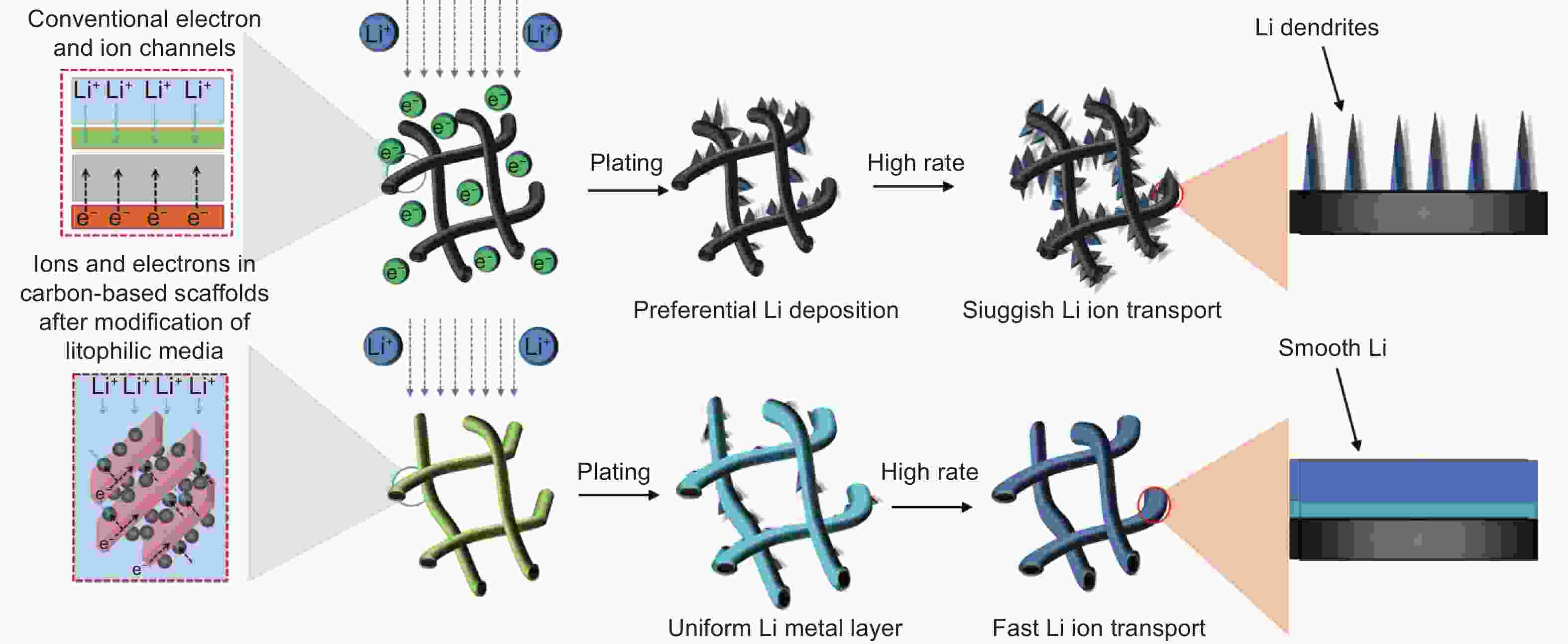
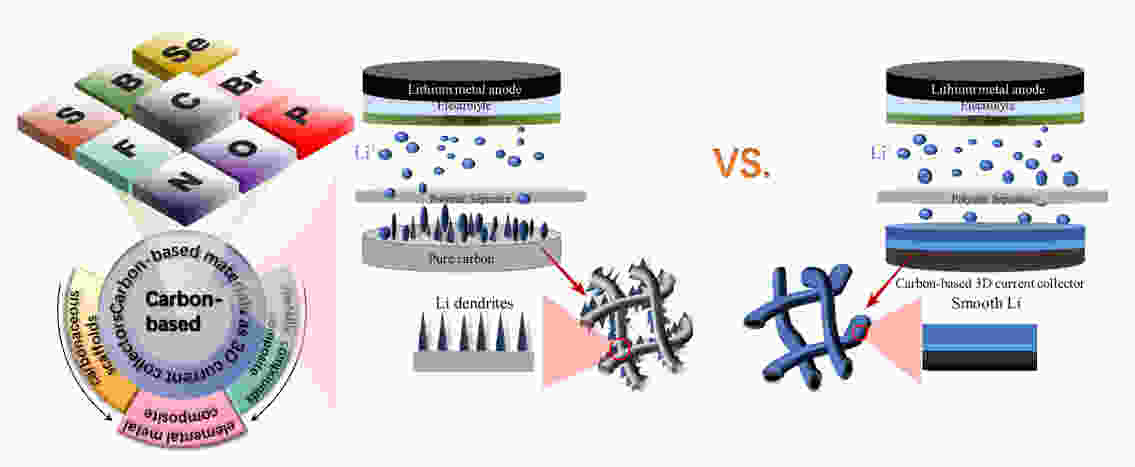
Advances in the use of carbonaceous scaffolds for constructing stable composite Li metal anodes
-
摘要: 因为锂金属电池(LMBs)具有高能量密度、高理论比容量和低氧化电位等优点,被认为是后锂离子电池(LIBs)中理想的能量存储装置之一。然而,锂金属阳极(LMA)面临着多种障碍,包括低库仑效率(CE)、大体积膨胀、锂枝晶的形成、低安全和低稳定性及短寿命,这些问题阻碍了LMBs的实际应用。由于低密度、高机械强度、稳定的化学性质和大比表面积等优势,碳基材料受到了广泛关注。建立复合碳基LMA是各种策略中的一种有效选择,因为其具有缓解体积膨胀、降低局部电流密度以及提供均匀Li+沉积的活性成核位点的能力。本文综述了复合碳基LMA的最新研究进展,包括碳基复合材料、元素金属及其化合物与碳基材料的复合物,以及它们与阳极界面稳定性和结构的关系。最后,本文总结并提出了关于将碳基材料作为LMA支架的观点和见解。Abstract: Compositing lithium metal anodes (LMAs) with carbon-based materials has been given much attention because of the latter’s low density, high mechanical strength, stable electrochemical properties, and large specific surface area. Such a composite LMA stands out because of its ability to reduce the volume expansion, lower the local current density, and provide active nucleation sites for uniform Li+ plating. Recent research advances in carbon-based materials as scaffolds to make composite anodes are reviewed, including composites with pure metals and their alloys, and compositing strategies to improve anode stability.
-
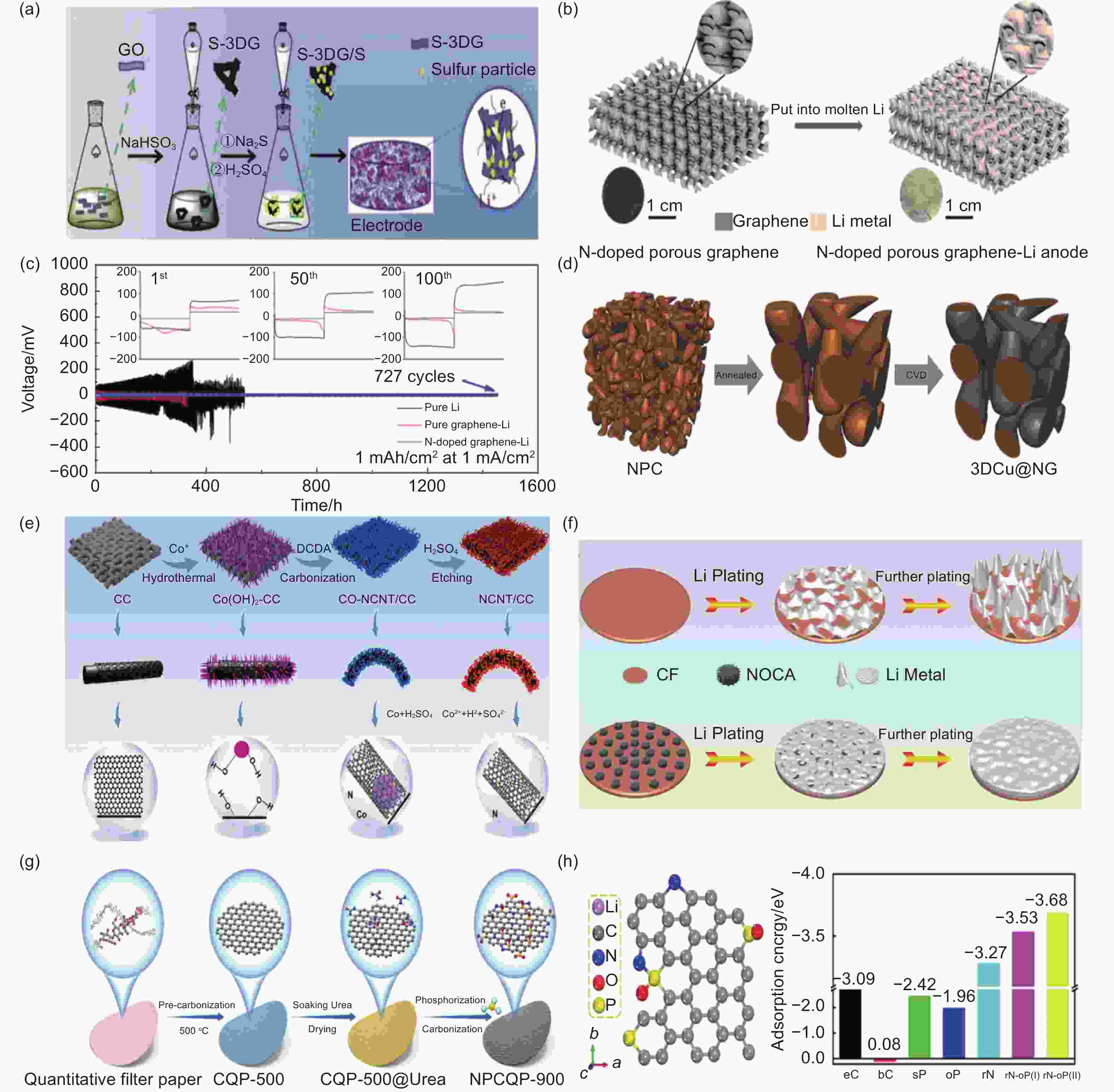
Figure 2. (a) Schematic diagram of the production steps of S-3DG/S and the transmission path and framework of Li and electrons[66]. (b) Schematic of LMA: nanoporous N-doped graphene synthesis[71]. (c) Li foil, graphene-Li, and N-doped graphene-Li symmetrical cells with 1 mAh cm−2 stripping/plating capacity and 1 mA cm−2 current density and voltage characteristics[71]. (d) Schematic diagram of 3DCu@NG manufacturing[72]. (e) The production steps of the NCNT-CC film[73]. (f) Schematic diagram of the deposition behavior of lithium deposition on CF and NOCA@CF[75]. (g) Schematic diagram of the NPCQP-900 synthesis process[76]. (h) Adsorption energy doped with carbon and lithium atoms[76]. ( Reprinted with permission )
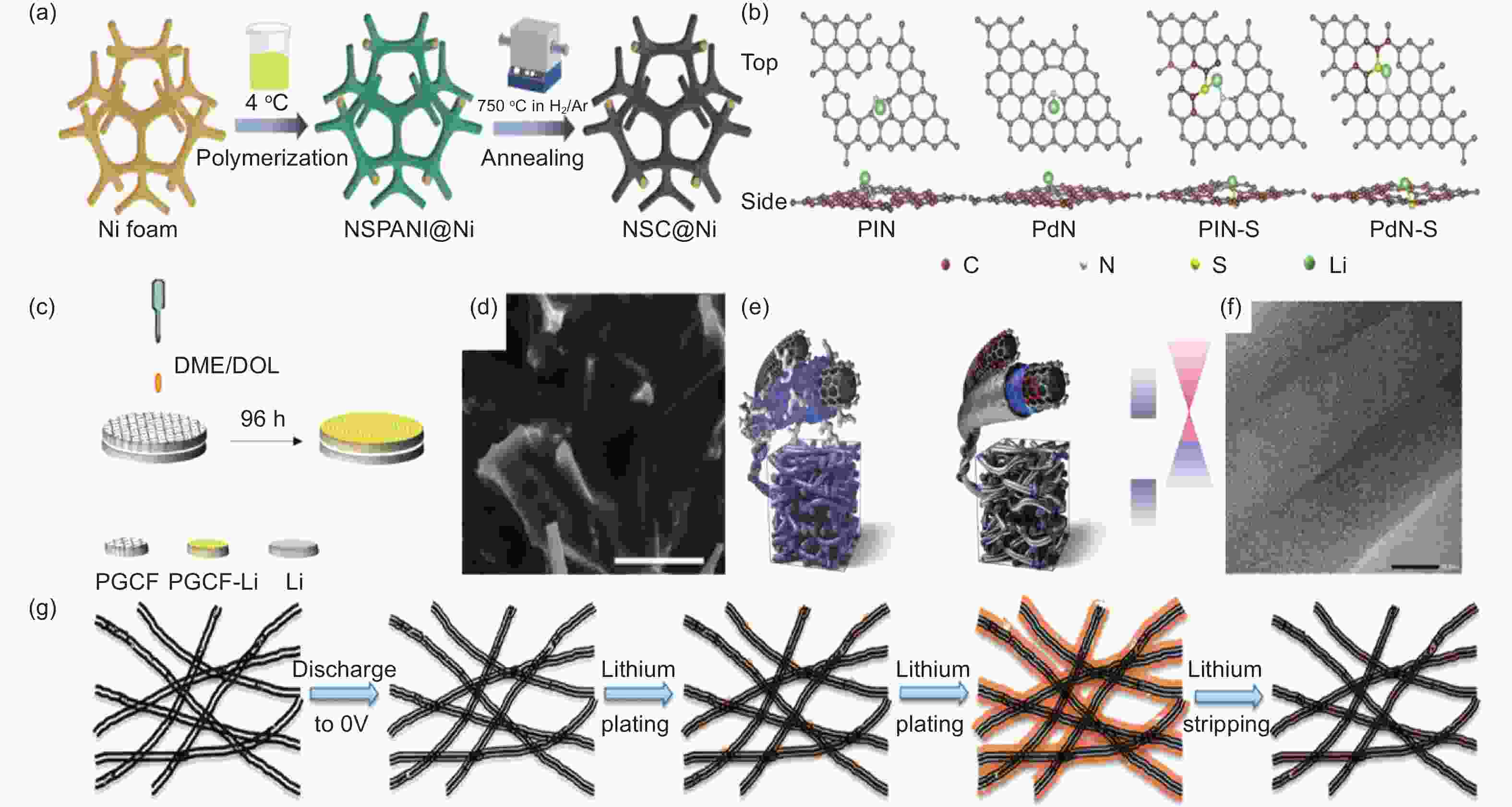
Figure 3. (a) Graphical depiction of the NSC@Ni synthesis method[81]. (b) Optimal configuration of lithium atoms adsorbed at PlN, PdN, PlN-S and PdN-S sites[81]. (c) The fabrication process of PGCF-Li[82]. (d)The SEM image of PGCF-Li is 10 mAh cm−2[82]. (e) Dual-function schematic diagram of a carbon surface with an electron-deficient structure[83]. (f) Physical characterization of the CNT sponge in HRTEM images[84]. (g) Morphology of carbon nanotube sponge during lithium plating/stripping: schematic representation of lithium discharged to carbon nanotube sponge’s electrochemical plating/stripping process[84]. ( Reprinted with permission )
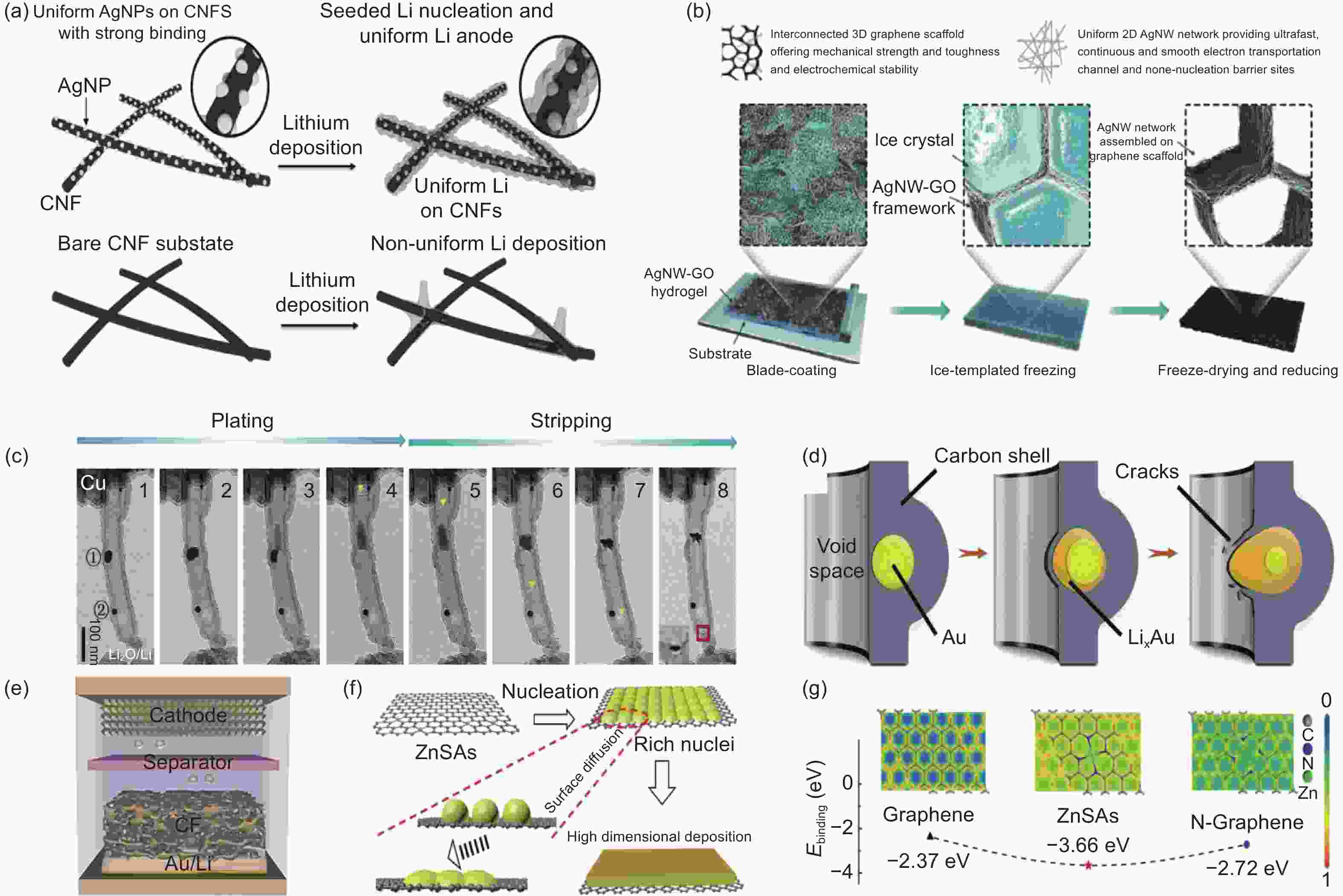
Figure 4. (a) A diagrammatic representation of the nanoseeding approach for the homogeneous deposition of lithium metal on a 3D host material Joule heat anchors AgNPs evenly to the CNF substrate, which is responsible for Li deposition and growth. Thus, lithium metal is guided into the 3D substrate to generate a homogeneous lithium anode[91]. (b) Schematic diagram of the 3D-AGBN host preparation process with the layered characteristics of the solution[94]. (c) The Li plating/stripping process within the Au@aCNT is shown in the TEM snapshot and the corresponding schematic[96]. (d) Schematic of embedded AuNP entering the interior of the tube by breaking through the separation layer upon lithiation[96]. (e) Li is directed deposition to the bottom of the Li/AuCF anode[97]. (f) Formation of multi-dimensional structures through deposition of single Zn atoms[98]. (g) A Schematic representation of the difference in electron density and surface binding energy between graphene, ZnSAs, and N-graphene[98]. ( Reprinted with permission )
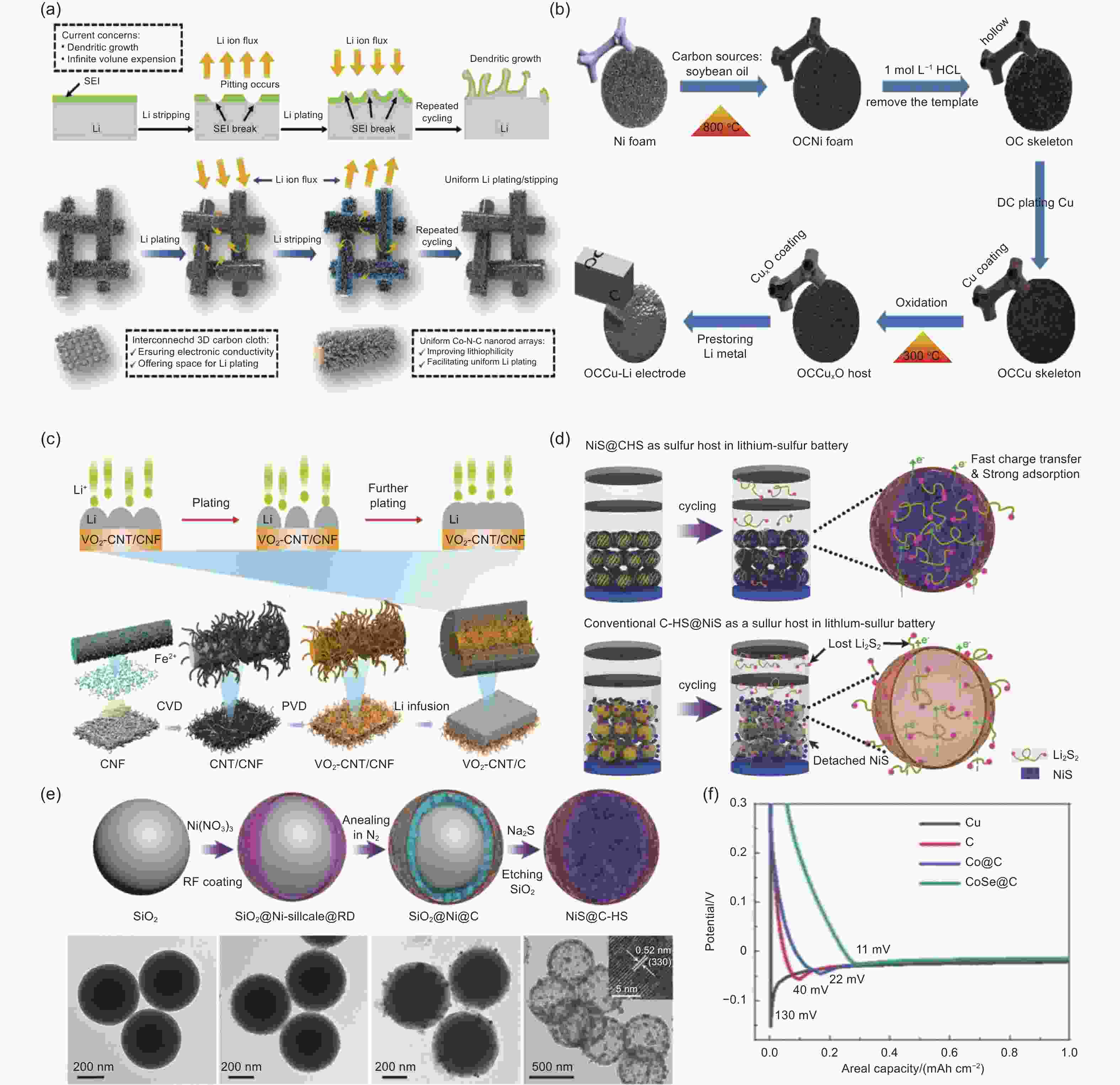
Figure 5. (a) A diagrammatic representation of the peeling and electroplating behavior lithium foil in a planar form and Li@NRA-CC electrode, mechanisms of breakdown by pitting corrosion, SEI fracture, and dendritic growth, and it describes the synergistic influence of interconnected 3D CC and Co-N-C NRAs on lithium stripping/plating behavior[108]. (b) Process flow diagram for the production of OCCu-Li electrode[109]. (c) Schematic of synthetic procedures of VO2-CNT/CNF@Li electrode[111]. (d) Schematic diagram of the NiS@C-HS manufacturing process[112]. (e) Schematic of the sulfur hosts NiS@C-HS and typical C-HS@NiS[112]. (f) Voltage profiles during initial Li plating on different substrates at 1 mA cm−2[113]. ( Reprinted with permission )
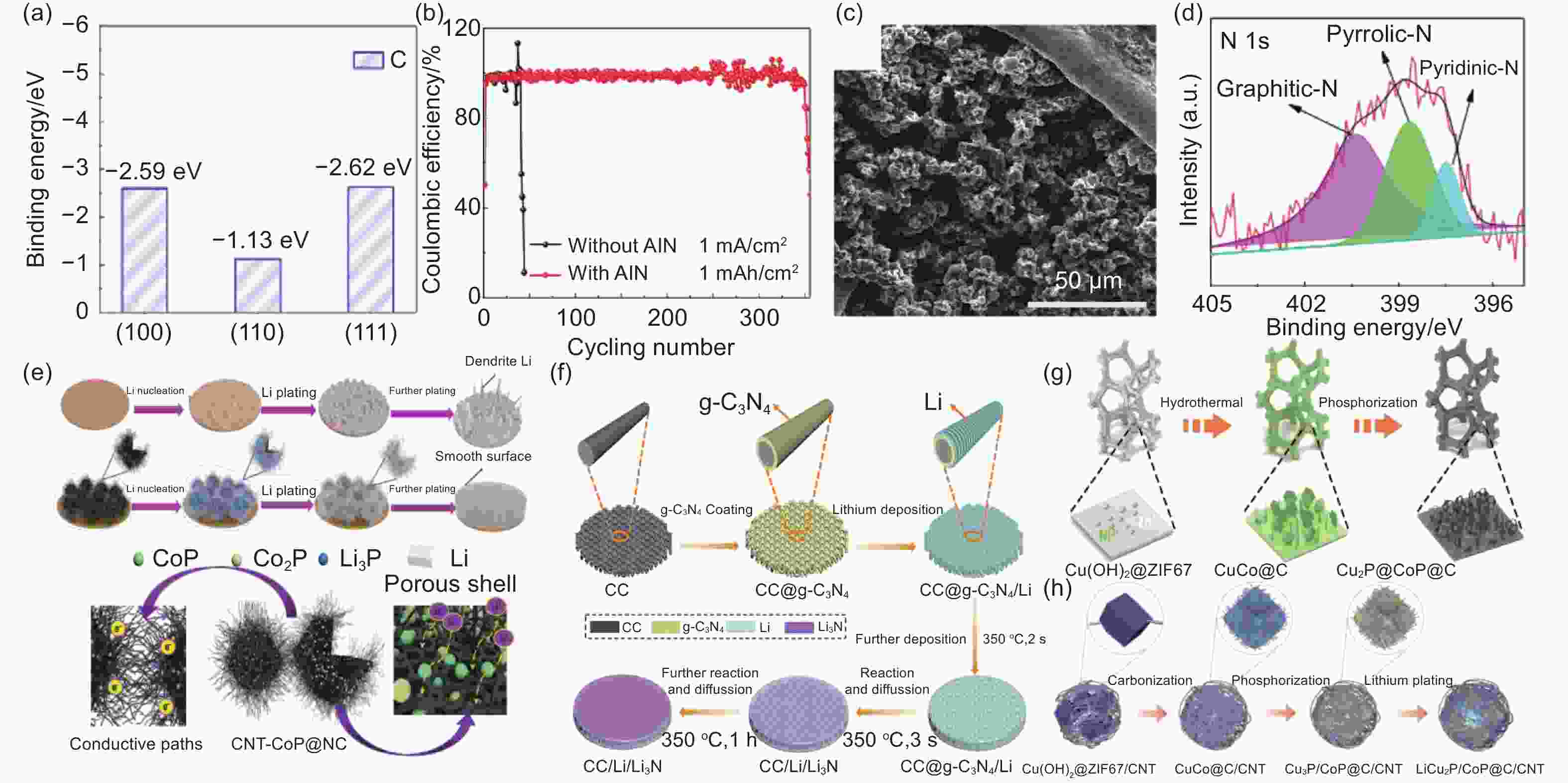
Figure 6. (a) Describe the first-principles calculation of CP binding energy[114]. (b) CP electrodes CE at 1 mA cm−2 and 1 mAh cm−2 at different current densities and different capacities[114] . (c) SEM image of surface topography after Li@NF 100 cycles at 1 mA cm−2[115]. (d) High-resolution XPS spectra of N 1s for CNT-CoP@NC[116]. (e) Lithium nucleation and electroplating process diagram on the Cu foil and CNT-CoP@NC electrode[116]. (f) Process diagram of the preparation process of the CC/Li/Li3N composite electrode[117]. (g) NZP/NF synthesis process diagram[119]. (h) A diagrammatic representation of the preparation process of LiCu3P/CoP@C/CNT[120]. ( Reprinted with permission )
Figure 7. (a) A diagrammatic representation of the reaction mechanism of Li-Ti3C2Tx-CC with a bare lithium electrode[126]. (b) Diagram of lithium plating on MXene@CNF film[127]. (c) Schematic diagram of the lithium stripping/plating process of AlF3@CNF interlayer. A potential gradient (∆E) is formed as the resistance between layers increases[128]. (d) Schematic diagram of lithium metal deposition on BGCF[129]. ( Reprinted with permission )
Figure 8. (a) schematic process of the Li plating on bare CuNW and GDY@CuNW current collectors[135]. (b) impedance variations at different cycles (1.0 mA cm−2 , 1.0 mAh cm−2 )[135]. (c) Raman spectra of GDY and Ni/GDY[136]. (d) Under the conditions of 1 mAh cm−2/1 mA cm−2, the Li plating and stripping efffciency of Ni/GDY electrodes[136]. (e) Schematic lithium plating on bare Cu foil and Cu-GDY NWs[137]. (f) Cycling performance of full cells with LFP at 0.5 C[137]. ( Reprinted with permission )
Table 1. Performance of LMBs using carbonaceous scaffolds as 3D current collectors
Current collector Half cell performance
(Cycle Number/h, CE)Operating conditions (Current censity/(mA cm−2),
Areal capacity/(mAh cm−2))Refs 3D-printed GO frameworks 300, 95.5% 1, 1 [65] Sulfur doped 3D graphene/sulfur particles 100, 93.9% 0.5, 1 [66] N-doped graphene modified 3D porous Cu 50, 97.0% 1, 2 [72] N-doped carbon nanotube modified carbon cloth 400, 99.7% 1, 1 [73] N/O dual-doped 3D porous carbon 350, 95.7% 1, 1 [75] N, P dual-doped carbon 200, 97.5% 1, 1 [76] 3D N, O co-doped carbon nanosphere 600, 98.2% 0.5, 0.5 [80] CNT sponge as a 3D porous 90, 98.5% 1, 2 [84] Table 2. Performance of LMBs using elemental metal composite as 3D current collectors
Table 3. Performance of LMBs using composite with metallic oxygen/sulphur/selenium compounds as 3D current collectors
Current collector Half cell performance
(Cycle number/h,CE)Operating conditions (Current density/(mA cm−2),
Areal capacity/(mAh cm−2))Refs Nanorod arrays modified carbon cloth 100, 97.5% 2, 4 [108] Vanadium oxide modified carbon nanotube films 500, 99% 1, 1 [111] 3D carbon aerogel decorated with cobalt selenide nanoparticles 100, 99.3% 6, 6 [113] Table 4. Performance of LMBs using composite with metal nitrides/phosphides as 3D current collectors
Current collector Half cell performance
(Cycle number/h, CE)Operating conditions (Current density/(mA cm−2),
Areal capacity/(mAh cm−2))Refs Aluminum nitride nanosheets as an additive and carbon
paper as 3D current collector350, 95.8% 1, 1 [114] Nitride decorated nickel foams 300, 97.0% 1, 3 [115] Construction of nickel phosphide nanosheets modified with nickel foam 280, 98.5% 1, 1 [119] Nitrogen-doped hollow porous polyhedron carbon 400, 96.9% 1, 1 [116] Table 5. Performance of LMBs using composite with metal-carbide composites and fluoride/bromide/iodide as 3D current collectors
Current collector Half cell performance
(Cycle number/h, CE)Operating conditions (Current density/(mA cm−2),
Areal capacity/(mAh cm−2))Reference Ti3C2Tx MXene films mixed with trace cellulose nanofibers 200, 98.9% 0.5, 2 [127] AlF3 particles embedded within carbon nanofibers 450, 97.2% 1, 1 [128] CuBr- and Br-doped graphene-like film modified
Cu foam300, 98.8% 2, 2 [129] Table 6. Performance of LMBs using composite with other carbon-based forms as 3D current collectors
Current collector Half cell performance
(Cycle number/h, CE)Operating conditions (Current density/(mA cm−2),
Areal capacity/(mAh cm−2))Reference The CuNW electrode modified by GDY nanofilms 200, 96.5% 0.5, 0.5 [135] Ni-anchored graphdiyne modified copper foam substrate 200, 98.5% 1, 1 [136] grew GDY nanofilms on a Cu nanowire network 500, 99.2% 1, 2 [137] -
[1] Gür T M. Review of electrical energy storage technologies, materials and systems: challenges and prospects for large-scale grid storage[J]. Energy & Environmental Science,2018,11(10):2696-2767 DOI: 10.1039/C8EE01419A . [2] Carley S, Konisky D M. The justice and equity implications of the clean energy transition[J]. Nature Energy,2020,5(8):569-577. doi: 10.1038/s41560-020-0641-6 [3] Friedlingstein P, O'Sullivan M, Jones M W, et al. Global carbon budget 2020[J]. Earth System Science Data,2020,12(4):3269-3340. doi: 10.5194/essd-12-3269-2020 [4] Zhao X, Ma X, Chen B, et al. Challenges toward carbon neutrality in China: Strategies and countermeasures [J]. Resources Conservation And Recycling, 2022, 176:105959 DOI: 10.1016/j.resconrec.2021.105959. [5] Fleischmann S, Mitchell J B, Wang R, et al. Pseudocapacitance: From fundamental understanding to high power energy storage materials[J]. Chemical Reviews,2020,120(14):6738-6782. doi: 10.1021/acs.chemrev.0c00170 [6] Koohi-Fayegh S, Rosen M A. A review of energy storage types, applications and recent developments[J]. Journal of Energy Storage,2020,27:101047. doi: 10.1016/j.est.2019.101047 [7] Reddy M V, Mauger A, Julien C M, et al. Brief history of early lithium-battery development [J]. Materials, 2020, 13(8):1884 DOI: 10.3390/ma13081884. [8] Cheng X B, Zhang R, Zhao C Z, et al. Toward safe lithium metal anode in rechargeable batteries: A review[J]. Chemical Reviews,2017,117(15):10403-10473. doi: 10.1021/acs.chemrev.7b00115 [9] Wang J B, Ren Z, Hou Y, et al. A review of graphene synthesisatlow temperatures by CVD methods[J]. New Carbon Materials,2020,35(3):193-208. doi: 10.1016/S1872-5805(20)60484-X [10] Jin L, Zhang H, Li S, et al. Exchange of li and AgNO3 enabling stable 3D lithium metal anodes with embedded lithophilic nanoparticles and a solid electrolyte interphase inducer[J]. Acs Applied Materials & Interfaces,2021,13(32):38425-38431 DOI: 10.1021/acsami.1c11733 . [11] Fan E, Li L, Wang Z, et al. Sustainable recycling technology for li-ion batteries and beyond: Challenges and future prospects[J]. Chemical Reviews,2020,120(14):7020-7063. doi: 10.1021/acs.chemrev.9b00535 [12] Wu F, Maier J, Yu Y. Guidelines and trends for next-generation rechargeable lithium and lithium-ion batteries[J]. Chemical Society Reviews,2020,49(5):1569-1614. doi: 10.1039/C7CS00863E [13] Zhang Y, Zuo T T, Popovic J, et al. Towards better Li metal anodes: Challenges and strategies[J]. Materials Today,2020,33:56-74. doi: 10.1016/j.mattod.2019.09.018 [14] Liang Y, Xiao Y, Yan C, et al. A bifunctional ethylene-vinyl acetate copolymer protective layer for dendrites-free lithium metal anodes[J]. Journal of Energy Chemistry,2020,48:203-207. doi: 10.1016/j.jechem.2020.01.027 [15] Zhang C, Huang Z, Lv W, et al. Carbon enables the practical use of lithium metal in a battery[J]. Carbon,2017,123:744-755. doi: 10.1016/j.carbon.2017.08.027 [16] Chen T, Wu H P, Wan J, et al. Synthetic poly-dioxolane as universal solid electrolyte interphase for stable lithium metal anodes[J]. Journal of Energy Chemistry,2021,62:172-178. doi: 10.1016/j.jechem.2021.03.018 [17] Wang Z, Qi F, Yin L, et al. An anion-tuned solid electrolyte interphase with fast ion transfer kinetics for stable lithium anodes [J]. Advanced Energy Materials, 2020, 10(14):1903843 DOI: 10.1002/aenm.201903843. [18] Qian H, Li X. Progress in functional solid electrolyte interphases for boosting li metal anode [J]. Acta Physico-Chimica Sinica, 2021, 37(2). [19] Zhang X L, Ruan Z Q, He Q T, et al. Three-dimensional (3D) nanostructured skeleton substrate composed of hollow carbon fiber/carbon nanosheet/ZnO for stable lithium anode[J]. ACS Applied Materials & Interfaces,2021,13(2):3078-3088. [20] Wu H, Jia H, Wang C, et al. Recent progress in understanding solid electrolyte interphase on lithium metal anodes[J]. Advanced Energy Materials,2021,11(5):1-15. [21] Zhai P, Wang T, Jiang H, et al. 3D artificial solid-electrolyte interphase for lithium metal anodes enabled by insulator-metal-insulator layered heterostructures[J]. Advanced Materials,2021,33(13):2006247. doi: 10.1002/adma.202006247 [22] Lu Q, Jie Y, Meng X, et al. Carbon materials for stable li metal anodes: Challenges, solutions, and outlook[J]. Carbon Energy,2021,3(6):957-975. doi: 10.1002/cey2.147 [23] Meda U S, Lal L, Sushantha M, et al. Solid electrolyte interphase (sei), a boon or a bane for lithium batteries: A review on the recent advances [J]. Journal of Energy Storage, 2021: 103564 DOI: 10.1016/j.est.2021.103564. [24] Shan X, Zhong Y, Zhang L, et al. A brief review on solid electrolyte interphase composition characterization technology for lithium metal batteries: Challenges and perspectives[J]. Journal Of Physical Chemistry C,2021,125(35):19060-19080. doi: 10.1021/acs.jpcc.1c06277 [25] Yan T, Li F, Xu C, et al. Highly uniform lithiated nafion thin coating on separator as an artificial SEI layer of lithium metal anode toward suppressed dendrite growth[J]. Electrochimica Acta,2022,410:140004. doi: 10.1016/j.electacta.2022.140004 [26] Jumi K I M, Jimin O, Kim J Y, et al. Recent progress and perspectives of solid electrolytes for lithium rechargeable batteries[J]. Journal of the Korean Electrochemical Society,2019,22(3):87-103. [27] Nie K, Hong Y, Qiu J, et al. Interfaces between cathode and electrolyte in solid state lithium batteries: Challenges and perspectives[J]. Frontiers In Chemistry,2018,6:616. doi: 10.3389/fchem.2018.00616 [28] Zhao C Z, Duan H, Huang J Q, et al. Designing solid-state interfaces on lithium-metal anodes: a review[J]. Science China-Chemistry,2019,62(10):1286-1299. doi: 10.1007/s11426-019-9519-9 [29] Kang D, Xiao M, Lemmon J P. Artificial solid-electrolyte interphase for lithium metal batteries[J]. Batteries & Supercaps,2021,4(3):445-455. [30] Jin C B, Shi P, Zhang X Q, et al. Advances in carbon materials for stable lithium metal batteries[J]. New Carbon Materials,2022,37(1):1-24. doi: 10.1016/S1872-5805(22)60573-0 [31] Liu Y, Zhai Y, Xia Y, et al. Recent progress of porous materials in lithium-metal batteries[J]. Small Structures,2021,2(5):2000118. doi: 10.1002/sstr.202000118 [32] Chen L, Ding K, Li K, et al. Crystalline porous materials-based solid-state electrolytes for lithium metal batteries[J]. Energy Chem,2022,4(3):100073. doi: 10.1016/j.enchem.2022.100073 [33] Li N, Wei W, Xie K, et al. Suppressing dendritic lithium formation using porous media in lithium metal-based batteries[J]. Nano Letters,2018,18(3):2067-2073. doi: 10.1021/acs.nanolett.8b00183 [34] Jin C, Sheng O, Luo J, et al. 3D lithium metal embedded within lithiophilic porous matrix for stable lithium metal batteries[J]. Nano Energy,2017,37:177-186. doi: 10.1016/j.nanoen.2017.05.015 [35] Yun Q, He Y B, Lv W, et al. Chemical dealloying derived 3D porous current collector for li metal anodes[J]. Advanced Materials,2016,28(32):6932-+. doi: 10.1002/adma.201601409 [36] Guo C, Zhang W, Tu J, et al. Construction of 3D copper-based collector and its application in lithium metal batteries[J]. Progress In Chemistry,2022,34(2):370-383. [37] Chen J Y, Zhao J, Lei L N, et al. Dynamic intelligent Cu current collectors for ultrastable lithium metal anodes[J]. Nano Letters,2020,20(5):3403-3410. doi: 10.1021/acs.nanolett.0c00316 [38] Zhang D, Dai A, Wu M, et al. Lithiophilic 3D porous cuzn current collector for stable lithium metal batteries[J]. ACS Energy Letters,2020,5(1):180-186. doi: 10.1021/acsenergylett.9b01987 [39] Lu Z, Liang Q, Wang B, et al. Graphitic carbon nitride induced micro-electric field for dendrite-free lithium metal anodes[J]. Advanced Energy Materials,2019,9(7):1803186. doi: 10.1002/aenm.201803186 [40] Pu J, Li J, Zhang K, et al. Conductivity and lithiophilicity gradients guide lithium deposition to mitigate short circuits[J]. Nature Communications,2019,10(1):1-10. doi: 10.1038/s41467-018-07882-8 [41] Shang J, Yu W, Wang L, et al. Metallic glass-fiber fabrics: a new type of flexible, super-lightweight, and 3D current collector for lithium batteries[J]. Advanced Materials,2023,35:2211748. [42] Jin S, Jiang Y, Ji H, et al. Advanced 3D current collectors for lithium-based batteries[J]. Advanced Materials,2018,30(48):1802014. doi: 10.1002/adma.201802014 [43] Xia Y, Hu W, Yao Y, et al. Application of electrodeposited Cu-metal nanoflake structures as 3D current collector in lithium-metal batteries[J]. Nanotechnology,2022,33(24):1361-6528. [44] Zhang L, Jin Q, Zhao K, et al. 3D hierarchical Cu@Ag nanostructure as a current collector for dendrite-free lithium metal anode[J]. Dalton Transactions,2022,51(43):16565-16573. doi: 10.1039/D2DT02937E [45] Yang S, Cheng Y, Xiao X, et al. Development and application of carbon fiber in batteries[J]. Chemical Engineering Journal,2020,384:123294. doi: 10.1016/j.cej.2019.123294 [46] Tang K, Xiao J, Li X, et al. Advances of carbon-based materials for lithium metal anodes[J]. Frontiers In Chemistry,2020,8:595972. doi: 10.3389/fchem.2020.595972 [47] Fu A, Wang C, Pei F, et al. Recent advances in hollow porous carbon materials for lithium-sulfur batteries[J]. Small,2019,15(10):1804786. doi: 10.1002/smll.201804786 [48] Wu Z, Sun K, Wang Z. A review of the application of carbon materials for lithium metal batteries[J]. Batteries-Basel,2022,8(11):246. doi: 10.3390/batteries8110246 [49] Deng W, Zhu W, Zhou X, et al. Graphene nested porous carbon current collector for lithium metal anode with ultrahigh areal capacity[J]. Energy Storage Materials,2018,15:266-273. doi: 10.1016/j.ensm.2018.05.005 [50] Chen H, Yang Y, Boyle D T, et al. Free-standing ultrathin lithium metal–graphene oxide host foils with controllable thickness for lithium batteries[J]. Nature Energy,2021,6(8):790-798. doi: 10.1038/s41560-021-00833-6 [51] Sun C, Liu Y, Sheng J, et al. Status and prospects of porous graphene networks for lithium-sulfur batteries[J]. Materials Horizons,2020,7(10):2487-2518. doi: 10.1039/D0MH00815J [52] Xu Q, Yang X, Rao M, et al. High energy density lithium metal batteries enabled by a porous graphene/MgF2 framework[J]. Energy Storage Materials,2020,26:73-82. doi: 10.1016/j.ensm.2019.12.028 [53] Lu K, Xu H, He H, et al. Modulating reactivity and stability of metallic lithium via atomic doping[J]. Journal of Materials Chemistry A,2020,8(20):10363-10369. doi: 10.1039/D0TA02176H [54] Chen X, Chen X R, Hou T Z, et al. Lithiophilicity chemistry of heteroatom-doped carbon to guide uniform lithium nucleation in lithium metal anodes[J]. Science Advances,2019,5(2):eaau7728. doi: 10.1126/sciadv.aau7728 [55] Yuan Y, Chen Z, Yu H, et al. Heteroatom-doped carbon-based materials for lithium and sodium ion batteries[J]. Energy Storage Materials,2020,32:65-90. doi: 10.1016/j.ensm.2020.07.027 [56] Wu Y, Rahm E, Holze R. Effects of heteroatoms on electrochemical performance of electrode materials for lithium ion batteries[J]. Electrochimica Acta,2002,47(21):3491-3507. doi: 10.1016/S0013-4686(02)00317-1 [57] Shao R, Zhu F, Cao Z, et al. Heteroatom-doped carbon networks enabling robust and flexible silicon anodes for high energy Li-ion batteries[J]. Journal of Materials Chemistry A,2020,8(35):18338-18347. doi: 10.1039/D0TA06647H [58] Pappas G S, Ferrari S, Huang X, et al. Heteroatom doped-carbon nanospheres as anodes in lithium ion batteries[J]. Materials,2016,9(1):35. doi: 10.3390/ma9010035 [59] Wang Y, Yuan C, Li K, et al. Freestanding porous silicon@ heteroatom-doped porous carbon fiber anodes for high-performance lithium-ion batteries[J]. ACS Applied Energy Materials,2022,5(9):11462-11471. doi: 10.1021/acsaem.2c01898 [60] Dai C, Sun G, Hu L, et al. Recent progress in graphene-based electrodes for flexible batteries[J]. Infomat,2020,2(3):509-526. doi: 10.1002/inf2.12039 [61] Xu Z, Zhang P, Chen J, et al. Growth and growth mechanism of oxide nanocrystals on electrochemically exfoliated graphene for lithium storage[J]. Energy Storage Materials,2019,18:174-181. doi: 10.1016/j.ensm.2018.08.023 [62] Seyyedin S T, Yaftian M R, Sovizi M R. Cobalt oxyhydroxide/graphene oxide nanocomposite for amelioration of electrochemical performance of lithium/sulfur batteries[J]. Journal of Solid State Electrochemistry,2017,21(3):649-656. doi: 10.1007/s10008-016-3411-4 [63] Zhang L, Ma T, Yang Y W, et al. Pomegranate-inspired graphene parcel enables high-performance dendrite-free lithium metal anodes[J]. Advanced Science,2022,9(28):2203178. doi: 10.1002/advs.202203178 [64] Zhang M, Shan Y, Kong Q, et al. Applications of metal-organic framework-graphene composite materials in electrochemical energy storage[J]. Flatchem,2022,32:100332. doi: 10.1016/j.flatc.2021.100332 [65] Yang Y, Ai L, Yu S, et al. 3D-printed porous go framework enabling dendrite-free lithium-metal anodes[J]. ACS Applied Energy Materials,2022,5(12):15666-15672. doi: 10.1021/acsaem.2c03267 [66] Li N, Gan F, Wang P, et al. In situ synthesis of 3D sulfur-doped graphene/sulfur as a cathode material for lithium-sulfur batteries[J]. Journal of Alloys and Compounds,2018,754:64-71. doi: 10.1016/j.jallcom.2018.04.018 [67] Zhu J, Tu W, Pan H, et al. Self-templating synthesis of hollow Co3O4 nanoparticles embedded in N, S-dual-doped reduced graphene oxide for lithium ion batteries[J]. ACS Nano,2020,14(5):5780-5787. doi: 10.1021/acsnano.0c00712 [68] Zhang F, Liu X, Yang M, et al. Novel S-doped ordered mesoporous carbon nanospheres toward advanced lithium metal anodes[J]. Nano Energy,2020,69:104443. doi: 10.1016/j.nanoen.2019.104443 [69] Choi Y J, Lee G W, Kim Y H, et al. Microspherical assembly of selectively pyridinic N-doped nanoperforated graphene for stable Li-metal anodes: Synergistic coupling of lithiophilic pyridinic N on perforation edges and low tortuosity via graphene nanoperforation[J]. Chemical Engineering Journal,2023,455:140770. doi: 10.1016/j.cej.2022.140770 [70] Fang Y, Hsieh Y Y, Khosravifar M, et al. Lithiophilic current collector based on nitrogen doped carbon nanotubes and three-dimensional graphene for long-life lithium metal batteries[J]. Materials Science and Engineering:B,2021,267:115067. doi: 10.1016/j.mseb.2021.115067 [71] Huang G, Han J, Zhang F, et al. Lithiophilic 3D nanoporous nitrogen-doped graphene for dendrite-free and ultrahigh-rate lithium-metal anodes[J]. Advanced Materials,2019,31(2):1805334. doi: 10.1002/adma.201805334 [72] Zhang R, Wen S, Wang N, et al. N-doped graphene modified 3D porous Cu current collector toward microscale homogeneous Li deposition for Li metal anodes[J]. Advanced Energy Materials,2018,8(23):1800914. doi: 10.1002/aenm.201800914 [73] Qiao L, Zhang R, Li Y, et al. Super-assembled hierarchical and stable N-doped carbon nanotube nanoarrays for dendrite-free lithium metal batteries[J]. ACS Applied Energy Materials,2021,5(1):815-824. [74] Wang H, An D, Tian P, et al. Incorporating quantum-sized boron dots into 3D cross-linked rGO skeleton to enable the activity of boron anode for favorable lithium storage[J]. Chemical Engineering Journal,2021,425:130659. doi: 10.1016/j.cej.2021.130659 [75] An Y, Tian Y, Li Y, et al. Heteroatom-doped 3D porous carbon architectures for highly stable aqueous zinc metal batteries and non-aqueous lithium metal batteries[J]. Chemical Engineering Journal,2020,400:125843. doi: 10.1016/j.cej.2020.125843 [76] Lu C, Tian M, Wei C, et al. Synergized N, P dual-doped 3D carbon host derived from filter paper for durable lithium metal anodes[J]. Journal of Colloid and Interface Science,2023,632:1-10. doi: 10.1016/j.jcis.2022.11.022 [77] Li H, Liu J, Zhang Y, et al. Mono-atom dispersed graphene foam with nitrogen-doped carbon nanospheres used in preparation of anode material for lithium-sulfur battery, is grown with carbon nanospheres doped with nitrogen atoms and single metal atoms, CN113104840-A [P/OL]. [78] Wang J, Han W Q. A review of heteroatom doped materials for advanced lithium-sulfur batteries[J]. Advanced Functional Materials,2022,32(2):07166. [79] Wu J, Pan Z, Zhang Y, et al. The recent progress of nitrogen-doped carbon nanomaterials for electrochemical batteries[J]. Journal of Materials Chemistry A,2018,6(27):12932-12944. doi: 10.1039/C8TA03968B [80] Gao C, Li J, Sun K, et al. Controllable lithium deposition behavior hollow of N, O co-doped carbon nanospheres for practical lithium metal batteries[J]. Chemical Engineering Journal,2021,412:128721. doi: 10.1016/j.cej.2021.128721 [81] Tang K, Xiao J, Long M, et al. Superlithiophilic N, S-codoped carbon on Ni foam as a stable 3D host for dendrite-free Li metal anodes[J]. Sustainable Materials and Technologies,2022,32:e00408. doi: 10.1016/j.susmat.2022.e00408 [82] Chen T, Jia W, Yao Z, et al. Partly lithiated graphitic carbon foam as 3D porous current collectors for dendrite-free lithium metal anodes[J]. Electrochemistry Communications,2019,107:106535. doi: 10.1016/j.elecom.2019.106535 [83] Kwon H, Lee J H, Roh Y, et al. An electron-deficient carbon current collector for anode-free Li-metal batteries[J]. Nature Communications,2021,12(1):5537. doi: 10.1038/s41467-021-25848-1 [84] Yang G, Li Y, Tong Y, et al. Lithium plating and stripping on carbon nanotube sponge[J]. Nano Letters,2019,19(1):494-499. doi: 10.1021/acs.nanolett.8b04376 [85] Huang Z, Kong D, Zhang Y, et al. Vertical graphenes grown on a flexible graphite paper as an all-carbon current collector towards stable li deposition [J]. Research, 2020:7163948 DOI: 10.34133/2020/7163948. [86] Meng Q, Deng B, Zhang H, et al. Heterogeneous nucleation and growth of electrodeposited lithium metal on the basal plane of single-layer graphene[J]. Energy Storage Materials,2019,16:419-425. doi: 10.1016/j.ensm.2018.06.024 [87] Zhai P, Wang T, Yang W, et al. Uniform lithium deposition assisted by single-atom doping toward high-performance lithium metal anodes [J]. Advanced Energy Materials, 2019, 9(18):1804019 DOI: 10.1002/aenm.201804019. [88] Ye H, Xin S, Yin Y X, et al. Stable li plating/stripping electrochemistry realized by a hybrid li reservoir in spherical carbon granules with 3D conducting skeletons[J]. Journal of the American Chemical Society,2017,139(16):5916-5922. doi: 10.1021/jacs.7b01763 [89] Sun Y, Zhao W, Wang X, et al. Progress of carbon and metal-based three-dimensional materials for dendrite-proof and interface-compatible lithium metal anode[J]. Applied Surface Science,2022,598:153785. doi: 10.1016/j.apsusc.2022.153785 [90] Tian B, Huang Z, Xu X, et al. Three-dimensional Ag/carbon nanotube-graphene foam for high performance dendrite free lithium/sodium metal anodes[J]. Journal of Materials Science & Technology,2023,132:50-58. [91] Yang C, Yao Y, He S, et al. Ultrafine silver nanoparticles for seeded lithium deposition toward stable lithium metal anode[J]. Advanced Materials,2017,29(38):1702714. doi: 10.1002/adma.201702714 [92] Sun Q, Zhai W, Hou G, et al. In situ synthesis of a lithiophilic ag-nanoparticles-decorated 3D porous carbon framework toward dendrite-free lithium metal anodes[J]. ACS Sustainable Chemistry & Engineering,2018,6(11):15219-15227. [93] Hou G, Ren X, Ma X, et al. Dendrite-free Li metal anode enabled by a 3D free-standing lithiophilic nitrogen-enriched carbon sponge[J]. Journal of Power Sources,2018,386:77-84. doi: 10.1016/j.jpowsour.2018.03.049 [94] Xue P, Liu S, Shi X, et al. A hierarchical silver-nanowire-graphene host enabling ultrahigh rates and superior long-term cycling of lithium-metal composite anodes[J]. Advanced Materials,2018,30(44):1804165. doi: 10.1002/adma.201804165 [95] Yan K, Lu Z, Lee H W, et al. Selective deposition and stable encapsulation of lithium through heterogeneous seeded growth[J]. Nature Energy,2016,1(3):16010. doi: 10.1038/nenergy.2016.10 [96] Lan X, Ye W, Zheng H, et al. Encapsulating lithium and sodium inside amorphous carbon nanotubes through gold-seeded growth[J]. Nano Energy,2019,66:104178. doi: 10.1016/j.nanoen.2019.104178 [97] Li D, Gao Y, Xie C, et al. Au-coated carbon fabric as Janus current collector for dendrite-free flexible lithium metal anode and battery[J]. Applied Physics Reviews,2022,9(1):011424. doi: 10.1063/5.0083830 [98] Xu K, Zhu M, Wu X, et al. Dendrite-tamed deposition kinetics using single-atom Zn sites for Li metal anode[J]. Energy Storage Materials,2019,23:587-593. doi: 10.1016/j.ensm.2019.03.025 [99] Matsui M. Study on electrochemically deposited Mg metal[J]. Journal of Power Sources,2011,196(16):7048-7055. doi: 10.1016/j.jpowsour.2010.11.141 [100] Ling C, Banerjee D, Matsui M. Study of the electrochemical deposition of Mg in the atomic level: Why it prefers the non-dendritic morphology[J]. Electrochimica Acta,2012,76:270-274. doi: 10.1016/j.electacta.2012.05.001 [101] Ding Y, Hu L, He D, et al. Design of multishell microsphere of transition metal oxides/carbon composites for lithium ion battery[J]. Chemical Engineering Journal,2020,380:122489. doi: 10.1016/j.cej.2019.122489 [102] Tan X, Wu Y, Lin X, et al. Application of MOF-derived transition metal oxides and composites as anodes for lithium-ion batteries[J]. Inorganic Chemistry Frontiers,2020,7(24):4939-4955. doi: 10.1039/D0QI00929F [103] Reddy R C K, Lin J, Chen Y, et al. Progress of nanostructured metal oxides derived from metal-organic frameworks as anode materials for lithium-ion batteries[J]. Coordination Chemistry Reviews,2020,420:213434. doi: 10.1016/j.ccr.2020.213434 [104] Yu B, Tao T, Mateti S, et al. Nanoflake arrays of lithiophilic metal oxides for the ultra-stable anodes of lithium-metal batteries[J]. Advanced Functional Materials,2018,28(36):1803023. doi: 10.1002/adfm.201803023 [105] Yu Z, Qu X, Dou A, et al. Carbon-coated cation-disordered rocksalt-type transition metal oxide composites for high energy Li-ion batteries[J]. Ceramics International,2021,47(2):1758-1765. doi: 10.1016/j.ceramint.2020.09.001 [106] Xiang M, Wu H, Liu H, et al. A flexible 3D multifunctional MgO-decorated carbon foam@ CNTs hybrid as self-supported cathode for high-performance lithium-sulfur batteries[J]. Advanced Functional Materials,2017,27(37):1702573. doi: 10.1002/adfm.201702573 [107] Zhang Y, Liu B, Hitz E, et al. A carbon-based 3D current collector with surface protection for Li metal anode[J]. Nano Research,2017,10(4):1356-1365. doi: 10.1007/s12274-017-1461-2 [108] Wang T S, Liu X, Wang Y, et al. High areal capacity dendrite-free li anode enabled by metal-organic framework-derived nanorod array modified carbon cloth for solid state li metal batteries[J]. Advanced Functional Materials,2021,31(2):2001973. doi: 10.1002/adfm.202001973 [109] Yue X Y, Bao J, Qiu Q Q, et al. Copper decorated ultralight 3D carbon skeleton derived from soybean oil for dendrite-free Li metal anode[J]. Chemical Engineering Journal,2020,391:123516. doi: 10.1016/j.cej.2019.123516 [110] Zeng L, Zhou T, Xu X, et al. General construction of lithiophilic 3D skeleton for dendrite-free lithium metal anode via a versatile MOF-derived route[J]. Science China-Materials,2022,65(2):337-348. doi: 10.1007/s40843-021-1764-x [111] Xu C, Wang H, Liu X, et al. Lithiophilic vanadium oxide coated three-dimensional carbon network design towards stable lithium metal anode[J]. Journal of Power Sources,2023,562:232778. doi: 10.1016/j.jpowsour.2023.232778 [112] Ye C, Zhang L, Guo C, et al. A 3D hybrid of chemically coupled nickel sulfide and hollow carbon spheres for high performance lithium-sulfur batteries[J]. Advanced Functional Materials,2017,27(33):1702524. doi: 10.1002/adfm.201702524 [113] He J, Manthiram A. 3D CoSe@ C aerogel as a host for dendrite-free lithium-metal anode and efficient sulfur cathode in Li-S full cells[J]. Advanced Energy Materials,2020,10(41):2002654. doi: 10.1002/aenm.202002654 [114] Gao C, Hong B, Sun K, et al. Self-suppression of lithium dendrite with aluminum nitride nanoflake additive in 3D carbon paper for lithium metal batteries[J]. Energy Technology,2020,8(7):1901463. doi: 10.1002/ente.201901463 [115] Zhu J, Chen J, Luo Y, et al. Lithiophilic metallic nitrides modified nickel foam by plasma for stable lithium metal anode[J]. Energy Storage Materials,2019,23:539-546. doi: 10.1016/j.ensm.2019.04.005 [116] Song Z, Liu Y, Wang Z, et al. Synergistic modulation of Li nucleation/growth enabled by CNTs-wrapped lithiophilic CoP/Co2P decorated hollow carbon polyhedron host for stable lithium metal anodes [J]. Nano Research, 2023:4961–4969 DOI: 10.1007/s12274-022-5179-4. [117] Cao W, Chen W, Lu M, et al. In situ generation of Li3N concentration gradient in 3D carbon-based lithium anodes towards highly-stable lithium metal batteries[J]. Journal of Energy Chemistry,2023,76:648-656. doi: 10.1016/j.jechem.2022.09.025 [118] Luo L, Li J, Yaghoobnejad Asl H, et al. A 3D lithiophilic Mo2N-modified carbon nanofiber architecture for dendrite-free lithium-metal anodes in a full cell[J]. Advanced Materials,2019,31(48):1904537. doi: 10.1002/adma.201904537 [119] Wang Z, Wang J, Mao Q, et al. Uniform lithium deposition and dissolution via metallic phosphides medium for stable cycling lithium metal batteries[J]. Chemical Engineering Journal,2021,407:126861. doi: 10.1016/j.cej.2020.126861 [120] Zhang X, Jin S, Seo M H, et al. Hierarchical porous structure construction for highly stable self-supporting lithium metal anode[J]. Nano Energy,2022,93:106905. doi: 10.1016/j.nanoen.2021.106905 [121] Zhang W, Jin H, Du Y, et al. Sulfur and nitrogen codoped Nb2C MXene for dendrite-free lithium metal battery[J]. Electrochimica Acta,2021,390:138812. doi: 10.1016/j.electacta.2021.138812 [122] Shi H, Yue M, Zhang C J, et al. 3D flexible, conductive, and recyclable Ti3C2Tx mxene-melamine foam for high-areal-capacity and long-lifetime alkali-metal anode[J]. ACS Nano,2020,14(7):8678-8688. doi: 10.1021/acsnano.0c03042 [123] Tian Y, An Y, Wei C, et al. Flexible and free-standing Ti3C2Tx mxene@zn paper for dendrite-free aqueous zinc metal batteries and nonaqueous lithium metal batteries[J]. ACS Nano,2019,13(10):11676-11685. doi: 10.1021/acsnano.9b05599 [124] Chen Q, Wei Y, Zhang X, et al. Vertically aligned mxene nanosheet arrays for high-rate lithium metal anodes[J]. Advanced Energy Materials,2022,12(18):2200072. doi: 10.1002/aenm.202200072 [125] Shen Y, Pu Z, Zhang Y, et al. MXene/ZnO flexible freestanding film as a dendrite-free support in lithium metal batteries[J]. Journal of Materials Chemistry A,2022,10(33):17199-17207. doi: 10.1039/D2TA04797G [126] Fang Y Z, Liang S, Zhang X, et al. Li (110) lattice plane evolution induced by a 3D MXene skeleton for stable lithium metal anodes[J]. Chemical Communications,2022,58(67):9373-9376. doi: 10.1039/D2CC03288K [127] Wang C Y, Zheng Z J, Feng Y Q, et al. Topological design of ultrastrong MXene paper hosted Li enables ultrathin and fully flexible lithium metal batteries[J]. Nano Energy,2020,74:104817. doi: 10.1016/j.nanoen.2020.104817 [128] Guo C, Yang H, Naveed A, et al. AlF 3-Modified carbon nanofibers as a multifunctional 3D interlayer for stable lithium metal anodes[J]. Chemical Communications,2018,54(60):8347-8350. doi: 10.1039/C8CC04422H [129] Duan H, Zhang J, Chen X, et al. Uniform nucleation of lithium in 3D current collectors via bromide intermediates for stable cycling lithium metal batteries[J]. Journal of the American Chemical Society,2018,140(51):18051-18057. doi: 10.1021/jacs.8b10488 [130] Shi H, Zhang C J, Lu P, et al. Conducting and lithiophilic mxene/graphene framework for high-capacity, dendrite-free lithium-metal anodes[J]. ACS Nano,2019,13(12):14308-14318. doi: 10.1021/acsnano.9b07710 [131] Xia Y, Mathis T S, Zhao M Q, et al. Thickness-independent capacitance of vertically aligned liquid-crystalline MXenes[J]. Nature,2018,557(7705):409-412. doi: 10.1038/s41586-018-0109-z [132] Tan J, Matz J, Dong P, et al. A growing appreciation for the role of lif in the solid electrolyte interphase[J]. Advanced Energy Materials,2021,11(16):2100046. doi: 10.1002/aenm.202100046 [133] Zhao J, Liao L, Shi F, et al. Surface fluorination of reactive battery anode materials for enhanced stability[J]. Journal of the American Chemical Society,2017,139(33):11550-11558. doi: 10.1021/jacs.7b05251 [134] Liu Z, He B, Zhang Z, et al. Lithium/graphene composite anode with 3D structural lif protection layer for high-performance lithium metal batteries[J]. ACS Applied Materials & Interfaces,2022,14(2):2871-2880. [135] Shang H, Zuo Z, Li Y. Highly lithiophilic graphdiyne nanofilm on 3D free-standing Cu nanowires for high-energy-density electrodes[J]. ACS Applied Materials & Interfaces,2019,11(19):17678-17685. [136] Kang H, Hua B, Gao P, et al. Ni/Graphdiyne composites inhibit dendrite growth in lithium metal anodes[J]. Electrochimica Acta,2023,440:141744. doi: 10.1016/j.electacta.2022.141744 [137] Zhu M, Yin C, Wang Q, et al. Columnar lithium deposition guided by graphdiyne nanowalls toward a stable lithium metal anode[J]. ACS Applied Materials & Interfaces,2022,14(50):55700-55708. -





 下载:
下载: