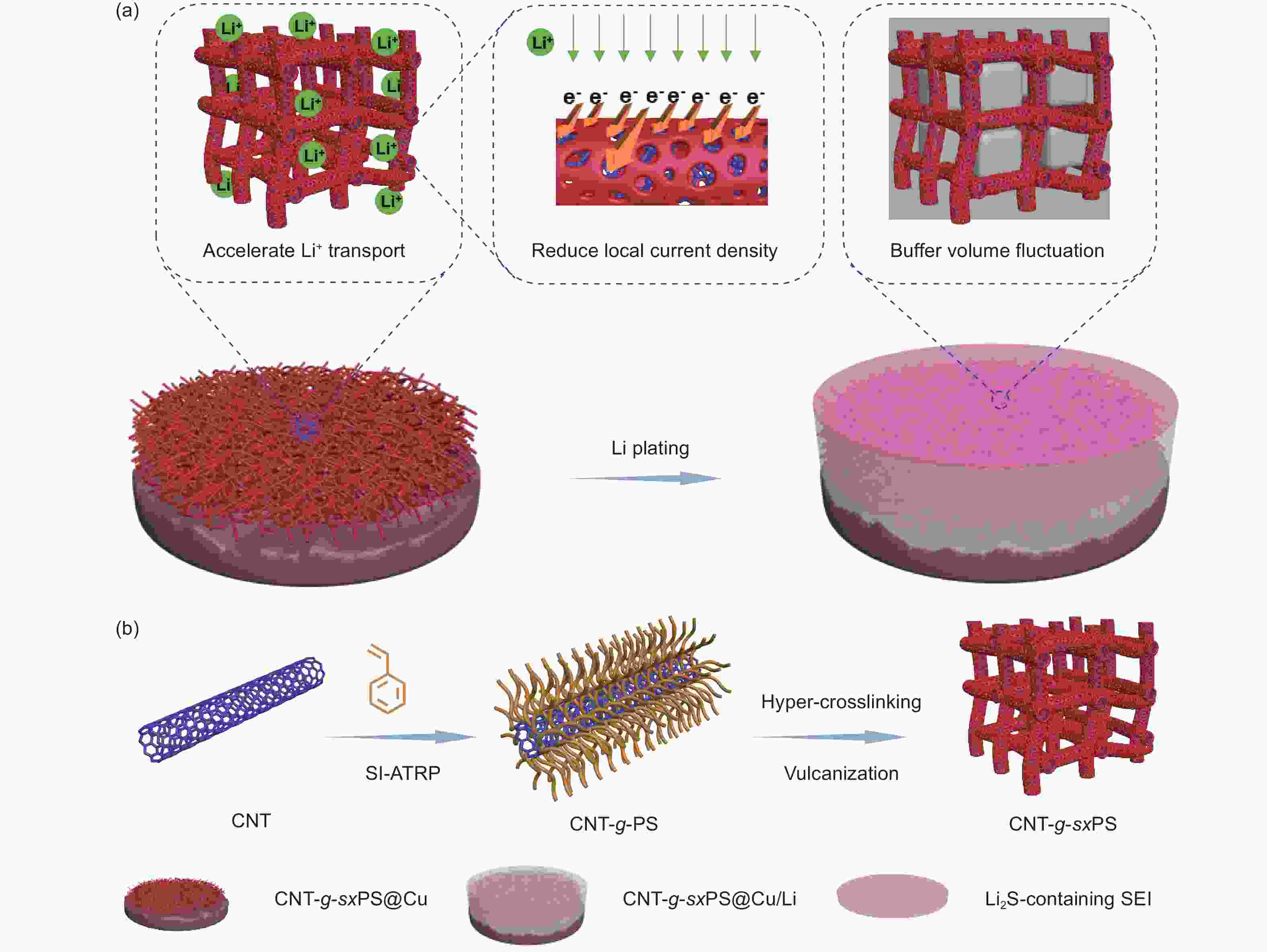
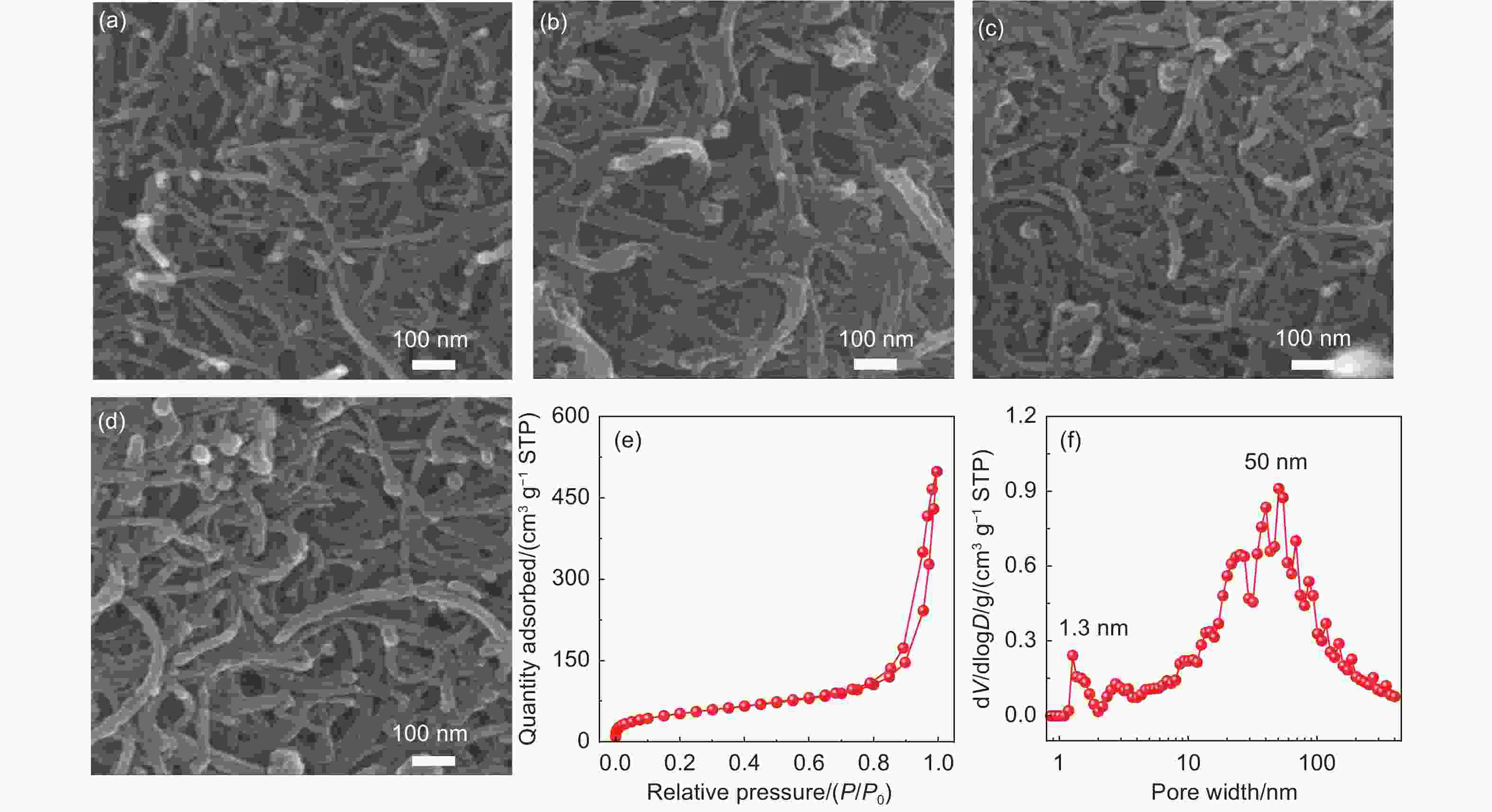
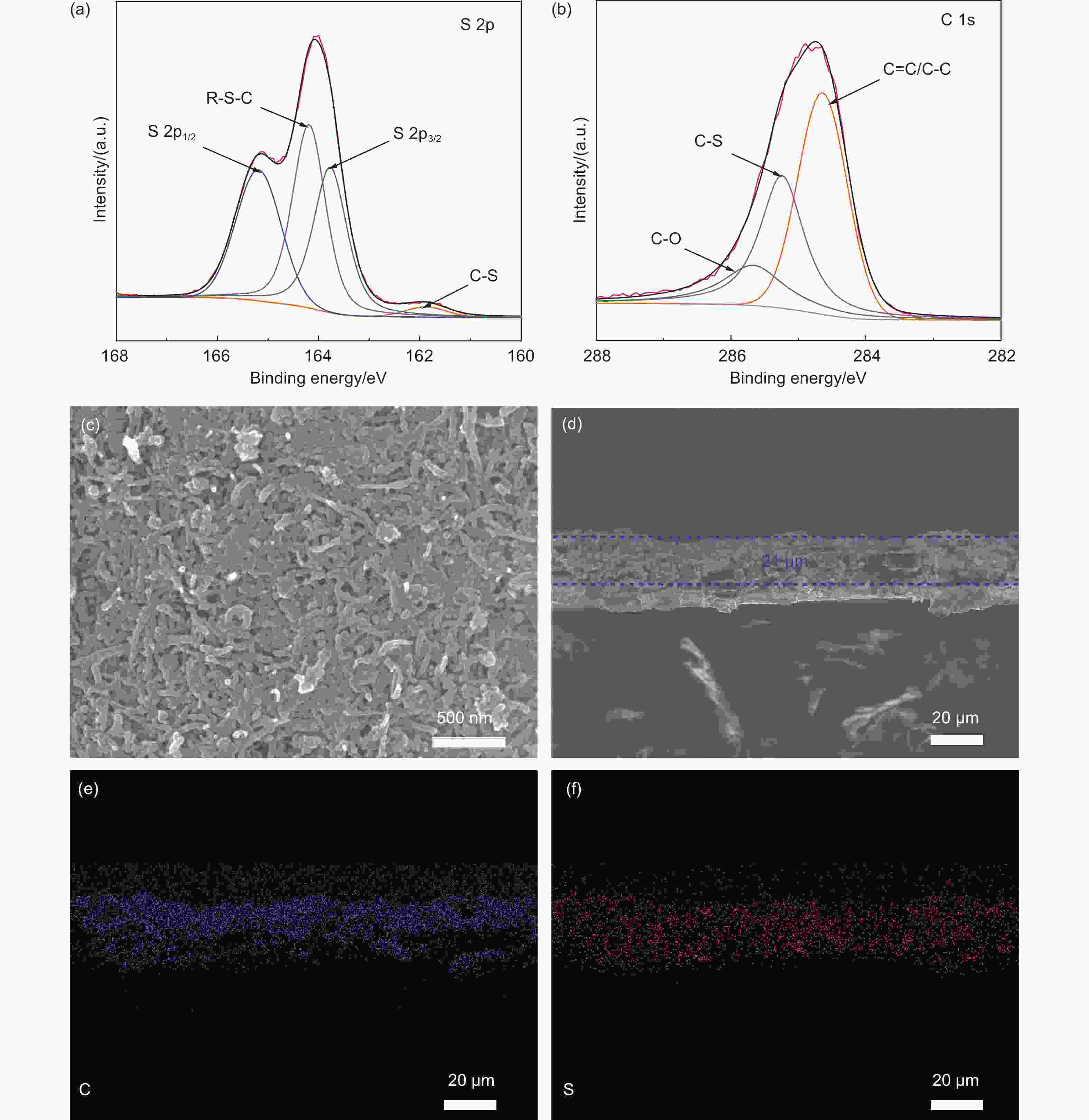
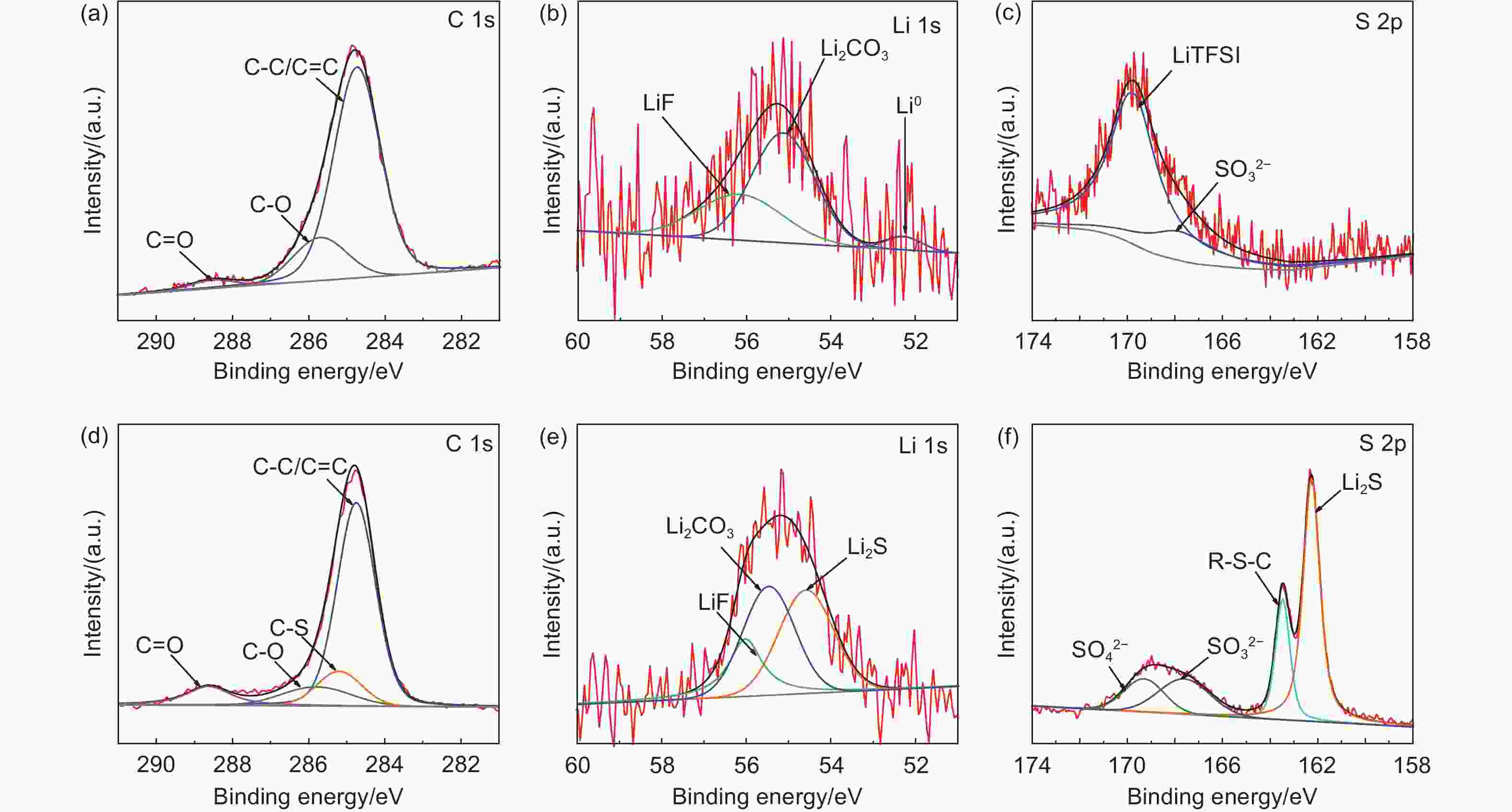
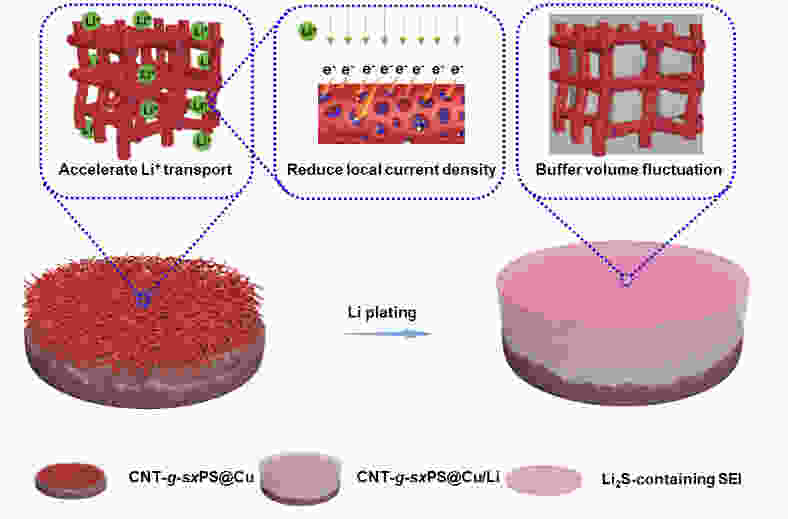
Fabrication of vulcanized cross-linked polystyrene grafted on carbon nanotubes for use as an advanced lithium host
-
摘要: 不可控的锂枝晶生长、严重的体积膨胀以及脆弱的固态电解质中间相(SEI)严重制约了锂金属电池(LMBs)的实际应用。在本研究中,作者成功设计合成了一类具有碳纳米管基底的硫化超交联聚苯乙烯刷(CNT-g-sxPS),并将其用作新型的三维锂金属载体。CNT-g-sxPS的层次化大孔、中孔和微孔能够促进锂离子的传输,缓解锂负极的体积变化,提供高比表面积以降低局部电流密度,从而实现快速且均匀的锂沉积/剥离。同时,孔骨架表面均匀分布的含硫基团可以与锂原位反应生成含Li2S的SEI,有利于构筑稳定的负极/电解液界面。此外,碳纳米管基底还能提供快速的电子传输路径。因此,利用CNT-g-sxPS负载的锂金属负极(CNT-g-sxPS@Cu/Li)组装的Li|Li对称电池在1 mA cm−2、1 mAh cm−2下可稳定循环超过500 h。当与磷酸铁锂正极(LFP)匹配时,利用CNT-g-sxPS@Cu/Li负极组装的全电池在1 C下循环600圈后仍然具有101 mAh g−1的放电比容量,容量保持率为77%。
-
关键词:
- 硫化超交联聚苯乙烯刷 /
- 碳纳米管基底 /
- 三维锂载体 /
- 含有Li2S的SEI /
- 锂金属电池
Abstract: We report the fabrication of vulcanized cross-linked polystyrene grafted on carbon nanotubes (CNTs) for use as an advanced three-dimensional Li host. First, polystyrene was grafted from Br-modified CNTs to form brush-like structure by surface-initiated atom-transfer radical polymerization. Polystyrene grafted on carbon nanotubes was then cross-linked using a Friedel-Crafts reaction and finally vulcanized with sulfur. Vulcanized cross-linked polystyrene grafted on carbon nanotubes was used as a support for the Li metal, and its macro-, meso- and microporous structure increased Li ion transport, buffered the volume changes of the Li anode, and provided a high specific surface area to reduce local current density, which assisted rapid and uniform Li plating/stripping. At the same time, the homogenously distributed sulfur in the support reacted with Li to produce a Li2S-containing SEI layer, while the CNTs provided conductive pathways for the rapid transmission of electrons. As a result, a Li|Li symmetric cell using this anode material and a Cu current collector had a stable cycling performance of more than 500 h at a current density of 1 mA cm−2. When LiFePO4 was used as the cathode, a full cell had a high discharge capacity of 101 mAh g−1 with a capacity retention of 77% after 600 cycles at 1 C. -
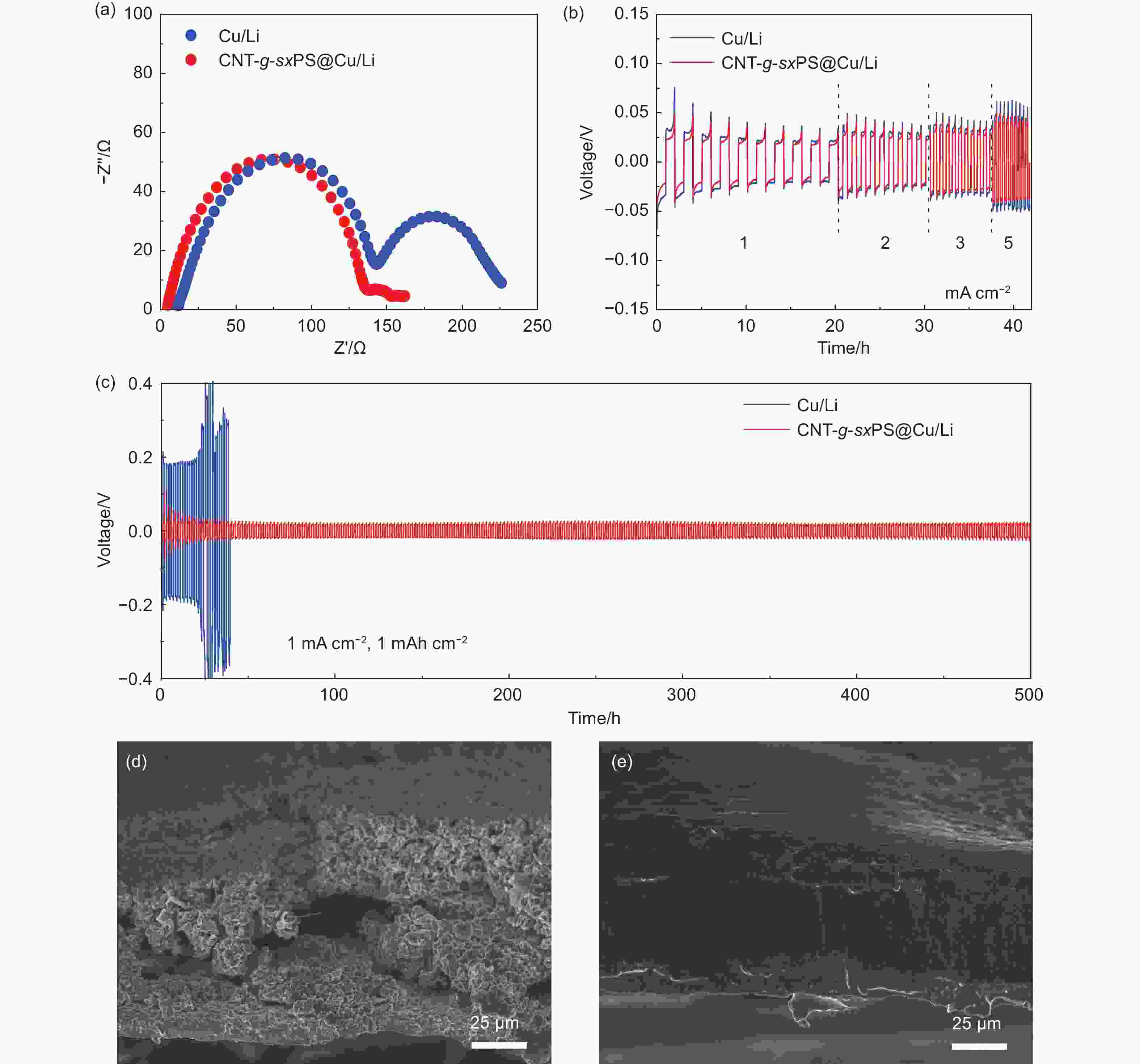
Figure 5. (a) Nyquist plots of Li|Li symmetric cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes. (b) Voltage profiles of symmetric Li|Li cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes at various current densities with a fixed capacity of 1 mAh cm−2. (c) Voltage profiles of Li|Li symmetric cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes at 1 mA cm−2 and 1 mAh cm−2. Cross-section SEM images of (d) bare Cu/Li and (e) CNT-g-sxPS@Cu/Li anodes after Li|Li symmetric cell tests for 50 cycles at 1 mA cm−2 and 1 mAh cm−2
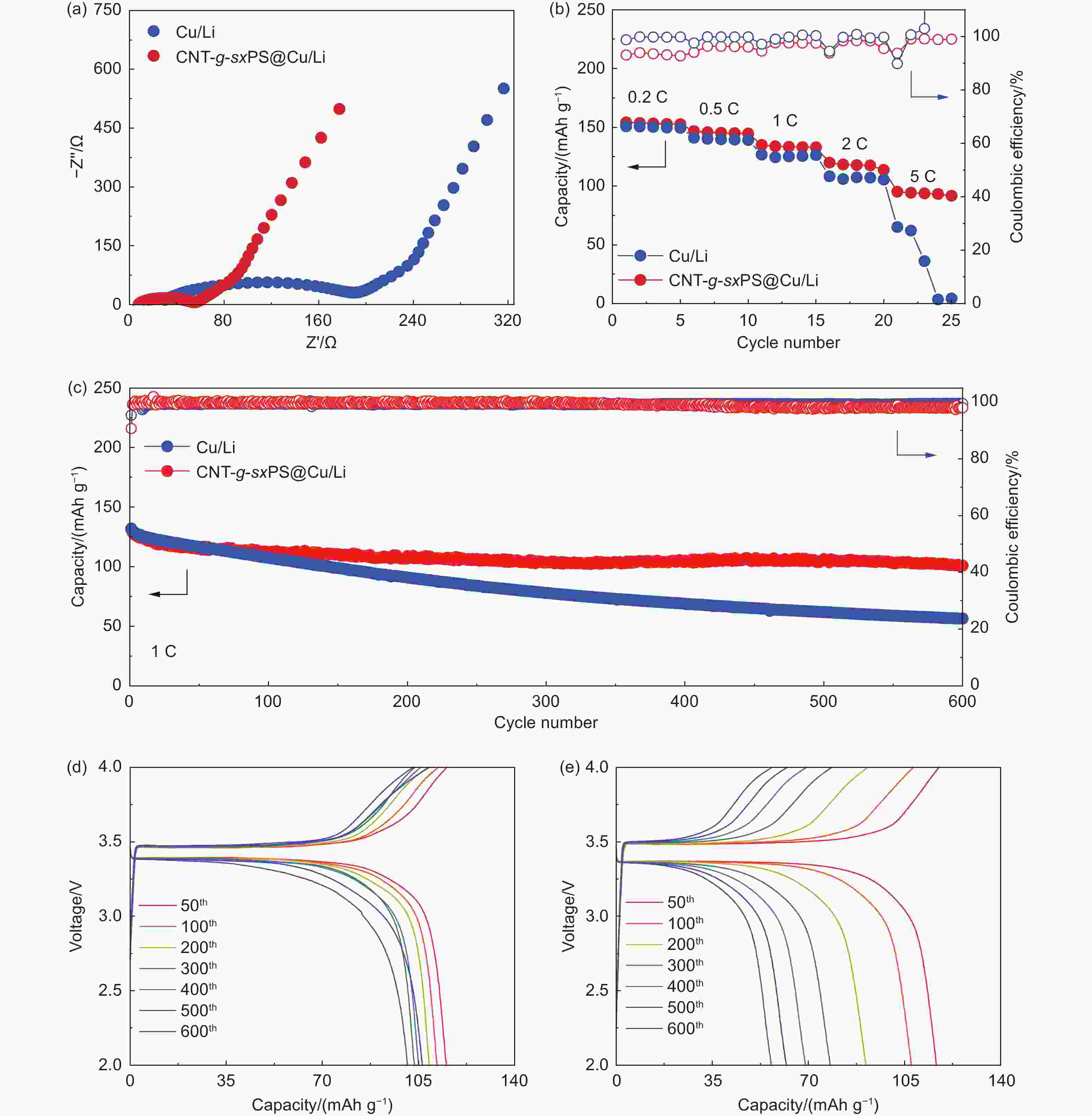
Figure 6. (a) Nyquist plots of Li|LFP full cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes. (b) Rate performance of Li|LFP full cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes. (c) Cycling performance of Li|LFP full cells with CNT-g-sxPS@Cu/Li and bare Cu/Li anodes at 1 C. Galvanostatic charge-discharge profiles of Li|LFP cells with (d) CNT-g-sxPS@Cu/Li and (e) bare Cu/Li anodes
-
[1] Tarascon J, Armand M. Issues and challenges facing rechargeable lithium batteries[J]. Nature,2001,414(6861):359-367. doi: 10.1038/35104644 [2] Zhang X, Yang Y, Zhou Z. Towards practical lithium-metal anodes[J]. Chemical Society Review,2020,49(10):3040-3071. doi: 10.1039/C9CS00838A [3] Li S, Huang J, Cui Y, et al. A robust all-organic protective layer towards ultrahigh-rate and large-capacity Li metal anodes[J]. Nature Nanotechnology,2022,17(6):613-621. doi: 10.1038/s41565-022-01107-2 [4] Chen X, Zhao B, Yan C, et al. Review on Li deposition in working batteries: from nucleation to early growth[J]. Advanced Materials,2021,33(8):e2004128. doi: 10.1002/adma.202004128 [5] Li C, Liu S, Shi C, et al. Two-dimensional molecular brush-functionalized porous bilayer composite separators toward ultrastable high-current density lithium metal anodes[J]. Nature Communications,2019,10(1):1363. doi: 10.1038/s41467-019-09211-z [6] He X, Bresser D, Passerini S, et al. The passivity of lithium electrodes in liquid electrolytes for secondary batteries[J]. Nature Reviews Materials,2021,6(11):1036-1052. doi: 10.1038/s41578-021-00345-5 [7] Liu Q, Liu R, Cui Y, et al. Dendrite-free and long-cycling lithium metal battery enabled by ultrathin, 2D shield-defensive, and single lithium-ion conducting polymeric membrane[J]. Advanced Materials,2022,34(33):e2108437. doi: 10.1002/adma.202108437 [8] Li S, Zhang L, Liu T, et al. A dendrite-free lithium-metal anode enabled by designed ultrathin MgF2 nanosheets encapsulated inside nitrogen-doped graphene-like hollow nanospheres[J]. Advanced Materials,2022,34(24):e2201801. doi: 10.1002/adma.202201801 [9] Fang Y, Zhang S, Wu Z, et al. A highly stable lithium metal anode enabled by Ag nanoparticle-embedded nitrogen-doped carbon macroporous fibers[J]. Science Advances,2021,7(21):eabg3626. doi: 10.1126/sciadv.abg3626 [10] Mei Y, Zhou J, Hao Y, et al. High-lithiophilicity host with micro/nanostructured active sites based on wenzel wetting model for dendrite‐free lithium metal anodes[J]. Advanced Functional Materials,2021,31(50):2106676. doi: 10.1002/adfm.202106676 [11] Lu Z, Liu S, Li C, et al. 3D porous carbon networks with highly dispersed SiOx by molecular-scale engineering toward stable lithium metal anodes[J]. Chemical Communication,2019,55(43):6034-6037. doi: 10.1039/C9CC01927H [12] Chen C, Guan J, Li N, et al. Lotus-root-like carbon fibers embedded with Ni-Co nanoparticles for dendrite-free lithium metal anodes[J]. Advanced Materials,2021,33(24):e2100608. doi: 10.1002/adma.202100608 [13] Huang G, Han J, Zhang F, et al. Lithiophilic 3D nanoporous nitrogen-doped graphene for dendrite-free and ultrahigh-rate lithium-metal anodes[J]. Advanced Materials,2019,31(2):e1805334. doi: 10.1002/adma.201805334 [14] Wang J, Li L, Hu H, et al. Toward dendrite-free metallic lithium anodes: from structural design to optimal electrochemical diffusion kinetics[J]. ACS Nano,2022,16(11):17729-17760. doi: 10.1021/acsnano.2c08480 [15] Fan L, Zhuang H, Zhang W, et al. Stable lithium electrodeposition at ultra-high current densities enabled by 3D PMF/Li composite Anode[J]. Advanced Energy Materials,2018,8(15):1703360. doi: 10.1002/aenm.201703360 [16] Hu Z, Deng W, He B, et al. Self-adaptive 3D skeleton with charge dissipation capability for practical Li metal pouch cells[J]. Nano Energy, 2022, 93(106805). [17] Ma Y, Wei L, He Y, et al. A "blockchain" synergy in conductive polymer-filled metal-organic frameworks for dendrite-free Li plating/stripping with high coulombic efficiency[J]. Angewandte Chemie International Edition,2022,61(12):e202116291. [18] Yang Z, Liu W, Chen Q, et al. Ultrasmooth and dense lithium deposition toward high-performance lithium-metal batteries[J]. Advanced Materials, 2023: e2210130. [19] Liang Y, Chen L, Zhuang D, et al. Fabrication and nanostructure control of super-hierarchical carbon materials from heterogeneous bottlebrushes[J]. Chemical Science,2017,8(3):2101-2106. doi: 10.1039/C6SC03961H [20] Adams B, Zheng J, Ren X, et al. Accurate determination of coulombic efficiency for lithium metal anodes and lithium metal batteries[J]. Advanced Energy Materials,2018,8(7):1702097. doi: 10.1002/aenm.201702097 [21] Chen G, Liu S, Chen S, et al. FTIR spectra, thermal properties, and dispersibility of a polystyrene/montmorillonite nanocomposite[J]. Macromolecular Chemistry and Physics,2001,202(7):1189-1193. doi: 10.1002/1521-3935(20010401)202:7<1189::AID-MACP1189>3.0.CO;2-M [22] Li Z, Wu D, Liang Y, et al. Synthesis of well-defined microporous carbons by molecular-scale templating with polyhedral oligomeric silsesquioxane moieties[J]. Journal of the American Chemical Society,2014,136(13):4805-4808. doi: 10.1021/ja412192v [23] Wu J, Huang J, Cui Y, et al. Rough endoplasmic reticulum inspired polystyrene-brush-based superhigh sulfur content cathodes enable lithium-sulfur cells with high mass and capacity loading[J]. Advanced Materials, 2023: e2211471. [24] Wu P, Sun M, Yu Y, et al. Physical and chemical dual-confinement of polysulfides within hierarchically meso-microporous nitrogen-doped carbon nanocages for advanced Li–S batteries[J]. RSC Advances,2017,7(68):42627-42633. doi: 10.1039/C7RA07918D [25] Chua C & Pumera M. Monothiolation and reduction of graphene oxide via one-pot synthesis: Hybrid catalyst for oxygen reduction[J]. ACS Nano,2015,9(4):4193-4199. doi: 10.1021/acsnano.5b00438 [26] Hu G, Sun Z, Shi C, et al. A sulfur-rich copolymer@CNT hybrid cathode with dual-confinement of polysulfides for high-performance lithium-sulfur batteries[J]. Advanced Materials,2017,29(11):1603835. doi: 10.1002/adma.201603835 [27] Munir A, Haq T, Qurashi A, et al. Ultrasmall Ni/NiO nanoclusters on thiol-functionalized and -exfoliated graphene oxide nanosheets for durable oxygen evolution reaction[J]. ACS Applied Energy Materials,2018,2(1):363-371. [28] Zhang T, Li J, Li X, et al. A silica-reinforced composite electrolyte with greatly enhanced interfacial lithium-ion transfer kinetics for high-performance lithium metal batteries[J]. Advanced Materials,2022,34(41):2205575. doi: 10.1002/adma.202205575 [29] Liang J, Li X, Zhao Y, et al. In situ Li3PS4 solid-state electrolyte protection layers for superior long-life and high-rate lithium-metal anodes[J]. Advanced Materials,2018,30(45):1804684. doi: 10.1002/adma.201804684 [30] Wang W, Yang Z, Zhang Y, et al. Highly stable lithium metal anode enabled by lithiophilic and spatial-confined spherical-covalent organic framework[J]. Energy Storage Materials, 2022, 46(374-383). [31] Tian M, Ben L, Yu H, et al. Designer cathode additive for stableinterphases on high-energy anodes[J]. Journal of the American Chemical Society,2022,144(33):15100-15110. doi: 10.1021/jacs.2c04124 -
 Supporting Information.pdf
Supporting Information.pdf

-





 下载:
下载: