Optimizing oxygen substituents of a carbon cathode for improved capacitive behavior in ethanol-based zinc-ion capacitors
-
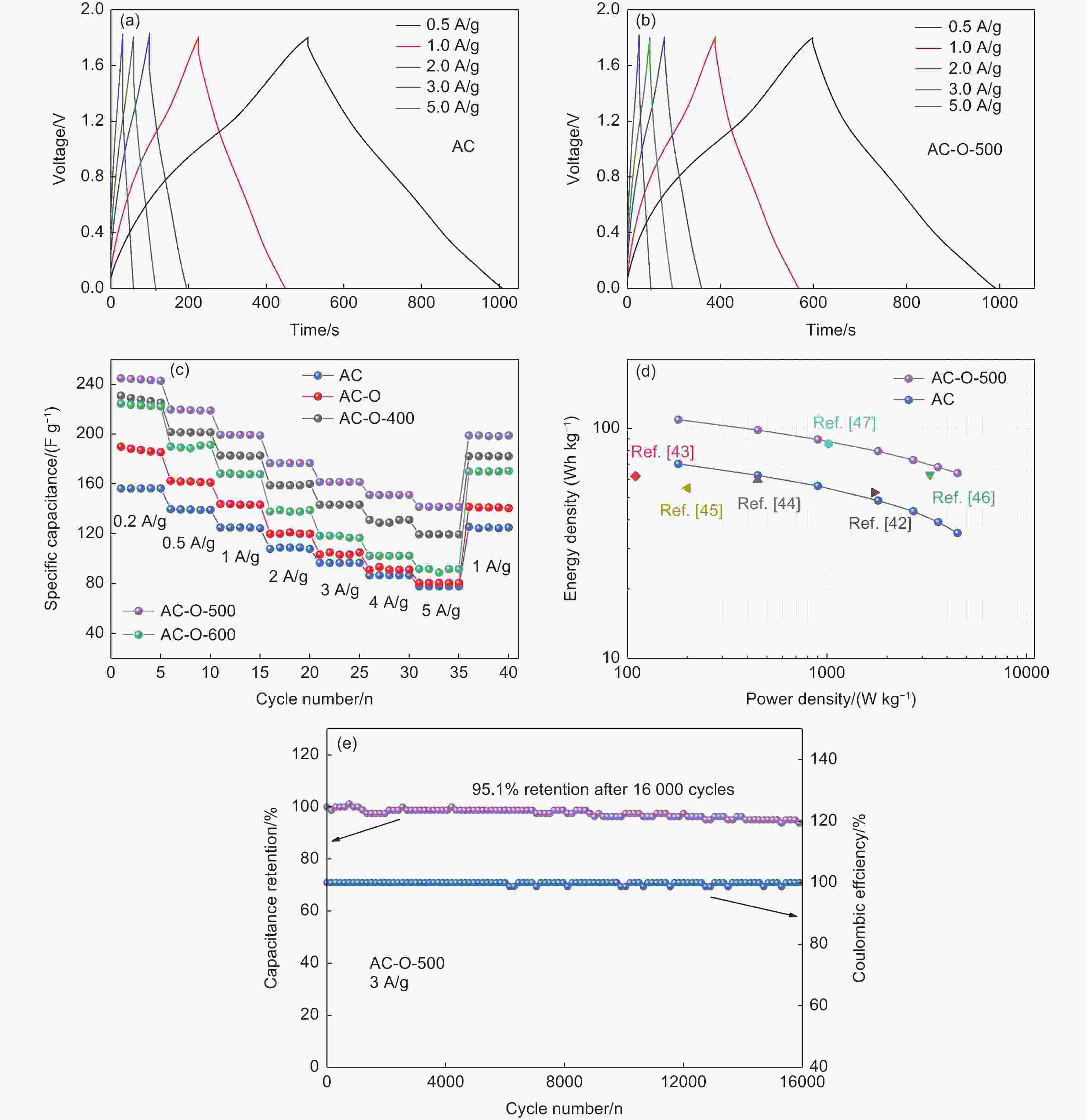
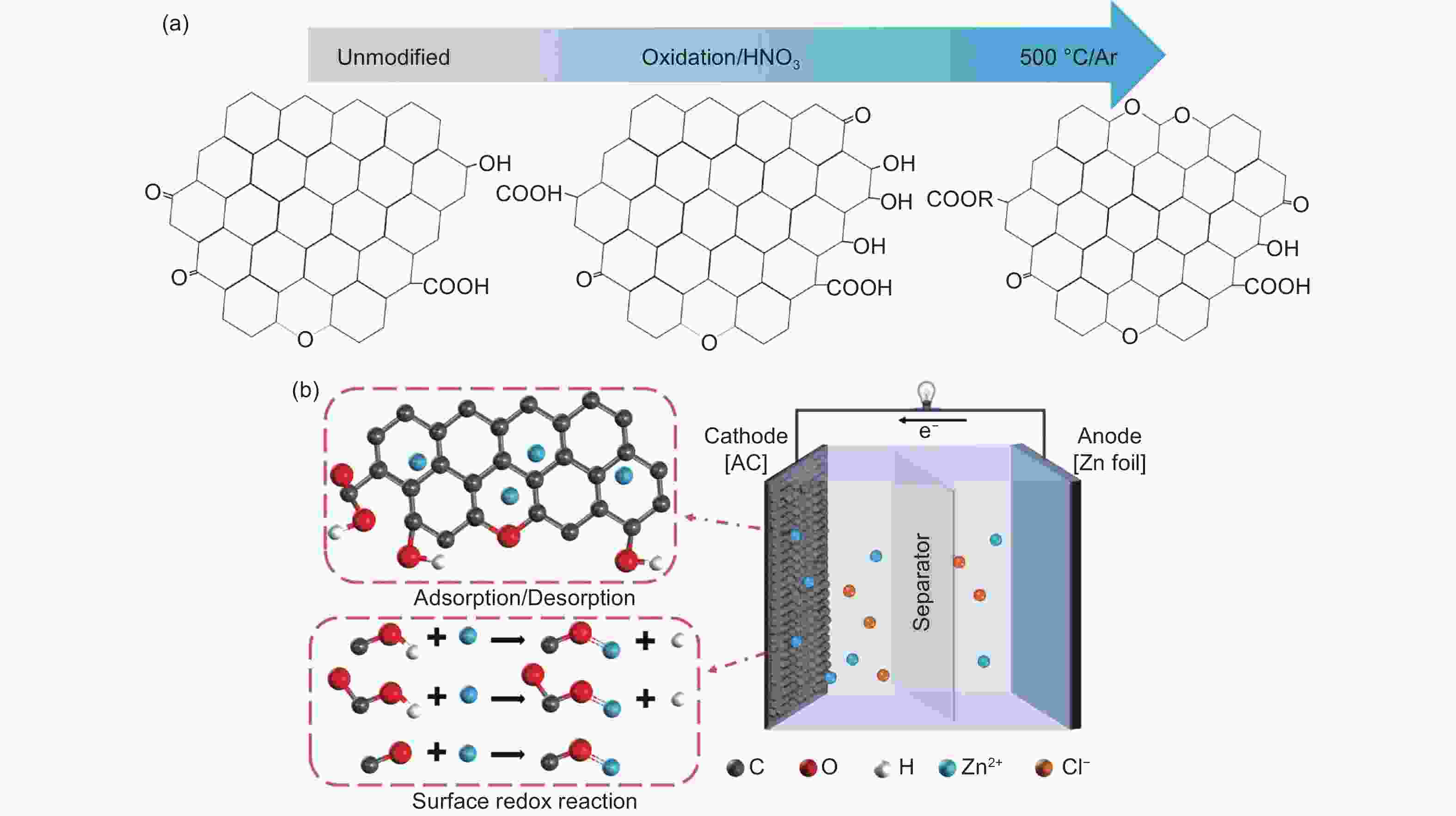
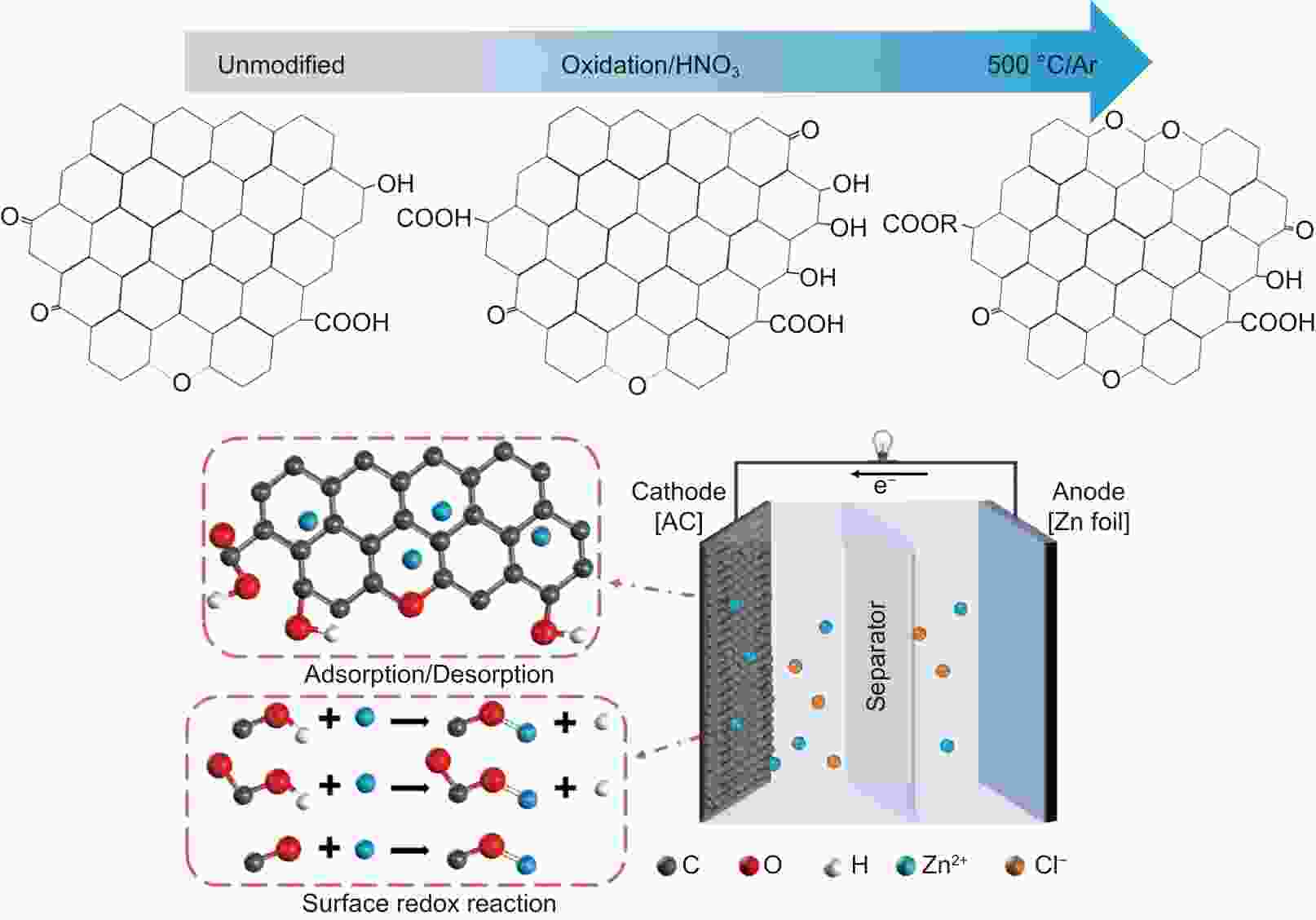
摘要: 锌离子电容器(ZICs)具有能量密度高、倍率性能好、循环寿命长、成本低等优点,近年来得到了广泛的研究。在碳基阴极表面引入氧官能团是提高水系ZICs电容性能的有效策略。然而,氧官能团的存在是否有利于提高乙醇(EtOH)基ZICs的电容性能,目前还没有被深入研究。本文采用硝酸氧化和进一步热处理的方法对阴极活性炭表面的氧官能团进行了优化。在ZnCl2/EtOH电解液中,优化后的样品在电流密度为1 A g−1时比电容达到195 F g−1,比未改性的样品 (125 F g−1)提高了56%。同时,ZICs也表现出良好的循环稳定性,在3 A g−1下的稳定循环次数超过16000次,并且保持100%的库仑效率。这是因为氧官能团,特别是羧基和酯基(―COO)的存在,为Zn2+氧化还原反应提供了丰富的电化学活性位点。因此,本研究通过优化氧官能团增强了炭阴极的电容性能,并为EtOH基ZICs的商业应用提供了研究基础。Abstract: Zinc ion capacitors (ZICs) have been widely studied in recent years due to their high energy density, excellent rate capability, long cycling life and low cost. The incorporation of oxygen functional groups (OFGs) on the surface of the carbon-based cathodes is an effective strategy for improving the capacitive performance of aqueous ZICs. However, whether their presence helps improve the capacitance of ethanol (EtOH)-based ZICs has not been investigated. In this work, a combination of nitric acid oxidation and thermal treatment was used to regulate the OFGs on the activated surface of the carbon cathode. The optimized sample had a high specific capacitance of 195 F g−1 at 1 A g−1 using ZnCl2/EtOH as the electrolyte, i.e., a 56% increase compared to an unmodified cathode (125 F g−1). ZICs also shown excellent stability for more than 16 000 cycles at 3 A g−1, while maintaining 100% coulombic efficiency. This significantly improved performance is attributed to the presence of OFGs, especially carboxyl and ester groups, which provide abundant electrochemical active sites for redox reaction with the zinc ions. This study reports a significant improvement in the specific capacitance of carbon cathodes for commercial EtOH-based ZIC systems.
-
Key words:
- Zinc-ion capacitors /
- Oxygen functional groups /
- Ethanol /
- Activated carbon /
- Specific capacitance
-
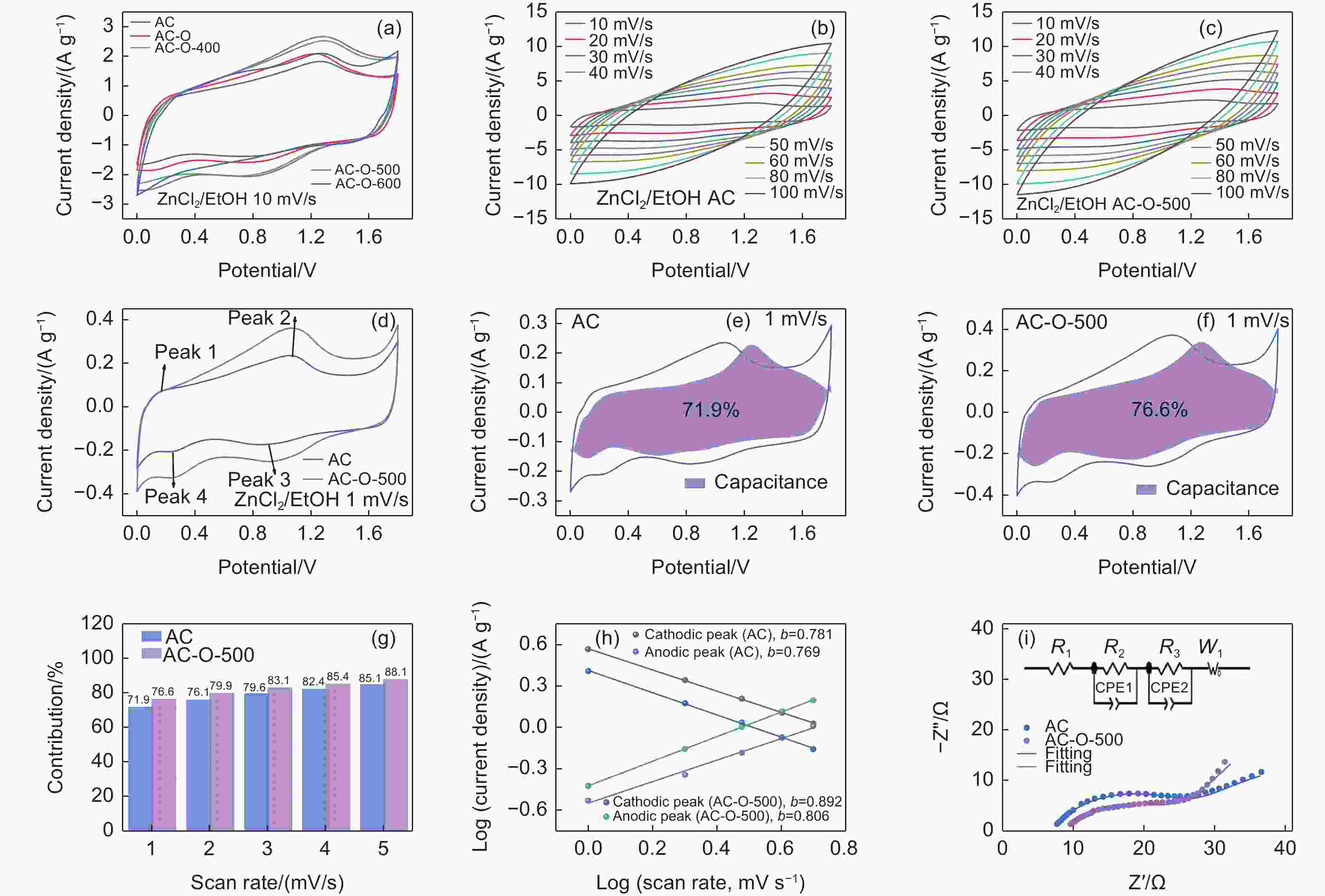
Figure 3. (a) CV curves at different scan rates for (b) AC and (c) AC-O-500, (d) CV comparison of AC and AC-O-500 at 1 mV s−1, (e, f) contribution ratio of the capacitive capacities, (g) contribution ratios, (h) b values in both the charge and discharge processes of AC and AC-O-500, (i) nyquist plots of AC and AC-O-500
Table 1. Structural parameters of AC, AC-O, AC-O-400, AC-O-500 and AC-O-600
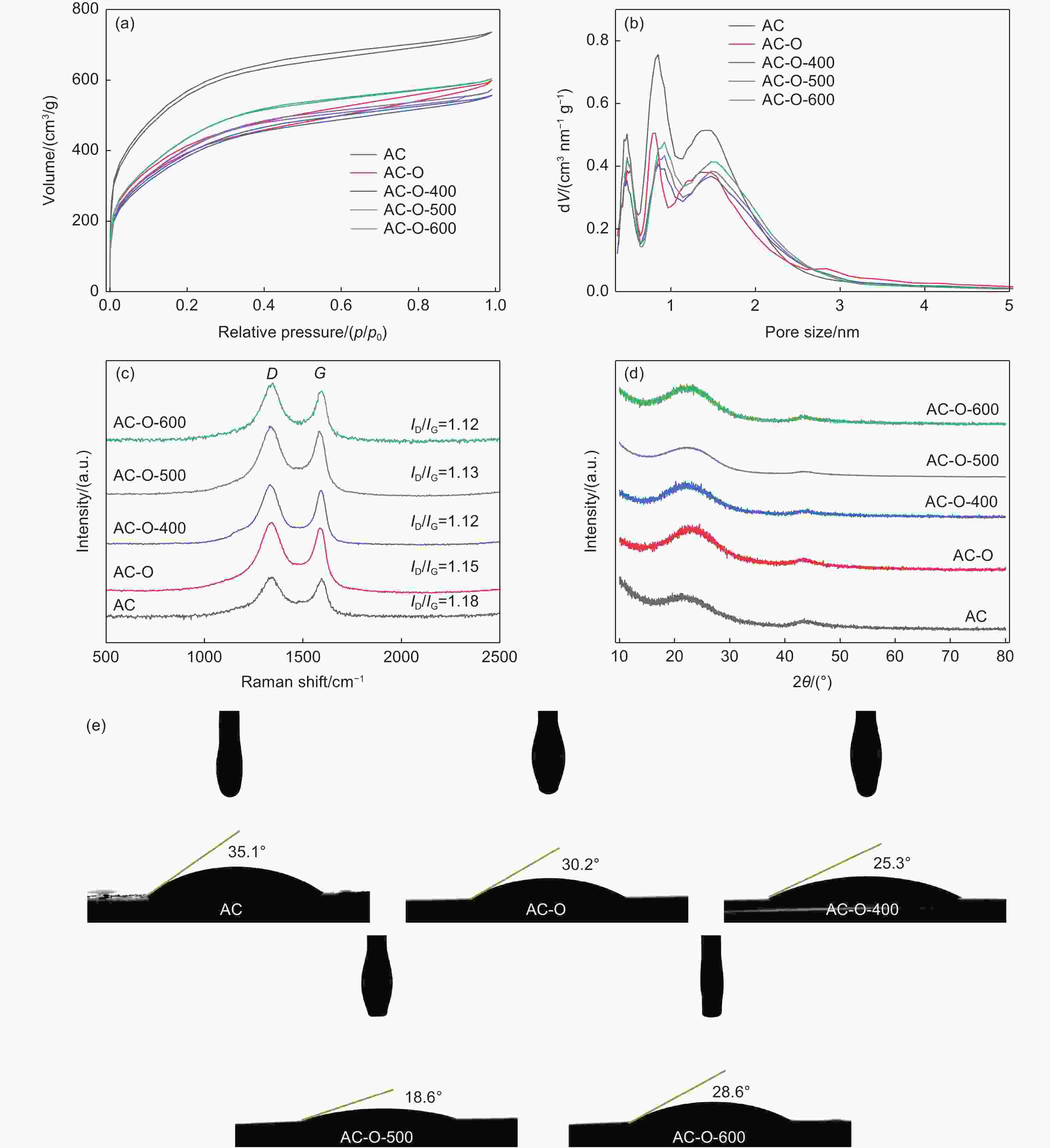
Samples SBETa/(m2 g−1) SMicrob/(m2 g−1) VTotalc/(cm3 g−1) VMicrod/(cm3 g−1) AC 1751.16 1500.97 0.904 0.734 AC-O 1240.78 1093.44 0.734 0.521 AC-O-400 1221.51 1001.25 0.684 0.498 AC-O-500 1286.62 1020.91 0.699 0.519 AC-O-600 1386.31 1086.03 0.741 0.558 Note: a-BET (Brunauer-Emmett-Teller) surface area. b-Micropore specific surface area obtained from the QSDFT method. c-Single-point total pore volume at p/p0 = 0.995. d-Micropore volume obtained from the QSDFT method. Table 2. Analysis of the fitted O 1s peaks of the OFGs on the AC samples from XPS spectra
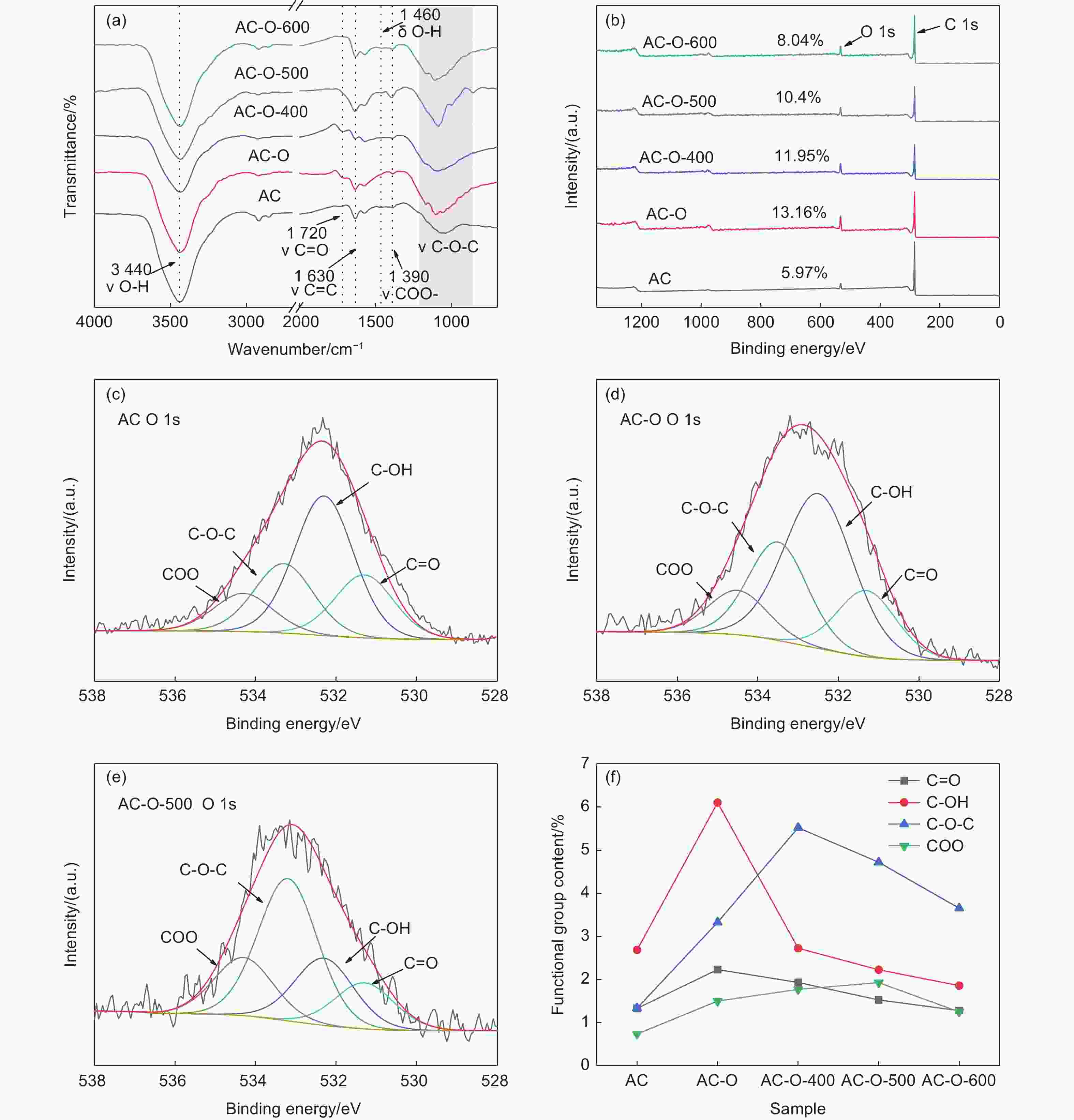
Samples C=O (531.3 eV)/% OH (532.3 eV)/% C―O―C (533.3 eV)/% COO (534.3 eV)/% AC 1.33 2.69 1.34 0.74 AC-O 2.23 6.10 3.33 1.50 AC-O-400 1.93 2.73 5.52 1.77 AC-O-500 1.53 2.23 4.72 1.93 AC-O-600 1.28 1.86 3.65 1.25 -
[1] Luo X Y, Chen S R, Hu T Z, et al. Renewable biomass-derived carbons for electrochemical capacitor applications[J]. SusMat,2021,1(2):211-240. doi: 10.1002/sus2.8 [2] Zhai Y P, Dou Y Q, Zhao D Y, et al. Carbon materials for chemical capacitive energy storage[J]. Advanced Materials,2011,23(42):4828-4850. doi: 10.1002/adma.201100984 [3] Larcher D, Tarascon J M. Towards greener and more sustainable batteries for electrical energy storage[J]. Nature Chemistry,2015,7(1):19-29. doi: 10.1038/nchem.2085 [4] Tang H, Yao J J, Zhu Y R. Recent developments and future prospects for zinc-ion hybrid capacitors: a review[J]. Advanced Energy Materials,2021,11(14):2003994. doi: 10.1002/aenm.202003994 [5] Wang H, Ye W, Yang Y, et al. Zn-ion hybrid supercapacitors: Achievements, challenges and future perspectives[J]. Nano Energy,2021,85:105942. doi: 10.1016/j.nanoen.2021.105942 [6] Li Z, Gadipelli S, Li H, et al. Tuning the interlayer spacing of graphene laminate films for efficient pore utilization towards compact capacitive energy storage[J]. Nature Energy,2020,5(2):160-168. doi: 10.1038/s41560-020-0560-6 [7] Dutta S, Bhaumik A, Wu K C W. Hierarchically porous carbon derived from polymers and biomass: effect of interconnected pores on energy applications[J]. Energy & Environmental Science,2014,7(11):3574-3592. [8] Zhi J, Zhao W, Liu X Y, et al. Highly conductive ordered mesoporous carbon based electrodes decorated by 3D graphene and 1D silver nanowire for flexible supercapacitor[J]. Advanced Functional Materials,2014,24(14):2013-2019. doi: 10.1002/adfm.201303082 [9] Zou K, Cai P, Wang B, et al. Insights into enhanced capacitive behavior of carbon cathode for lithium ion capacitors: The coupling of pore size and graphitization engineering[J]. Nanomicro Letters,2020,12(1):121. [10] Liu Q Y, Zhang H Z, Xie J H, et al. Recent progress and challenges of carbon materials for Zn-ion hybrid supercapacitors[J]. Carbon Energy,2020,2(4):521-539. doi: 10.1002/cey2.69 [11] Jin J, Geng X, Chen Q, et al. A better Zn-ion storage device: recent progress for Zn-ion hybrid supercapacitors[J]. Nanomicro Letters,2022,14(1):64. [12] Ma X P, Cheng J Y, Dong L B, et al. Multivalent ion storage towards high-performance aqueous zinc-ion hybrid supercapacitors[J]. Energy Storage Materials,2019,20:335-342. doi: 10.1016/j.ensm.2018.10.020 [13] Han P X, Xu G J, Han X Q, et al. Lithium ion capacitors in organic electrolyte system: scientific problems, material development, and key technologies[J]. Advanced Energy Materials,2018,8(26):1801243. doi: 10.1002/aenm.201801243 [14] Dong L B, Yang W, Yang W, et al. Multivalent metal ion hybrid capacitors: A review with a focus on zinc-ion hybrid capacitors[J]. Journal of Materials Chemistry A,2019,7(23):13810-13832. doi: 10.1039/C9TA02678A [15] Wu S L, Chen Y T, Jiao T P, et al. An aqueous Zn-ion hybrid supercapacitor with high energy density and ultrastability up to 80000 cycles[J]. Advanced Energy Materials,2019,9(47):1902915. doi: 10.1002/aenm.201902915 [16] Wang F, Borodin O, Gao T, et al. Highly reversible zinc metal anode for aqueous batteries[J]. Nature Materials,2018,17(6):543-549. doi: 10.1038/s41563-018-0063-z [17] Hao J N, Li X L, Zeng X H, et al. Deeply understanding the Zn anode behaviour and corresponding improvement strategies in different aqueous Zn-based batteries[J]. Energy & Environmental Science,2020,13(11):3917-3949. [18] Yuan L B, Hao J N, Kao C C, et al. Regulation methods for the Zn/electrolyte interphase and the effectiveness evaluation in aqueous Zn-ion batteries[J]. Energy & Environmental Science,2021,14(11):5669-5689. [19] He Y, Zhuang X, Lei C, et al. Porous carbon nanosheets: Synthetic strategies and electrochemical energy related applications[J]. Nano Today,2019,24:103-119. doi: 10.1016/j.nantod.2018.12.004 [20] Luo X Y, Chen Y, Mo Y. A review of charge storage in porous carbon-based supercapacitors[J]. New Carbon Materials,2021,36(1):49-68. doi: 10.1016/S1872-5805(21)60004-5 [21] Ao W, Fu J, Mao X, et al. Microwave assisted preparation of activated carbon from biomass: A review[J]. Renewable and Sustainable Energy Reviews,2018,92:958-979. doi: 10.1016/j.rser.2018.04.051 [22] Luo X Y, Zheng H, Lai W D, et al. Defect engineering of carbons for energy conversion and storage applications[J]. Energy & Environmental Materials,2023,6:1-22. doi: 10.1002/eem2.12402 [23] Zheng Y W, Zhao W, Jia D D, et al. Porous carbon prepared via combustion and acid treatment as flexible zinc-ion capacitor electrode material[J]. Chemical Engineering Journal,2020,387:124161. doi: 10.1016/j.cej.2020.124161 [24] Wang Y G, Xia Y Y. Recent progress in supercapacitors: from materials design to system construction[J]. Advanced Materials,2013,25(37):5336-5342. doi: 10.1002/adma.201301932 [25] Tang X N, Liu C Z, Chen X R, et al. Graphene aerogel derived by purification-free graphite oxide for high performance supercapacitor electrodes[J]. Carbon,2019,146:147-154. doi: 10.1016/j.carbon.2019.01.096 [26] Shao Y Y, Sun Z T, Tian Z N, et al. Regulating oxygen substituents with optimized redox activity in chemically reduced graphene oxide for aqueous Zn‐ion hybrid capacitor[J]. Advanced Functional Materials,2020,31(6):2007843. [27] Lin Z Y, Liu Y, Yao Y G, et al. Superior capacitance of functionalized graphene[J]. The Journal of Physical Chemistry C,2011,115(14):7120-7125. doi: 10.1021/jp2007073 [28] Hu P Y, Luo X Y, Hu T Z, et al. Ethanol solvent used in constructing ultra-low-temperature zinc ion capacitors with a long cycling life[J]. ACS Applied Materials & Interfaces,2023,15:5180-5190. [29] Sevilla M, Mokaya R. Energy storage applications of activated carbons: supercapacitors and hydrogen storage[J]. Energy & Environmental Science,2014,7(4):1250-1280. doi: 10.1039/c3ee43525c [30] Wu J P, Liu R R, Li M, et al. Boosting effects of hydroxyl groups on porous carbon for improved aqueous zinc-ion capacitors[J]. Journal of Energy Storage,2022,48:103996. doi: 10.1016/j.est.2022.103996 [31] Raymundo-Piñero E, Kierzek K, Machnikowski J, et al. Relationship between the nanoporous texture of activated carbons and their capacitance properties in different electrolytes[J]. Carbon,2006,44(12):2498-2507. doi: 10.1016/j.carbon.2006.05.022 [32] Shi X, Zhang H Z, Zeng S Q S, et al. Pyrrolic-dominated nitrogen redox enhances reaction kinetics of pitch-derived carbon materials in aqueous zinc ion hybrid supercapacitors[J]. ACS Materials Letters,2021,3(9):1291-1299. doi: 10.1021/acsmaterialslett.1c00325 [33] Zhang X R, Yang S, Jiang Y H, et al. Multi-dimensional graded electrodes with enhanced capacitance and superior cyclic stability[J]. Journal of Power Sources,2021,481:228911. doi: 10.1016/j.jpowsour.2020.228911 [34] Li X R, Jiang Y H, Wang P Z, et al. Effect of the oxygen functional groups of activated carbon on its electrochemical performance for supercapacitors[J]. New Carbon Materials,2020,35(3):232-243. doi: 10.1016/S1872-5805(20)60487-5 [35] Li J X, Han K H, Wang D, et al. Fabrication of high performance structural N-doped hierarchical porous carbon for supercapacitors[J]. Carbon,2020,164:42-50. doi: 10.1016/j.carbon.2020.03.044 [36] Zhong H M, Wang T T, Chen Y, et al. Understanding alkaline hydrogen evolution promoted by mesopores in three-dimensional graphene-like materials from perspective of capacitance effects[J]. Carbon,2022,199:13-22. doi: 10.1016/j.carbon.2022.07.054 [37] Zhang H Z, Liu Q Y, Fang Y B, et al. Boosting Zn-ion energy storage capability of hierarchically porous carbon by promoting chemical adsorption[J]. Advanced Materials,2019,31(44):1904948. doi: 10.1002/adma.201904948 [38] Li Y, Yang W, Yang W, et al. Towards high-energy and anti-self-discharge Zn-ion hybrid supercapacitors with new understanding of the electrochemistry[J]. Nanomicro Letters,2021,13(1):95. [39] Jiang Y Q, Liu J P. Definitions of pseudocapacitive materials: A brief review[J]. Energy & Environmental Materials,2019,2(1):30-37. [40] Yin J, Zhang W L, Wang W X, et al. Electrochemical zinc ion capacitors enhanced by redox reactions of porous carbon cathodes[J]. Advanced Energy Materials,2020,10(37):2001705. doi: 10.1002/aenm.202001705 [41] Yang B J, Chen J T, Liu L Y, et al. 3D nitrogen-doped framework carbon for high-performance potassium ion hybrid capacitor[J]. Energy Storage Materials,2019,23:522-529. doi: 10.1016/j.ensm.2019.04.008 [42] Huo S, Zhao Y, Zong M, et al. Boosting supercapacitor and capacitive deionization performance of hierarchically porous carbon by polar surface and structural engineering[J]. Journal of Materials Chemistry A,2020,8(5):2505-2517. doi: 10.1039/C9TA12170F [43] Wang H, Wang M, Tang Y B. A novel zinc-ion hybrid supercapacitor for long-life and low-cost energy storage applications[J]. Energy Storage Materials,2018,13:1-7. doi: 10.1016/j.ensm.2017.12.022 [44] Zhang H Z, Fang Y B, Yang F, et al. Aromatic organic molecular crystal with enhanced π–π stacking interaction for ultrafast Zn-ion storage[J]. Energy & Environmental Science,2020,13(8):2515-2523. [45] Chen S M, Ma L T, Zhang K, et al. A flexible solid-state zinc ion hybrid supercapacitor based on co-polymer derived hollow carbon spheres[J]. Journal of Materials Chemistry A,2019,7(13):7784-7790. doi: 10.1039/C9TA00733D [46] Dong S Y, Shen L F, Li H S, et al. Flexible sodium-ion pseudocapacitors based on 3D Na2Ti3O7 nanosheet arrays/carbon textiles anodes[J]. Advanced Functional Materials,2016,26(21):3703-3710. doi: 10.1002/adfm.201600264 [47] Dong X L, Chen L, Su X L, et al. Flexible aqueous lithium-ion battery with high safety and large volumetric energy density[J]. Angewandte Chemie International Edition,2016,55(26):7474-7477. doi: 10.1002/anie.201602766 [48] Zeng Y X, Lin Z Q, Meng Y, et al. Flexible ultrafast aqueous rechargeable Ni//Bi battery based on highly durable single-crystalline bismuth nanostructured anode[J]. Advanced Materials,2016,28(41):9188-9195. doi: 10.1002/adma.201603304 [49] Wang Z Q, Zhou M, Qin L P, et al. Simultaneous regulation of cations and anions in an electrolyte for high-capacity, high-stability aqueous zinc-vanadium batteries[J]. eScience,2022,2(2):209-218. doi: 10.1016/j.esci.2022.03.002 [50] Xiong D B, Li X F, Shan H, et al. Controllable oxygenic functional groups of metal-free cathodes for high performance lithium ion batteries[J]. Journal of Materials Chemistry A,2015,3(21):11376-11386. doi: 10.1039/C5TA01574J [51] Yuan S T, Huang X H, Wang H, et al. Structure evolution of oxygen removal from porous carbon for optimizing supercapacitor performance[J]. Journal of Energy Chemistry,2020,51:396-404. doi: 10.1016/j.jechem.2020.04.004 [52] Phattharasupakun N, Wutthiprom J, Suktha P, et al. High-performance supercapacitors of carboxylate-modified hollow carbon nanospheres coated on flexible carbon fibre paper: Effects of oxygen-containing group contents, electrolytes and operating temperature[J]. Electrochimica Acta,2017,238:64-73. doi: 10.1016/j.electacta.2017.03.208 [53] Ternero-Hidalgo J J, Rosas J M, Palomo J, et al. Functionalization of activated carbons by HNO3 treatment: Influence of phosphorus surface groups[J]. Carbon,2016,101:409-419. doi: 10.1016/j.carbon.2016.02.015 [54] Wang G M, Wang H Y, Lu X H, et al. Solid-state supercapacitor based on activated carbon cloths exhibits excellent rate capability[J]. Advanced Materials,2014,26(17):2676-2682. doi: 10.1002/adma.201304756 [55] Liu C, Wu J C, Zhou H, et al. Great enhancement of carbon energy storage through narrow pores and hydrogen-containing functional groups for aqueous Zn-ion hybrid supercapacitor[J]. Molecules,2019,24(14):2589. doi: 10.3390/molecules24142589 [56] Ha S H, Jeong Y S, Lee Y J. Free standing reduced graphene oxide film cathodes for lithium ion batteries[J]. ACS Applied Materials & Interfaces,2013,5(23):12295-12303. [57] Zou K Y, Cai P, Deng X L, et al. Highly stable zinc metal anode enabled by oxygen functional groups for advanced Zn-ion supercapacitors[J]. Chemical Communications,2021,57(4):528-531. doi: 10.1039/D0CC07526D [58] Chen C M, Zhang Q, Yang M G, et al. Structural evolution during annealing of thermally reduced graphene nanosheets for application in supercapacitors[J]. Carbon,2012,50(10):3572-3584. doi: 10.1016/j.carbon.2012.03.029 [59] Burgess R, Buono C, Davies P R, et al. The functionalisation of graphite surfaces with nitric acid: Identification of functional groups and their effects on gold deposition[J]. Journal of Catalysis,2015,323:10-18. doi: 10.1016/j.jcat.2014.12.021 [60] Sun T, Li Z J, Zhi Y F, et al. Poly(2, 5-Dihydroxy-1, 4-Benzoquinonyl Sulfide) as an efficient cathode for high-performance aqueous zinc-organic batteries[J]. Advanced Functional Materials,2021,31(16):2010049. doi: 10.1002/adfm.202010049 [61] Lee J, Abbas M A, Bang J H. Exploring the capacitive behavior of carbon functionalized with cyclic ethers: A rational strategy to exploit oxygen functional groups for enhanced capacitive performance[J]. ACS Applied Materials & Interfaces,2019,11(15):14126-14135. [62] Wu K H, Wang D W, Gentle I R. Revisiting oxygen reduction reaction on oxidized and unzipped carbon nanotubes[J]. Carbon,2015,81:295-304. doi: 10.1016/j.carbon.2014.09.060 [63] Deng X Y, Li J J, Shan Z, et al. N, O co-doped hierarchical carbon cathode for high-performance Zn-ion hybrid supercapacitors with enhanced pseudocapacitance[J]. Journal of Materials Chemistry A,2020,8(23):11617-11625. doi: 10.1039/D0TA02770G -
 Supporting Information2023-0030.pdf
Supporting Information2023-0030.pdf

-





 下载:
下载: