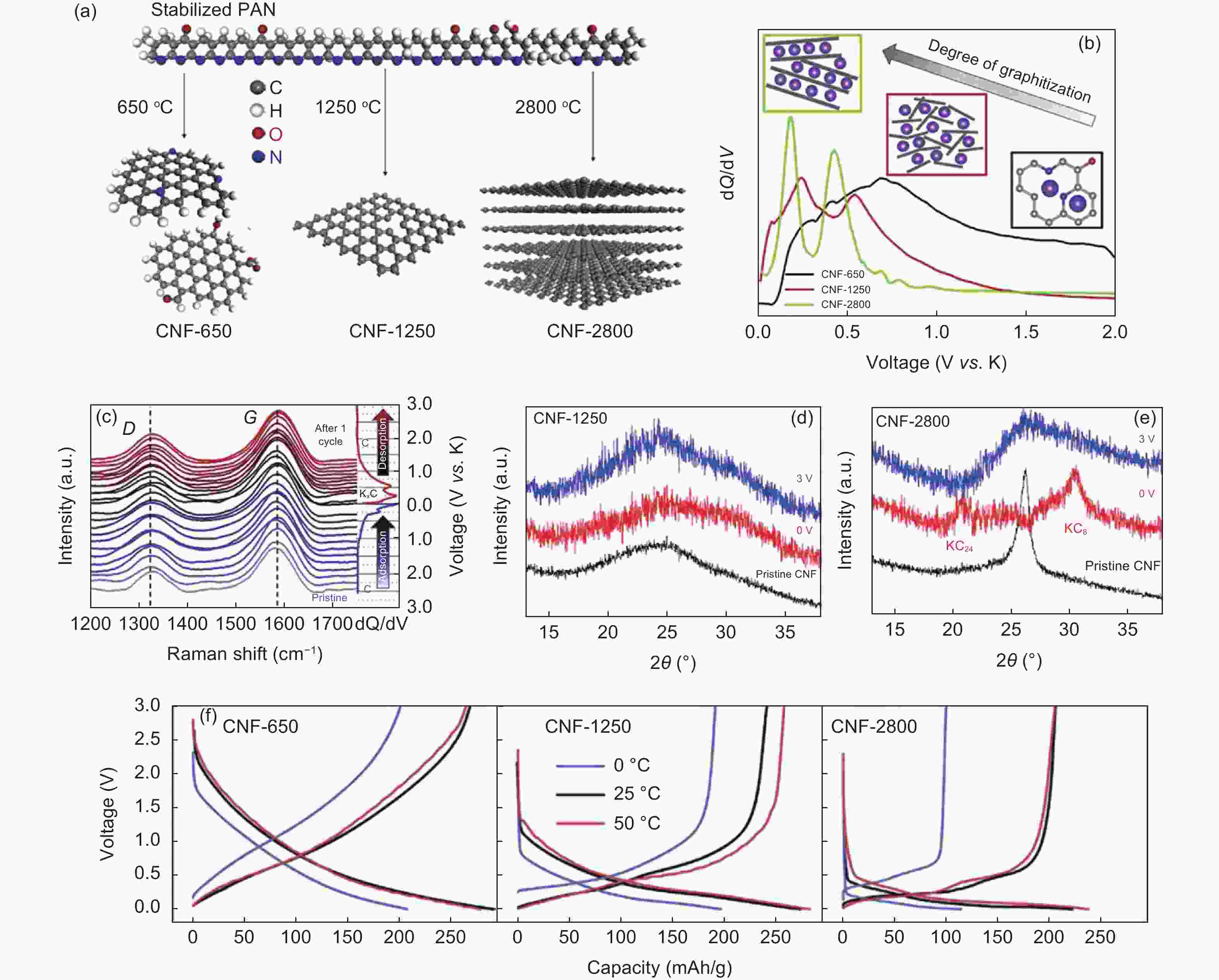
Figure 6. (a) Correlation between the microstructure of CNFs and the annealing temperature. (b) The dQ/dV curves of CNF films after oxidation, the inset illustrates the main K+ storage mechanism in three different regions. (c) Selective in-situ Raman spectra of CNF-1250 with corresponding dQ/dV plot. The in-situ XRD pattern recorded on the first discharge/charge for (d) CNF-1250 and (e) CNF-2800. (f) Discharge/charge curves of CNFs at different working temperatures[67](Reproduced with permission).
Figure 7. TEM images and SAED patterns of hard carbon samples: (a) HC700 and (b) HC2000. (c) Schematic illustration of microstructures of the hard carbon samples prepared at 700 and 2000 °C. (d) Powder XRD patterns of carbon samples prepared at different HTTs. (e) Interlayer distances of carbon layers[73]. (f) The charge-discharge curves of SP-HCs. (g) The dQ/dV curves of SP-HCs. (h) The K+ storage mechanism in hard carbon[74]. (i) the capacities of sloping-voltage and plateau-voltage regions in relation to HTT[75]. (j) Schematic diagram of K+ storage mechanism in CQDHC[38](Reproduced with permission).
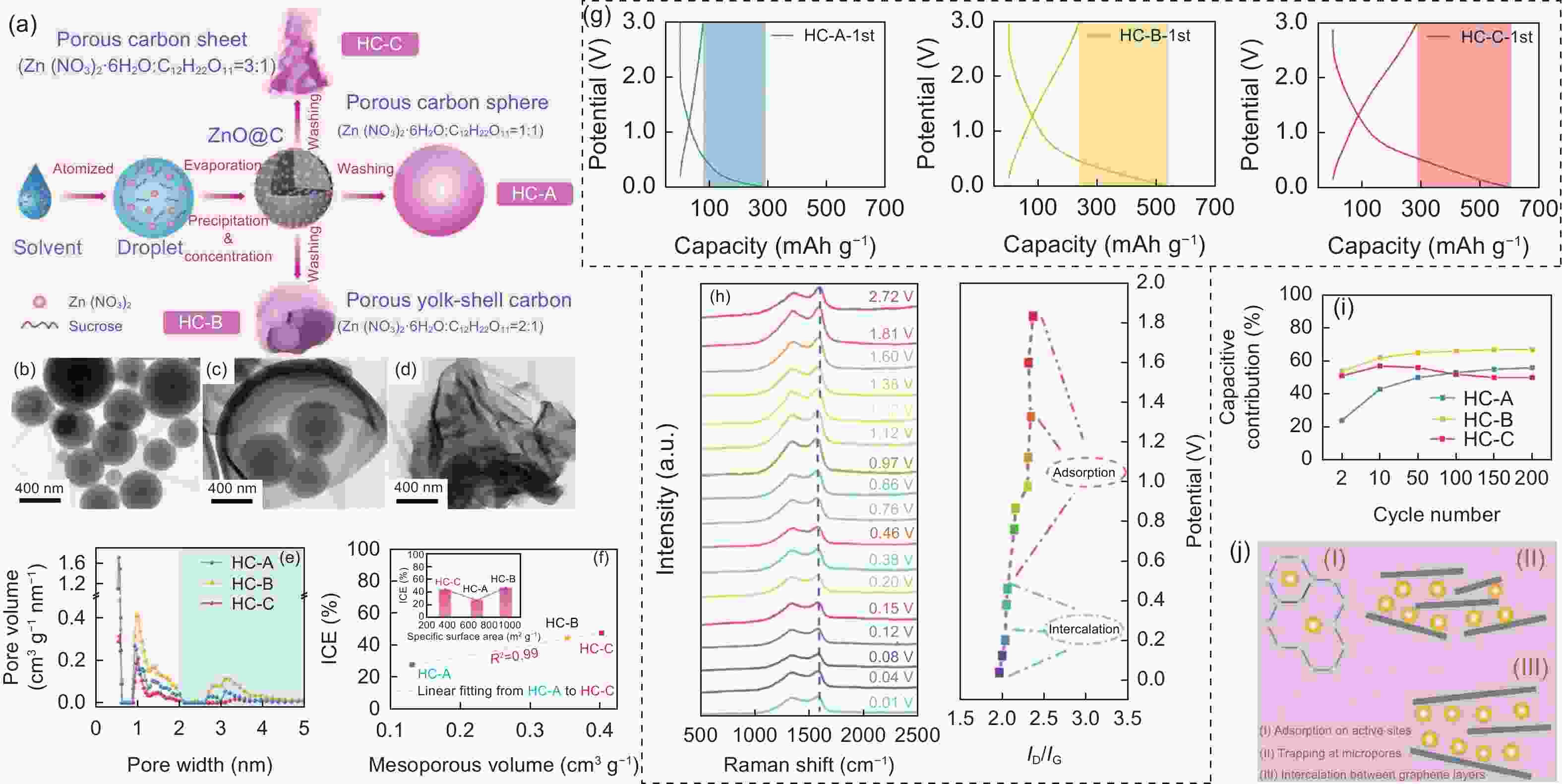
Figure 8. (a) Schematic diagram of the synthesis procedure for the porous carbons by spray pyrolysis. TEM images of (b) HC-A, (c) HC-B and (d) HC-C. (e) Pore size distribution curves, (f) relationship between mesopore volume and ICE and (g) first charge/discharge curves of HC-A, HC-B and HC-C electrode at 0.1 A g−1. (h) In-situ Raman spectra of the HC-B electrode, and the corresponding variation of ID/IG ratio. (i) Capacitive contribution of different HCs during cycling process. (j) Scheme illustration of K+ storage mechanisms in HCs[57](Reproduced with permission).
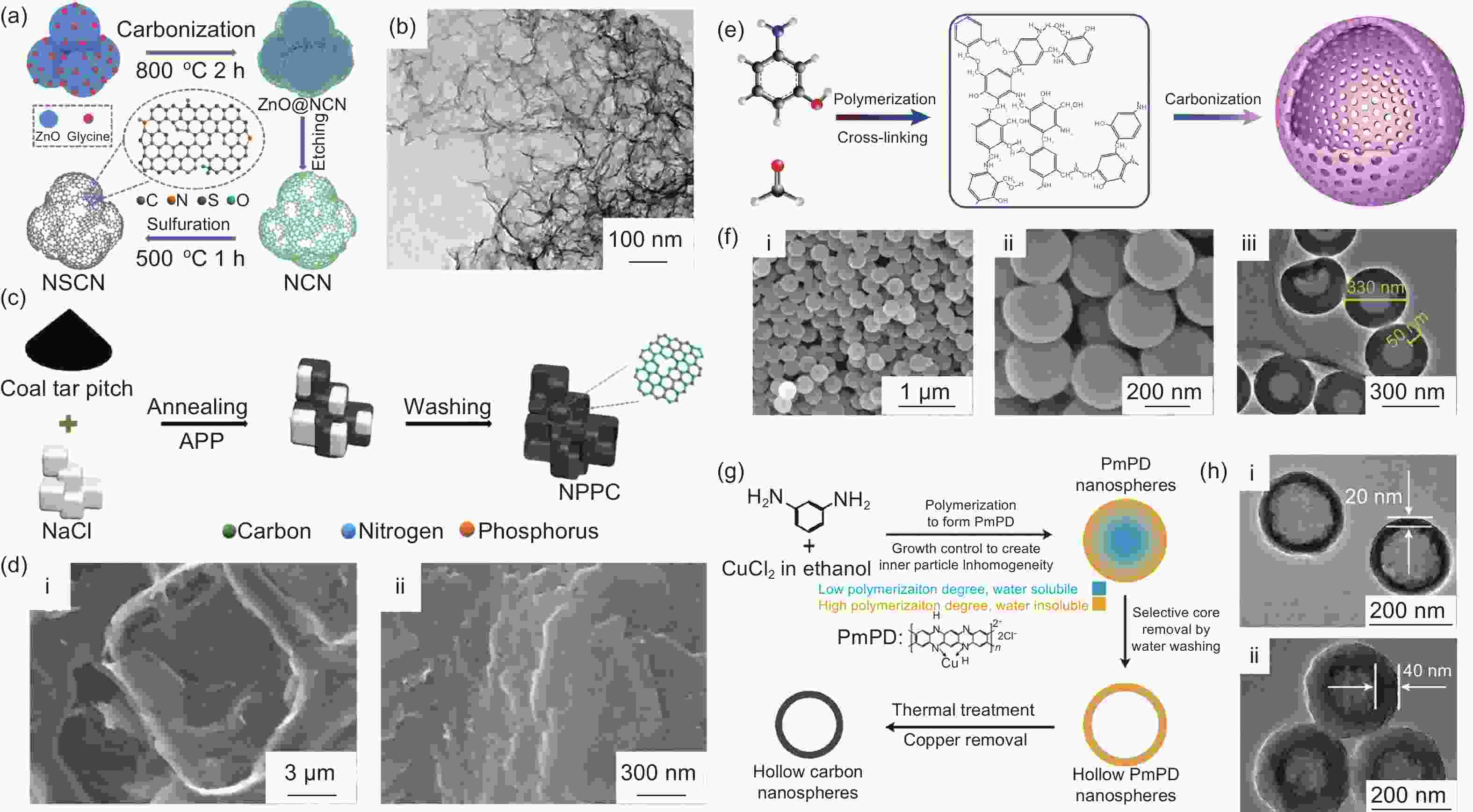
Figure 9. (a) Schematic illustration of the synthesis process of NSCN. (b) SEM image of NSCN [36]. (c) Schematic illustration for the synthesis of NPPC. (d) (i) SEM and (ii) high-magnification SEM images of NPPC[59]. (e) Preparation principle of NHCS. (f) (i), (ii) SEM images, and (iii) TEM image of NHCS[86]. (g) Synthesis protocol for hollow carbon nanospheres. (h) TEM image of the hollow carbon spheres by reaction for (i) 20 min and (ii) 40 min[87](Reproduced with permission).
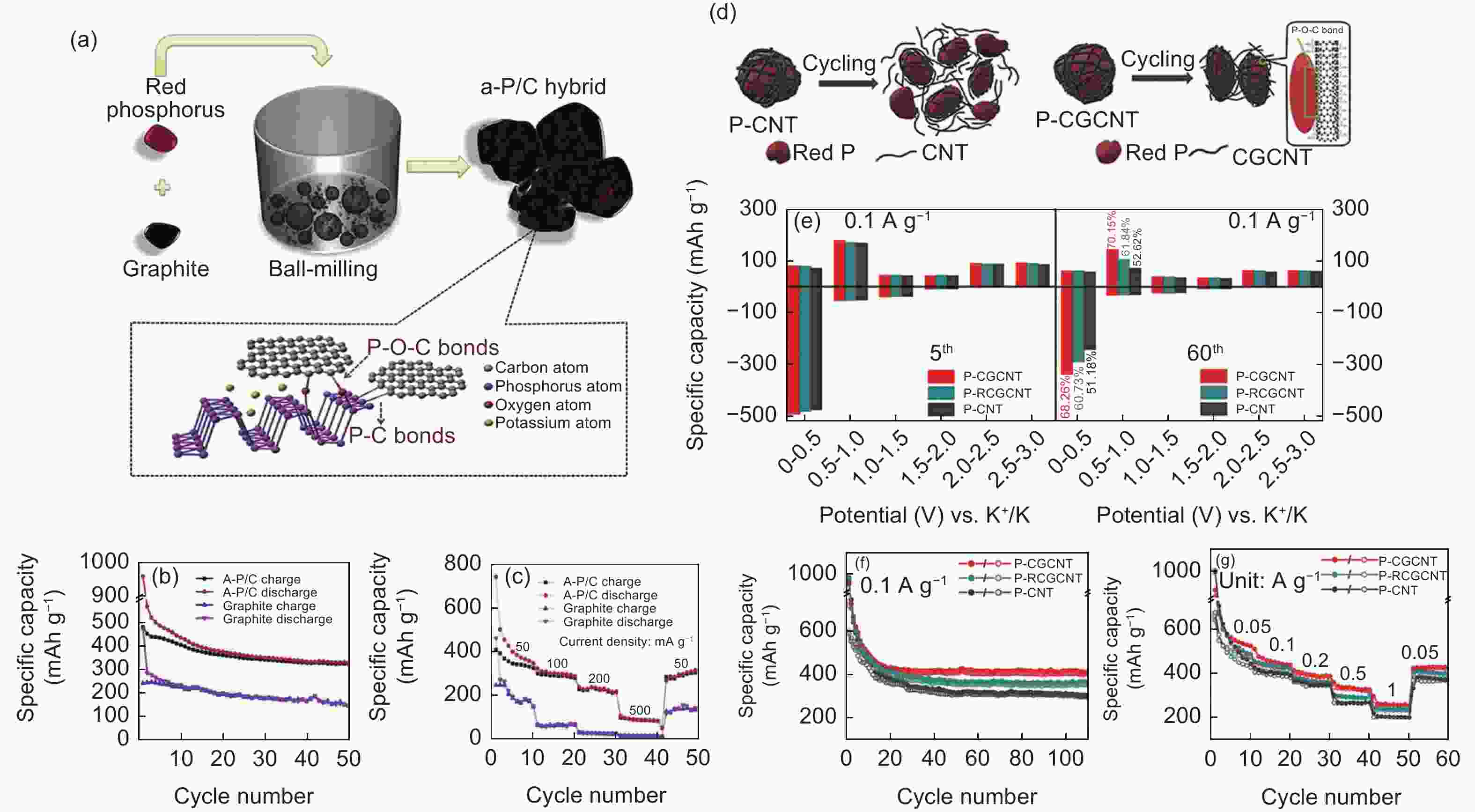
Figure 10. (a) Schematic diagram of the synthesis of the P/C composite. (b) The cycling performance and (c) the rate performance of the P/C and pure graphite electrodes[62]. (d) Schematic illustration of the structure evolution of P-CNT and P-CGCNT electrodes during cycling. (e) The capacity retention in different voltage ranges of P-CGCNT and P-CNT electrodes at the 5th and 60th cycles. (f) The cycling performance and (g) the rate performance of P-CGCNT, P-RCGCNT, and P-CNT electrodes[40](Reproduced with permission).
Figure 12. (a) Schematic illustration of the construction of CNSs. (b) Schematic illustration of the alignment of carbon atoms on the nanobubble surface to create the order-in-disorder structure. Schematic illustration of the K+ storage mechanism in CNSs obtained by (c) low and (d) high pyrolysis temperatures. (e) TEM images of CNS-1000. (f) Rate performance of CNS electrodes. (g) The variation of specific surface area, ID/IG and electric conductivity of CNSs with the pyrolysis temperature[10](Reproduced with permission).
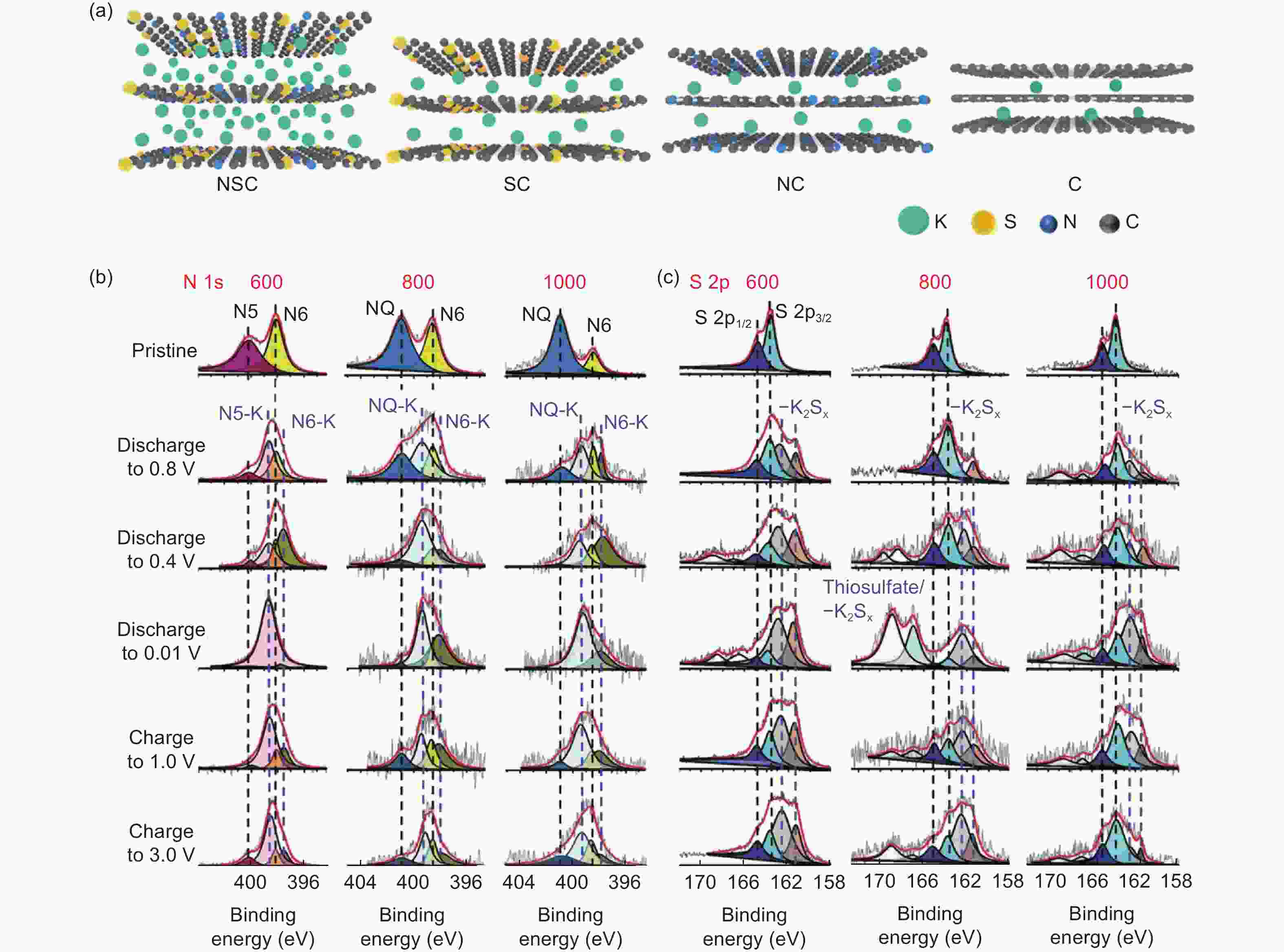
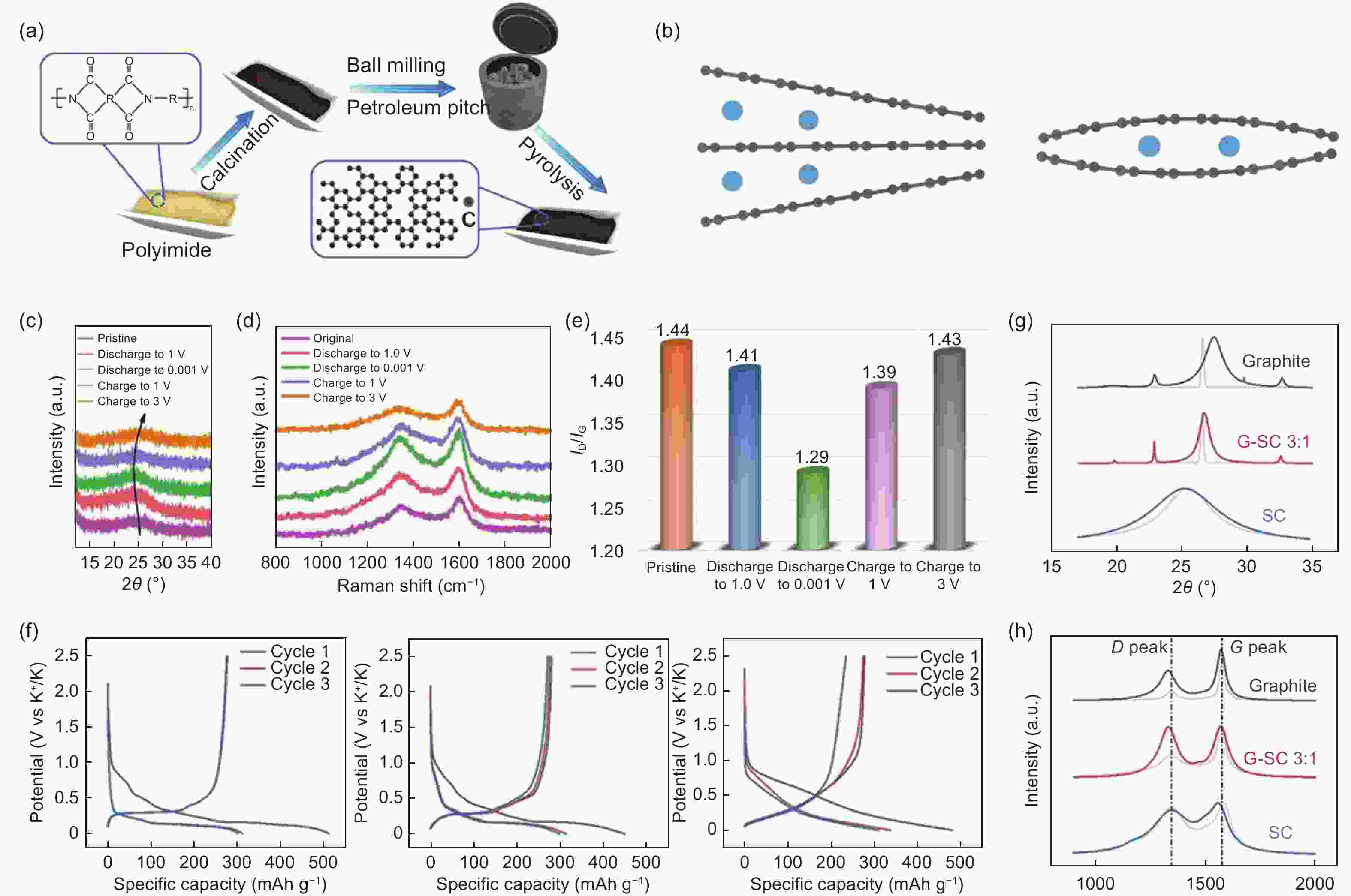
Figure 13. (a) Schematic illustration of the preparation of the hard-soft carbon composites[63]. (b) Unfavorable intercalation with accumulated K+ at one edge of graphite and local bending of the graphite layer locking K+[28]. (c) Ex-situ XRD patterns and (d) Raman spectra of PI-700-P28 electrode at typical charge/discharge stages and their pristine states. (e) ID/IG ratios of PI-700-P28 electrode at different charge/discharge states. (f) GCD curves at 0.1 C of G (left), G-SC 3∶1 (middle) and SC (right). (g) Ex-situ XRD patterns of G, G-SC 3∶1 and SC. (h) Ex-situ Raman spectra of G, G-SC 3∶1 and SC (Reproduced with permission).
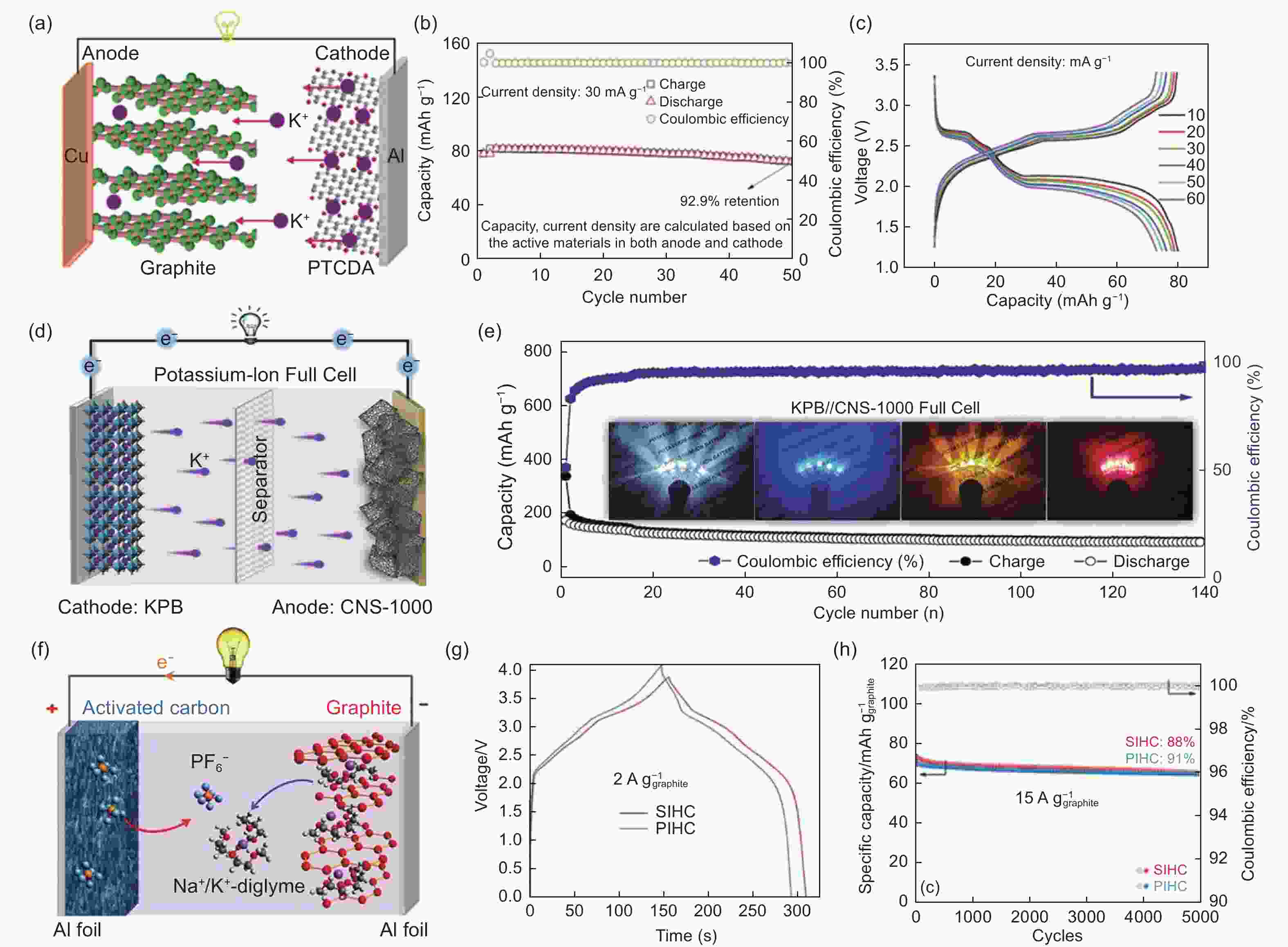
Figure 14. Potassium-ion full cell and potassium-ion hybrid capacitor based on the carbon electrode. (a) Schematic illustration of the graphite/PTCDA full cell. (b) Cycle stability at 30 mA g−1. (c) Charge/discharge curves at different current densities[69]. (d) Schematic illustration of the hard carbon/KPB full-cell. (e) The long cycle performance of the full cell at 0.1 A g−1[10]. (f) Schematic illustration for the working mechanism of PIHC. (g) Charge/discharge curves at 2 A g−1 and (h) prolonged cycling of the PIHC[111](Reproduced with permission).
Table 1. A comparison of the K+ storage performance for different carbon materials.
Materials Capacity
(mAh g−1)Potential
(V)ICE
(%)Rate capability
(mAh g−1)Capacity retention Mass loading
(mg cm−2)Ref. Graphite Graphite 273 at 0.028 A g−1 ~0.2 57.4 80 at 0.28 A g−1 50.8% after 50 cycles ~2 [45] Expanded graphite 267 at 0.05 A g−1 ~0.3 81.6 175 at 0.2 A g−1 99% after 500 cycles ~2 [26] N-doped graphitic carbon 266 at 0.05 A g−1 ~0.3 48.7 228.9 at 2 A g−1 188.9 after 2200 cycles 1-1.1 [46] Polynanocr-ystalline graphite 224 at 0.02 A g−1 ~0.2 54.1 99 at 0.2 A g−1 50% after 240 cycles ~2 [47] Graphite 260 at 0.028 A g−1 ~0.3 81.5 250 at 1.4 A g−1 99.9% after 300 cycles ~0.7 [48] Soft carbon Soft carbon 264 at 0.028 A g−1 ~0.5 56.4 140 at 1.4 A g−1 81.4% after 50 cycles ~2 [45]
N-doped carbon nanofibers248 at 0.025 A g−1 ~0.65 49 101 at 20 A g−1 \ ~1.5 [49] N-doped soft carbon 303 at 0.05 A g−1 ~0.5 30.9 141 at 5 A g−1 85.5% after 500 cycles ~1 [50] N/S-doped soft carbon 359 at 0.1 A g−1 ~0.5 50 115 at 5 A g−1 79.5% after 200 cycles 1-1.2 [51] Pitch-derived soft carbon (1200 °C) 296 at 0.028 A g−1 ~0.2 65 115.2 at 1.4 A g−1 93.2% after 50 cycles \ [52] Pitch-derived soft carbon (1400 °C) 194 at 0.025 A g−1 ~0.27 70 110 at 0.5 A g−1 75% after 100 cycles ~2 [53] Pitch-derived soft carbon (1500 °C) 240 at 0.028 A g−1 ~0.24 \ 80 at 1.4 A g−1 67.9% after 500 cycles ~2 [19] Hard carbon Rhodanine-derived hard carbon 425 at 0.05 A g−1 \ \ 237.4 at 1 A g−1 90.4% after 10th to 400th cycles ~0.7 [10] Biomass-derived hard carbon 442.4 at 0.03 A g−1 ~0.6 \ 175 at 2 A g−1 \ ~0.17 [54] quantum dots in hard carbon 325 at 0.1 A g−1 \ 39 110 at 1 A g−1 75% after 150 cycles ~0.3 [38] N/S-doped hard carbon 250 at 0.1 A g−1 ~0.5 35.2 174 at 3 A g−1 66% after 1200 cycles \ [55] N/O-doped hard carbon 230 at 0.05 A g−1 \ 45.4 118 at 3 A g−1 72% after 1100 cycles ~0.9 [39] N/P-doped hard carbon 312 at 0.05 A g−1 \ 44 179 at 5 A g−1 80% after 500 cycles \ [56] Porous carbon Porous hard carbon 237.6 at 0.1 A g−1 ~0.8 44.4 81.6 at 2 A g−1 \ 1.2-1.6 [57] Porous hard carbon 259 at 0.05 A g−1 ~0.5 44.6 214 at 2 A g−1 84% after 5000 cycles 1.1-1.3 [58] Mesoporous carbon 460 at 0.05 A g−1 \ \ 110 at 4 A g−1 71.3% after 2000 cycles 0.8-1.2 [16] Nanocapsules carbon 293 at 0.05 A g−1 ~0.5 30.9 151 at 5 A g−1 85.5% after 500 cycles ~1. [50] N/S co-doped nanocapsules carbon 408 at 0.05 A g−1 \ 33 149 at 5 A g−1 81% after 10th to 2000th cycles ~1.2 [36]
N/P co-doped porous carbon301 at 0.025 A g−1 ~0.5 60.04 126 at 10 A g−1 81.8% after 400 cycles 1-1.4 [59] Hierarchical porous carbon 211.5 at 0.05 A g−1 \ 24.1 76.7 at 10 A g−1 53% after 100 cycles 0.8-1 [60] Heteroatom-doping carbon N-doped carbon 248 at 0.025 A g−1 ~0.65 49 101 at 20 A g−1 \ ~1.5 [49] S-doped carbon 310 at 0.05 A g−1 ~0.75 42 182.7 at 2 A g−1 68% after 2000 cycles ~2.4 [61] P-doped carbon 323.5 at 0.05 A g−1 ~0.5 50.3 90 at 0.5 A g−1 53.7% after 500 cycles \ [62] P-doped carbon 402.6 at 0.1 A g−1 ~0.75 \ 258 at 1 A g−1 \ \ [40] N/O co-doped carbon 365 at 0.025 A g−1 \ 38 118 at 3 A g−1 69.5% after 1100 cycles ~0.9 [39] N/S co-doped carbon 359 at 0.1 A g−1 \ 50 115 at 5 A g−1 92% after 1000 cycles 1-1.2 [51] Hybrid and composite carbon sp2-sp3 carbon 425 at 0.05 A g−1 \ \ 237.4 at 1 A g−1 90.4% after 10th to 400th cycles ~0.7 [10] sp-sp2 carbon 505 at 0.05 A g−1 \ \ 150 at 5 A g−1 90% after 2000 cycles ~0.8 [102] Hard-soft carbon 376.8 at 0.025 A g−1 ~0.68 71.0 101.2 at 4 A g−1 \ ~1.2 [63] Graphite-soft carbon 280 at 0.028 A g−1 ~0.8 67.3 \ \ 2-2.5 [28] Graphite-hard carbon 253 at 0.05 A g−1 ~0.18 61.8 215.7 at 0.2 A g−1 97.5% after 1000 cycles \ [29] Graphene-hard carbon 297.89 at 0.1 A g−1 ~0.25 \ 220 at 1 A g−1 99.4% after 3200 cycles ~1.2 [66] -
[1] Wu Y M, Zhao H T, Wu Z G, et al. Rational design of carbon materials as anodes for potassium-ion batteries[J]. Energy Storage Materials,2021,34:483-507. doi: 10.1016/j.ensm.2020.10.015 [2] Guo R, Liu X, Wen B, et al. Engineering mesoporous structure in amorphous carbon boosts potassium storage with high initial coulombic efficiency[J]. Nano-Micro Letters,2020,12(1):148. doi: 10.1007/s40820-020-00481-7 [3] Zhu C Y, Ye Y W, Guo X, et al. Design and synthesis of carbon-based nanomaterials for electrochemical energy storage[J]. New Carbon Materials,2022,37(1):59-92. doi: 10.1016/S1872-5805(22)60579-1 [4] Shaker M, Ghazvini A A S, Cao W Q, et al. Biomass-derived porous carbons as supercapacitor electrodes - A review[J]. New Carbon Materials,2021,36(3):546-572. doi: 10.1016/S1872-5805(21)60038-0 [5] Sepulveda N A, Jenkins J D, Edington A, et al. The design space for long-duration energy storage in decarbonized power systems[J]. Nature Energy,2021,6(5):506-516. doi: 10.1038/s41560-021-00796-8 [6] Chen Y X, Shi L L, Yuan Q, et al. Crystallization-induced morphological tuning toward denim-like graphene nanosheets in a KCl-copolymer solution[J]. ACS Nano,2018,12(4):4019-4024. doi: 10.1021/acsnano.8b01708 [7] Wu X, Chen Y X, Xing Z, et al. Advanced carbon‐based anodes for potassium‐ion batteries[J]. Advanced Energy Materials,2019,9(21):1900343. doi: 10.1002/aenm.201900343 [8] Wang D K, Zhang J P, Dong Y, et al. Progress on graphitic carbon materials for potassium-based energy storage[J]. New Carbon Materials,2021,36(3):435-448. doi: 10.1016/S1872-5805(21)60039-2 [9] Ju Z C, Li P Z, Ma G Y, et al. Few layer nitrogen-doped graphene with highly reversible potassium storage[J]. Energy Storage Materials,2018,11:38-46. doi: 10.1016/j.ensm.2017.09.009 [10] Chen Y X, Xi B J, Huang M, et al. Defect-selectivity and "order in disorder" engineering in carbon for durable and fast potassium storage[J]. Advanced Materials,2021,34(7):2108621. [11] Rajagopalan R, Tang Y, Ji X, et al. Advancements and challenges in potassium ion batteries: A comprehensive review[J]. Advanced Functional Materials,2020,30(12):1909486. doi: 10.1002/adfm.201909486 [12] Lei K X, Li F J, Mu C N, et al. High K-storage performance based on the synergy of dipotassium terephthalate and ether-based electrolytes[J]. Energy & Environmental Science,2017,10(2):552-557. [13] Kubota K, Dahbi M, Hosaka T, et al. Towards K-ion and Na-ion batteries as "beyond Li-ion"[J]. Chemical record (New York, N.Y.) ,2018,18(4):459-479. doi: 10.1002/tcr.201700057 [14] Zhang L P, Wang W, Lu S F, et al. Carbon anode materials: A detailed comparison between Na‐ion and K‐ion batteries[J]. Advanced Energy Materials,2021,11(11):2003640. doi: 10.1002/aenm.202003640 [15] Gao F, Zang Y H, Wang Y, et al. A review of the synthesis of carbon materials for energy storage from biomass and coal/heavy oil waste[J]. New Carbon Materials,2021,36(1):34-48. doi: 10.1016/S1872-5805(21)60003-3 [16] Tan H, Du X Q, Zhou R, et al. Rational design of microstructure and interphase enables high-capacity and long-life carbon anodes for potassium ion batteries[J]. Carbon,2021,176:383-389. doi: 10.1016/j.carbon.2021.02.003 [17] Liu S, Kang L, Zhang J, et al. Carbonaceous anode materials for non-aqueous sodium- and potassium-ion hybrid capacitors[J]. ACS Energy Letters,2021,6(11):4127-4154. doi: 10.1021/acsenergylett.1c01855 [18] Zhang J, Lai L, Wang H, et al. Energy storage mechanisms of anode materials for potassium ion batteries[J]. Materials Today Energy,2021,21:100747. doi: 10.1016/j.mtener.2021.100747 [19] Wu S, Song Y, Lu C, et al. An adsorption-insertion mechanism of potassium in soft carbon[J]. Small,2021,18(4):2105275. [20] Chen J F, Feng J M, Dong L, et al. Nanoporous coal via Ni-catalytic graphitization as anode materials for potassium ion battery[J]. Journal of Electroanalytical Chemistry,2020,862:113902. doi: 10.1016/j.jelechem.2020.113902 [21] Du J C, Gao S S, Shi P H, et al. Three-dimensional carbonaceous for potassium ion batteries anode to boost rate and cycle life performance[J]. Journal of Power Sources,2020,451:227727. doi: 10.1016/j.jpowsour.2020.227727 [22] Zhang W C, Liu Y J, Guo Z P, et al. Approaching high-performance potassium-ion batteries via advanced design strategies and engineering[J]. Science Advances,2019,5(5):eaav7412. doi: 10.1126/sciadv.aav7412 [23] Yin B, Liang S, Yu D, et al. Increasing accessible subsurface to improving rate capability and cycling stability of sodium-ion batteries[J]. Advanced Materials,2021,33(37):2100808. doi: 10.1002/adma.202100808 [24] Liu Z, Zhang L H, Sheng L Z, et al. Edge-nitrogen-rich carbon dots pillared graphene blocks with ultrahigh volumetric/gravimetric capacities and ultralong life for sodium-ion storage[J]. Advanced Energy Materials,2018,8(30):1802042. doi: 10.1002/aenm.201802042 [25] Liu L Y, Lin Z F, Chane-Ching J Y, et al. 3D rGO aerogel with superior electrochemical performance for K–ion battery[J]. Energy Storage Materials,2019,19:306-313. doi: 10.1016/j.ensm.2019.03.013 [26] An Y L, Fei H F, Zeng G F, et al. Commercial expanded graphite as a low–cost, long-cycling life anode for potassium–ion batteries with conventional carbonate electrolyte[J]. Journal of Power Sources,2018,378:66-72. doi: 10.1016/j.jpowsour.2017.12.033 [27] Sultana I, Rahman M M, Ramireddy T, et al. High capacity potassium-ion battery anodes based on black phosphorus[J]. Journal of Materials Chemistry A,2017,5(45):23506-23512. doi: 10.1039/C7TA02483E [28] Zhang S, Teck A A, Guo Z Y, et al. Carbon composite anodes with tunable microstructures for potassium‐ion batteries[J]. Batteries & Supercaps,2021,4(4):663-670. [29] Tian S, Zhang Y, Yang C H, et al. Nitrogen-doped carbon nanosheet coated multilayer graphite as stabilized anode material of potassium-ion batteries with high performances[J]. Electrochimica Acta,2021,380:138254. doi: 10.1016/j.electacta.2021.138254 [30] Xu S, Cai L, Niu P, et al. The creation of extra storage capacity in nitrogen-doped porous carbon as high-stable potassium-ion battery anodes[J]. Carbon,2021,178:256-264. doi: 10.1016/j.carbon.2021.03.039 [31] Yu F, Huang T, Zhang P P, et al. Design and synthesis of electrode materials with both battery-type and capacitive charge storage[J]. Energy Storage Materials,2019,22:235-255. doi: 10.1016/j.ensm.2019.07.023 [32] Zhang Y, Tao L, Xie C, et al. Defect engineering on electrode materials for rechargeable batteries[J]. Advanced Materials,2020,32(7):1905923. doi: 10.1002/adma.201905923 [33] Wu J, Zhang X X, Li Z, et al. Toward high‐performance capacitive potassium‐ion storage: A superior anode material from silicon carbide‐derived carbon with a well‐developed pore structure[J]. Advanced Functional Materials,2020,30(40):2004348. doi: 10.1002/adfm.202004348 [34] Chen Y X, Shi L L, Li A, et al. Capacity enhancement of porous carbon electrodes during long-term cycling in lithium-ion batteries[J]. Journal of The Electrochemical Society,2017,164(9):2000-A2006. doi: 10.1149/2.1151709jes [35] Chen Y X, Shi L L, Guo S S, et al. A general strategy towards carbon nanosheets from triblock polymers as high-rate anode materials for lithium and sodium ion batteries[J]. Journal of Materials Chemistry A,2017,5(37):19866-19874. doi: 10.1039/C7TA06453E [36] Bi H H, He X J, Yang L, et al. Interconnected carbon nanocapsules with high N/S co-doping as stable and high-capacity potassium-ion battery anode[J]. Journal of Energy Chemistry,2022,66:195-204. doi: 10.1016/j.jechem.2021.08.016 [37] Chen Y, Shi L, Li D, et al. Undercooling-directed nacl crystallization: An approach towards nanocavity-linked graphene networks for fast lithium and sodium storage[J]. Nanoscale,2020,12(14):7622-7630. doi: 10.1039/D0NR01126F [38] Guo Y Y, Feng Y F, Li H, et al. Carbon quantum dots in hard carbon: an approach to achieving PIB anodes with high potassium adsorption[J]. Carbon,2022,189(15):142-151. [39] Yang J, Ju Z, Jiang Y, et al. Enhanced capacity and rate capability of nitrogen/oxygen dual-doped hard carbon in capacitive potassium-ion storage[J]. Advanced Materials,2018,30(4):1700104. doi: 10.1002/adma.201700104 [40] Peng D Q, Chen Y X, Ma H L, et al. Enhancing the cycling stability by tuning the chemical bonding between phosphorus and carbon nanotubes for potassium-ion battery anodes[J]. ACS Applied Materials & Interfaces,2020,12(33):37275-37284. [41] Zhang W C, Lu J Guo Z P. Challenges and future perspectives on sodium and potassium ion batteries for grid-scale energy storage[J]. Materials Today,2021,50:400-417. doi: 10.1016/j.mattod.2021.03.015 [42] Wang B, Peng Y, Yuan F, et al. A comprehensive review of carbons anode for potassium-ion battery: Fast kinetic, structure stability and electrochemical[J]. Journal of Power Sources,2021,484:229244. doi: 10.1016/j.jpowsour.2020.229244 [43] Jiang Y, Yang Y, Xu R, et al. Ultrafast potassium storage in F-induced ultra-high edge-defective carbon nanosheets[J]. ACS Nano,2021,15(6):10217-10227. doi: 10.1021/acsnano.1c02275 [44] Liu H, Du H L, Zhao W, et al. Fast potassium migration in mesoporous carbon with ultrathin framework boosting superior rate performance for high-power potassium storage[J]. Energy Storage Materials,2021,40:490-498. doi: 10.1016/j.ensm.2021.05.037 [45] Jian Z, Luo W, Ji X. Carbon electrodes for K-ion batteries[J]. Journal of the American Chemical Society,2015,137(36):11566-9. doi: 10.1021/jacs.5b06809 [46] Wang B, Gu L, Yuan F, et al. Edge-enrich N-doped graphitic carbon: Boosting rate capability and cyclability for potassium ion battery[J]. Chemical Engineering Journal,2022,432(15):134321. [47] Xing Z, Qi Y, Jian Z, et al. Polynanocrystalline graphite: A new carbon anode with superior cycling performance for K-ion batteries[J]. ACS Applied Materials & Interfaces,2017,9(5):4343-4351. [48] Hosaka T, Kubota K, Kojima H, et al. Highly concentrated electrolyte solutions for 4 V class potassium-ion batteries[J]. Chemical Communications,2018,54(60):8387-8390. doi: 10.1039/C8CC04433C [49] Xu Y, Zhang C, Zhou M, et al. Highly nitrogen doped carbon nanofibers with superior rate capability and cyclability for potassium ion batteries[J]. Nature Communications,2018,9(1):1720. doi: 10.1038/s41467-018-04190-z [50] Liu C, Xiao N, Li H J, et al. Nitrogen-doped soft carbon frameworks built of well-interconnected nanocapsules enabling a superior potassium-ion batteries anode[J]. Chemical Engineering Journal,2020,382(15):121759. [51] Liu Q, Han F, Zhou J, et al. Boosting the potassium-ion storage performance in soft carbon anodes by the synergistic effect of optimized molten salt medium and N/S dual-doping[J]. ACS Applied Materials & Interfaces,2020,12(18):20838-20848. [52] Liu Y, Lu Y X, Xu Y S, et al. Pitch-derived soft carbon as stable anode material for potassium ion batteries[J]. Advanced Materials,2020,32(17):2000505. doi: 10.1002/adma.202000505 [53] Tan H, Zhou R Zhang B. Understanding potassium ion storage mechanism in pitch-derived soft carbon and the consequence on cyclic stability[J]. Journal of Power Sources,2021,506:230179. doi: 10.1016/j.jpowsour.2021.230179 [54] Wang P F, Gong Z, Ye K, et al. N-rich biomass carbon derived from hemp as a full carbon-based potassium ion hybrid capacitor anode[J]. Applied Surface Science,2021,553:149569. doi: 10.1016/j.apsusc.2021.149569 [55] Liu Y, Dai H D, Wu L, et al. A large scalable and low‐cost sulfur/nitrogen dual‐doped hard carbon as the negative electrode material for high‐performance potassium‐ion batteries[J]. Advanced Energy Materials,2019,9(34):1901379. doi: 10.1002/aenm.201901379 [56] Tao S, Xu W, Zheng J H, et al. Soybean roots-derived N, P co-doped mesoporous hard carbon for boosting sodium and potassium-ion batteries[J]. Carbon,2021,178:233-242. doi: 10.1016/j.carbon.2021.03.022 [57] Li W Z, Zhang R, Chen Z, et al. Microstructure-dependent K+ storage in porous hard carbon[J]. Small,2021,17(21):2100397. doi: 10.1002/smll.202100397 [58] Yuan F, Zhang D, Li Z, et al. Unraveling the intercorrelation between micro/mesopores and K migration behavior in hard carbon [J]. Small, 2022, 18 (12): 2107113. DOI: 10.1002/smll. 202107113. [59] Ma X Q, Xiao N, Xiao J, et al. Nitrogen and phosphorus dual-doped porous carbons for high-rate potassium ion batteries[J]. Carbon,2021,179:33-41. doi: 10.1016/j.carbon.2021.03.067 [60] Wu X, Lam C W K, Wu N Q, et al. Multiple templates fabrication of hierarchical porous carbon for enhanced rate capability in potassium-ion batteries[J]. Materials Today Energy,2019,11:182-191. doi: 10.1016/j.mtener.2018.11.009 [61] Xu Y, Ruan J, Pang Y, et al. Homologous strategy to construct high-performance coupling electrodes for advanced potassium-ion hybrid capacitors[J]. Nano-Micro Letters,2020,13(1):14. [62] Wu X, Zhao W, Wang H, et al. Enhanced capacity of chemically bonded phosphorus/carbon composite as an anode material for potassium-ion batteries[J]. Journal of Power Sources,2018,378:460-467. doi: 10.1016/j.jpowsour.2017.12.077 [63] Wang M Y, Zhu Y Y, Zhang Y, et al. Cost-effective hard–soft carbon composite anodes with promising potassium ions storage performance[J]. Electrochimica Acta,2021,368:137649. doi: 10.1016/j.electacta.2020.137649 [64] Cao B, Zhang Q, Liu H, et al. Graphitic carbon nanocage as a stable and high power anode for potassium-ion batteries[J]. Advanced Energy Materials,2021,8(25):1801149. [65] Tai Z X, Zhang Q, Liu Y J, et al. Activated carbon from the graphite with increased rate capability for the potassium ion battery[J]. Carbon,2017,123:54-61. doi: 10.1016/j.carbon.2017.07.041 [66] Liu Z, Wang J, Jia X, et al. Graphene armored with a crystal carbon shell for ultrahigh-performance potassium ion batteries and aluminum batteries[J]. ACS Nano,2019,13(9):10631-10642. doi: 10.1021/acsnano.9b04893 [67] Lin X Y, Huang J Q, Zhang B. Correlation between the microstructure of carbon materials and their potassium ion storage performance[J]. Carbon,2019,143:138-146. doi: 10.1016/j.carbon.2018.11.001 [68] Zeng S, Zhou X F, Wang B, et al. Freestanding CNT-modified graphitic carbon foam as a flexible anode for potassium ion batteries[J]. Journal of Materials Chemistry A,2019,7(26):15774-15781. doi: 10.1039/C9TA03245B [69] Fan L, Ma R, Zhang Q, et al. Graphite anode for a potassium-ion battery with unprecedented performance[J]. Angewandte Chemie International Edition,2019,58(31):10500-10505. doi: 10.1002/anie.201904258 [70] Wang H H, Yang G, Chen Z, et al. Nitrogen configuration dependent holey active sites toward enhanced K+ storage in graphite foam[J]. Journal of Power Sources,2019,419:82-90. doi: 10.1016/j.jpowsour.2019.02.029 [71] Shen Y P, Huang C, Li Y H, et al. Enhanced sodium and potassium ions storage of soft carbon by a S/O co-doped strategy[J]. Electrochimica Acta,2021,367:137526. doi: 10.1016/j.electacta.2020.137526 [72] Ou M Y, Zhang Y, Zhu Y, et al. Local structures of soft carbon and electrochemical performance of potassium-ion batteries[J]. ACS Applied Materials & Interfaces,2021,13(24):28261-28269. [73] Kubota K, Shimadzu S, Yabuuchi N, et al. Structural analysis of sucrose-derived hard carbon and correlation with the electrochemical properties for lithium, sodium, and potassium insertion[J]. Chemistry of Materials,2020,32(7):2961-2977. doi: 10.1021/acs.chemmater.9b05235 [74] Chen C, Wu M Q, Wang Y S, et al. Insights into pseudographite-structured hard carbon with stabilized performance for high energy K-ion storage[J]. Journal of Power Sources,2019,444:227310. doi: 10.1016/j.jpowsour.2019.227310 [75] Alvin S, Cahyadi H S, Hwang J, et al. Revealing the intercalation mechanisms of lithium, sodium, and potassium in hard carbon[J]. Advanced Energy Materials,2020,10(20):2000283. doi: 10.1002/aenm.202000283 [76] Wang B, Zhang Z Y, Yuan F, et al. An insight into the initial coulombic efficiency of carbon-based anode materials for potassium-ion batteries[J]. Chemical Engineering Journal,2022,428:131093. doi: 10.1016/j.cej.2021.131093 [77] Zhang H H, Luo C, He H N, et al. Nano-size porous carbon spheres as a high-capacity anode with high initial coulombic efficiency for potassium-ion batteries[J]. Nanoscale Horizons,2020,5(5):895-903. doi: 10.1039/D0NH00018C [78] Xu Y S, Duan S Y, Sun Y G, et al. Recent developments in electrode materials for potassium-ion batteries[J]. Journal of Materials Chemistry A,2019,7(9):4334-4352. doi: 10.1039/C8TA10953B [79] Chen J C, Xiao G C, Duan G G, et al. Structural design of carbon dots/porous materials composites and their applications[J]. Chemical Engineering Journal,2021,421(1):127743. [80] Zhu J W, Mu S C. Defect engineering in carbon‐based electrocatalysts: Insight into intrinsic carbon defects[J]. Advanced Functional Materials,2020,30(25):2001097. doi: 10.1002/adfm.202001097 [81] Li J P, Li Y J, Ma X D, et al. A honeycomb-like nitrogen-doped carbon as high-performance anode for potassium-ion batteries[J]. Chemical Engineering Journal,2020,384(15):123328. [82] Liu S T, Yang B B, Zhou J S, et al. Nitrogen-rich carbon-onion-constructed nanosheets: an ultrafast and ultrastable dual anode material for sodium and potassium storage[J]. Journal of Materials Chemistry A,2019,7(31):18499-18509. doi: 10.1039/C9TA04699B [83] Benzigar M R, Talapaneni S N, Joseph S, et al. Recent advances in functionalized micro and mesoporous carbon materials: synthesis and applications[J]. Chemical Society Reviews,2018,47(8):2680-2721. doi: 10.1039/C7CS00787F [84] Lee J, Oh J, Jeon Y, et al. Multi-heteroatom-doped hollow carbon attached on graphene using LiFePO4 nanoparticles as hard templates for high-performance lithium-sulfur batteries[J]. ACS Applied Materials & Interfaces,2018,10(31):26485-26493. [85] Wang W K, Zhao W W, Chen T T, et al. All‐in‐one hollow flower‐like covalent organic frameworks for flexible transparent devices[J]. Advanced Functional Materials,2021,31(29):2010306. doi: 10.1002/adfm.202010306 [86] Qiu D P, Guan J Y, Li M, et al. Kinetics enhanced nitrogen‐doped hierarchical porous hollow carbon spheres boosting advanced potassium‐ion hybrid capacitors[J]. Advanced Functional Materials,2019,29(32):1903496. doi: 10.1002/adfm.201903496 [87] Tao X S, Sun Y G, Liu Y, et al. Facile synthesis of hollow carbon nanospheres and their potential as stable anode materials in potassium-ion batteries[J]. ACS Applied Materials & Interfaces,2020,12(11):13182-13188. [88] Lin Q, Zhang J, Lv W, et al. A functionalized carbon surface for high-performance sodium-ion storage[J]. Small,2020,16(15):1902603. doi: 10.1002/smll.201902603 [89] Share K, Cohn A P, Carter R, et al. Role of nitrogen-doped graphene for improved high-capacity potassium ion battery anodes[J]. ACS Nano,2016,10(10):9738-9744. doi: 10.1021/acsnano.6b05998 [90] Shen W, Wang C, Xu Q J, et al. Nitrogen-doping-induced defects of a carbon coating layer facilitate Na-storage in electrode materials[J]. Advanced Energy Materials,2015,5(1):1400982. doi: 10.1002/aenm.201400982 [91] Zhang W L, Sun M L, Yin J, et al. Accordion-like carbon with high nitrogen doping for fastand stable K ion storage[J]. Advanced Energy Materials,2021,11(41):2101928. doi: 10.1002/aenm.202101928 [92] Tian K, Wang J, Cao L, et al. Single-site pyrrolic-nitrogen-doped sp2-hybridized carbon materials and their pseudocapacitance[J]. Nature Communications,2020,11(1):3884. doi: 10.1038/s41467-020-17727-y [93] Li J L, Qin W, Xie J P, et al. Sulphur-doped reduced graphene oxide sponges as high-performance free-standing anodes for K-ion storage[J]. Nano Energy,2018,53:415-424. doi: 10.1016/j.nanoen.2018.08.075 [94] Chen W M, Wan M, Liu Q, et al. Heteroatom‐doped carbon materials: synthesis, mechanism, and application for sodium‐ion batteries[J]. Small Methods,2018,3(4):1800323. [95] Cui R C, Xu B, Dong H J, et al. N/O dual-doped environment-friendly hard carbon as advanced anode for potassium-ion batteries[J]. Advanced Science,2020,7(5):1902547. doi: 10.1002/advs.201902547 [96] Tao L, Yang Y P, Wang H L, et al. Sulfur-nitrogen rich carbon as stable high capacity potassium ion battery anode: Performance and storage mechanisms[J]. Energy Storage Materials,2020,27:212-225. doi: 10.1016/j.ensm.2020.02.004 [97] Wang T D, Li Q, Feng Q T, et al. Carbon defects applied to potassium-ion batteries: A density functional theory investigation[J]. Nanoscale,2021,13(32):13719-13734. doi: 10.1039/D1NR03604A [98] Dong Y, Lin X J, Wang D K, et al. Modulating the defects of graphene blocks by ball-milling for ultrahigh gravimetric and volumetric performance and fast sodium storage[J]. Energy Storage Materials,2020,30:287-295. doi: 10.1016/j.ensm.2020.05.016 [99] Dong Y, Zhang S, Du X, et al. Boosting the electrical double‐layer capacitance of graphene by self‐doped defects through ball‐milling[J]. Advanced Functional Materials,2019,29(24):1901127. doi: 10.1002/adfm.201901127 [100] Yuan R, Dong Y, Zhang S, et al. Efficient utilization of the active sites in defective graphene blocks through functionalization synergy for compact capacitive energy storage[J]. ACS Applied Materials & Interfaces,2021,13(48):57092-57099. [101] Yang Z, Ren X, Song Y, et al. Germanium‐carbdiyne: A 3D well‐defined sp‐hybridized carbon‐based material with superhigh Li storage property[J]. Energy & Environmental Materials,2022(0):1-8. [102] Li G, Li Y, Liu H, et al. Architecture of graphdiyne nanoscale films[J]. Chemical Communications,2010,46(19):3256-3258. doi: 10.1039/b922733d [103] Yi Y Y, Li J Q, Zhao W, et al. Temperature‐mediated engineering of graphdiyne framework enabling high‐performance potassium storage[J]. Advanced Functional Materials,2020,30(31):2003039. doi: 10.1002/adfm.202003039 [104] Meng Y T, Nie C H, Guo W J, et al. Inorganic cathode materials for potassium ion batteries[J]. Materials Today Energy,2022, 25:100982. [105] Liu S, Kang L, Jun S C. Challenges and strategies toward cathode materials for rechargeable potassium-ion batteries[J]. Advanced Materials,2021,33(47):2004689. doi: 10.1002/adma.202004689 [106] Qin L, Xiao N, Zheng J F, et al. Localized high‐concentration electrolytes boost potassium storage in high‐loading graphite[J]. Advanced Energy Materials,2019,9(44):1902618. doi: 10.1002/aenm.201902618 [107] Ji B, Zhang F, Song X, et al. A novel potassium-ion-based dual-ion battery[J]. Advanced Materials,2017,29(19):1700519. doi: 10.1002/adma.201700519 [108] Fan L, Liu Q, Chen S, et al. Potassium-based dual ion battery with dual-graphite electrode[J]. Small,2017,13(30):1701011. doi: 10.1002/smll.201701011 [109] Beltrop K, Beuker S, Heckmann A, et al. Alternative electrochemical energy storage: Potassium-based dual-graphite batteries[J]. Energy & Environmental Science,2017,10(10):2090-2094. [110] Ding X, Zhang F, Ji B, et al. Potassium dual-ion hybrid batteries with ultrahigh rate performance and excellent cycling stability[J]. ACS Applied Materials & Interfaces,2018,10(49):42294-42300. [111] Liu X, Elia G A, Qin B S, et al. High-power Na-ion and K-ion hybrid capacitors exploiting cointercalation in graphite negative electrodes[J]. ACS Energy Letters,2019,4(11):2675-2682. doi: 10.1021/acsenergylett.9b01675 [112] Fan L, Lin K, Wang J, et al. A nonaqueous potassium-based battery-supercapacitor hybrid device[J]. Advanced Materials,2018,30(20):1800804. doi: 10.1002/adma.201800804 [113] Zhang C, Liu X, Li Z, et al. Nitrogen‐doped accordion‐like soft carbon anodes with exposed hierarchical pores for advanced potassium‐ion hybrid capacitors[J]. Advanced Functional Materials,2021,31(23):2101470. doi: 10.1002/adfm.202101470 [114] Wang D K, Zhang J P, Li X T, et al. Woven microsphere architected by carbon nanotubes as high-performance potassium ion batteries anodes[J]. Chemical Engineering Journal,2022,429:132272. doi: 10.1016/j.cej.2021.132272 -






 下载:
下载: