A review of synthesis method and application of MXenes as host in lithium metal anodes
-
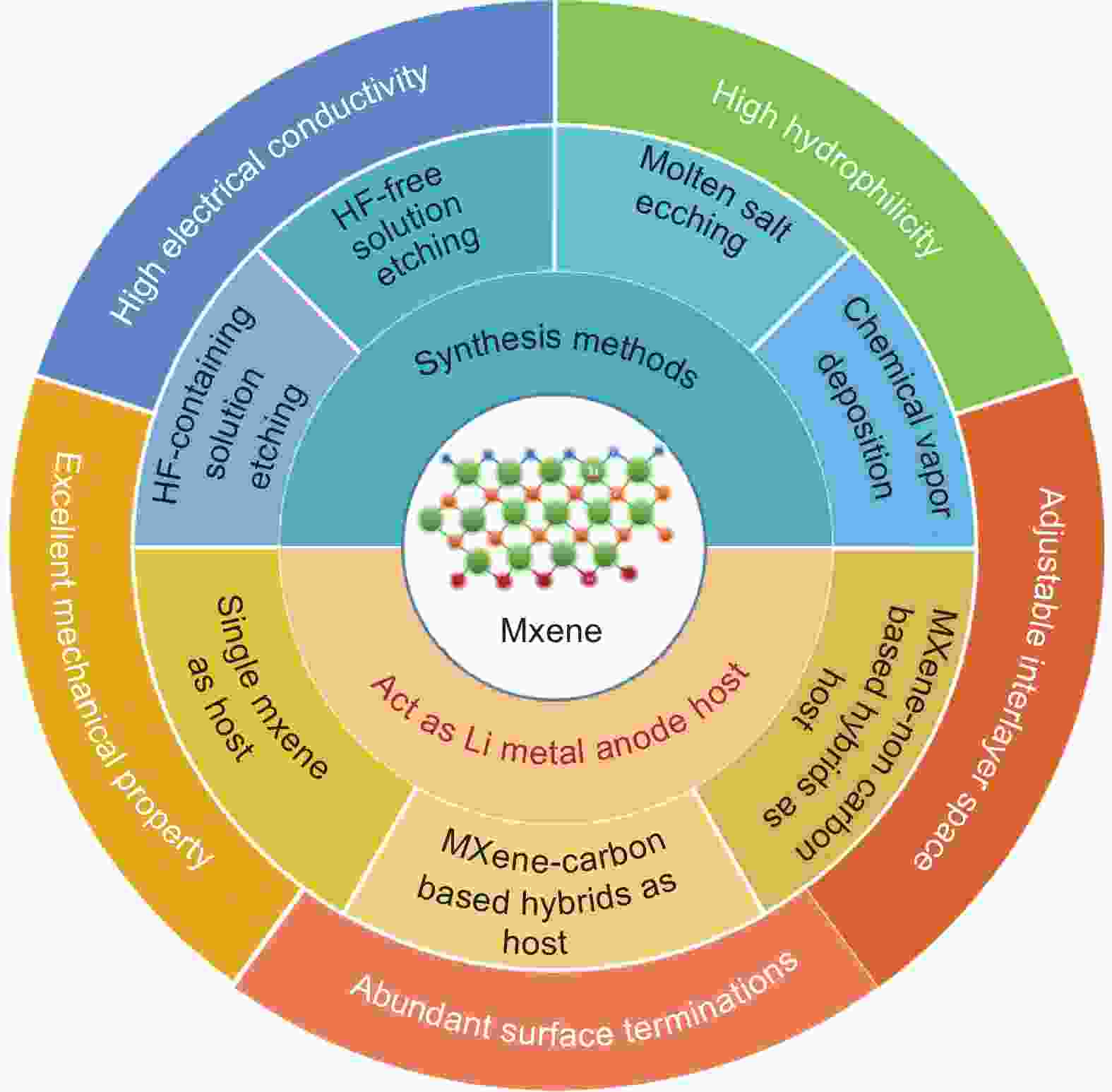
摘要: 锂金属直接用作负极时,在循环过程中面临枝晶生长和体积膨胀的问题,导致固态电解质界面(SEI)层断裂和重复形成,消耗活性物质和电解质,进而降低电池的库仑效率并导致容量快速衰减。设计具有快速传质和足够存储空间的基体是促进锂的均匀沉积、减少SEI重复生长和死锂形成的有效方法。具有二维层状结构的MXenes由于具有优异的导电性、可调控的层间距、丰富的亲锂表面官能团和优异的机械性能而被认为是良好的锂金属宿主。本综述首先总结了MXenes的多种合成方法,包括借助外部试剂蚀刻前驱体MAX相、化学气相沉积、UV诱导蚀刻和机械化学等方法。不同的合成方法会形成不同表面官能团和层状结构的MXene,进而影响锂金属的成核和生长行为。随后,介绍了纯MXene,MXene-碳杂化物和MXene-非碳杂化物在锂金属负极宿主中的应用,主要关注其缓解锂金属负极体积变化并抑制锂枝晶生长方面的性能。最后,对一些改性策略和潜在的研究方向进行了总结和展望。Abstract: Severe dendritic growth and volume expansion are easily induced during the cycling process when lithium metal is used as an anode electrode directly. These problems cause the solid electrolyte interface (SEI) layer to break and re-form, which consumes the active lithium metal and electrolyte, thereby reducing the Coulomb efficiency and rapid capacity. Designing a host matrix with rapid mass transfer and enough storage space to promote lithium homogeneous deposition, hence reducing the repeated SEI growth and the formation of dead lithium, is an effective method to address the concerns mentioned above issues. MXenes with two-dimensional layered structures have been regarded as feasible hosts for stabilizing lithium due to their superior electrical conductivity, sizeable interlayer space, abundant lithiophilic surface functional groups, and excellent mechanical properties. In this review, we first summarized the multiple synthesis methods of MXenes, including etching the precursor MAX phase, chemical vapor deposition, UV-induced etching, and mechanochemical et al. Various synthesis methods would induce different surface termination and lamellar structures, affecting lithium metal nucleation and growth behavior. Subsequently, pure MXene, MXene-carbon and MXene-non carbon hybrid compounds applied for lithium metal anode hosts were introduced, focusing on alleviating noticeable volume changes and inhibiting lithium dendrite growth. Finally, some modification strategies and potential research prospects were summarized and prospected.
-
Key words:
- MXene /
- 2D materials /
- Synthetize /
- Lithium anodes /
- Carbon hybridization
-
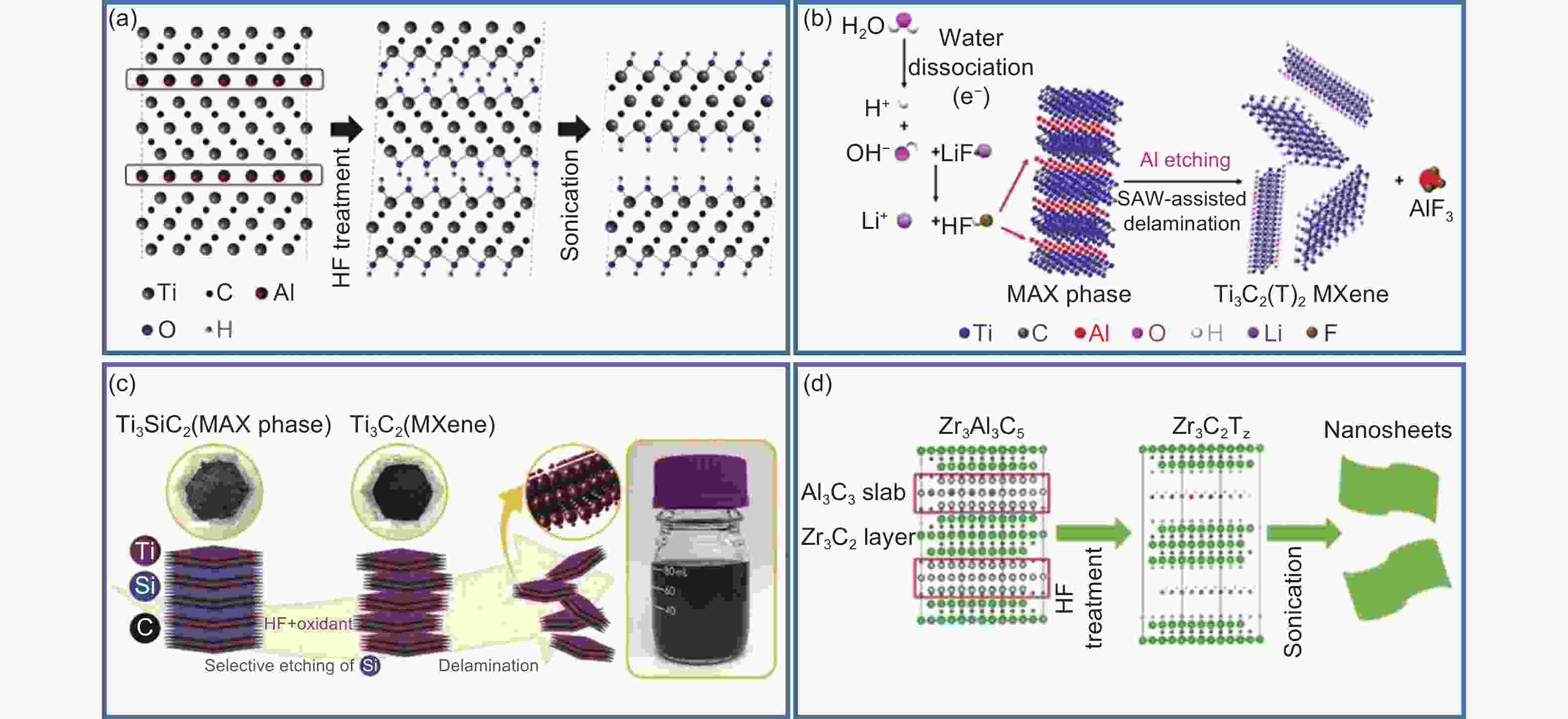
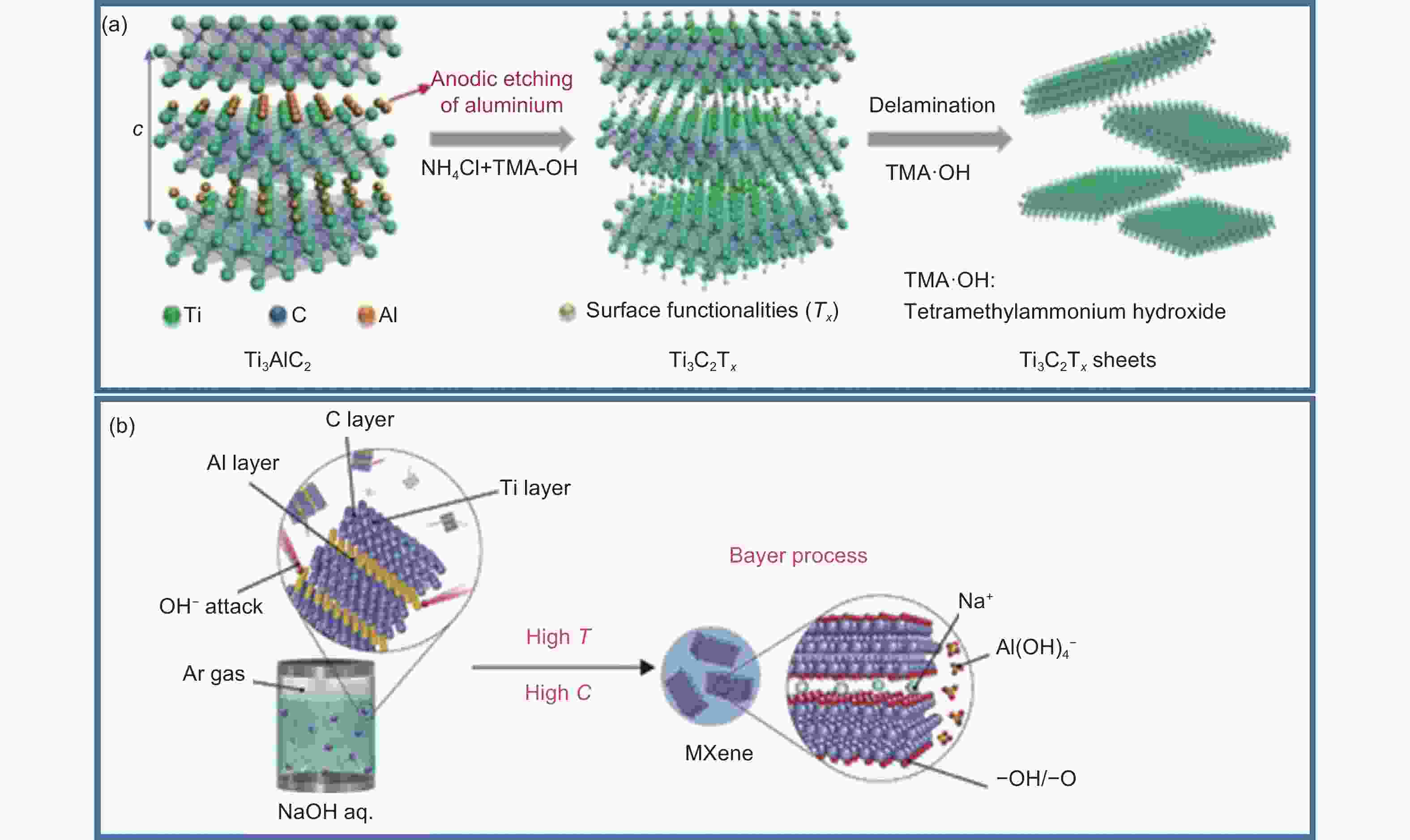
Figure 2. (a) Schematic of the preparation of MXene by HF acid solution etching[53]. (b) Schematic of the synthesis of Ti3C2(T)z MXene through selective etching with HF generated in-situ protons and LiF[57]. (c) Schematic of etching Si to synthesize MXene[62]. (d) Schematic of using HF to selectively remove Al3C3 for preparing Ti-free based Zr3C2Tz[63]. Reprinted with permission
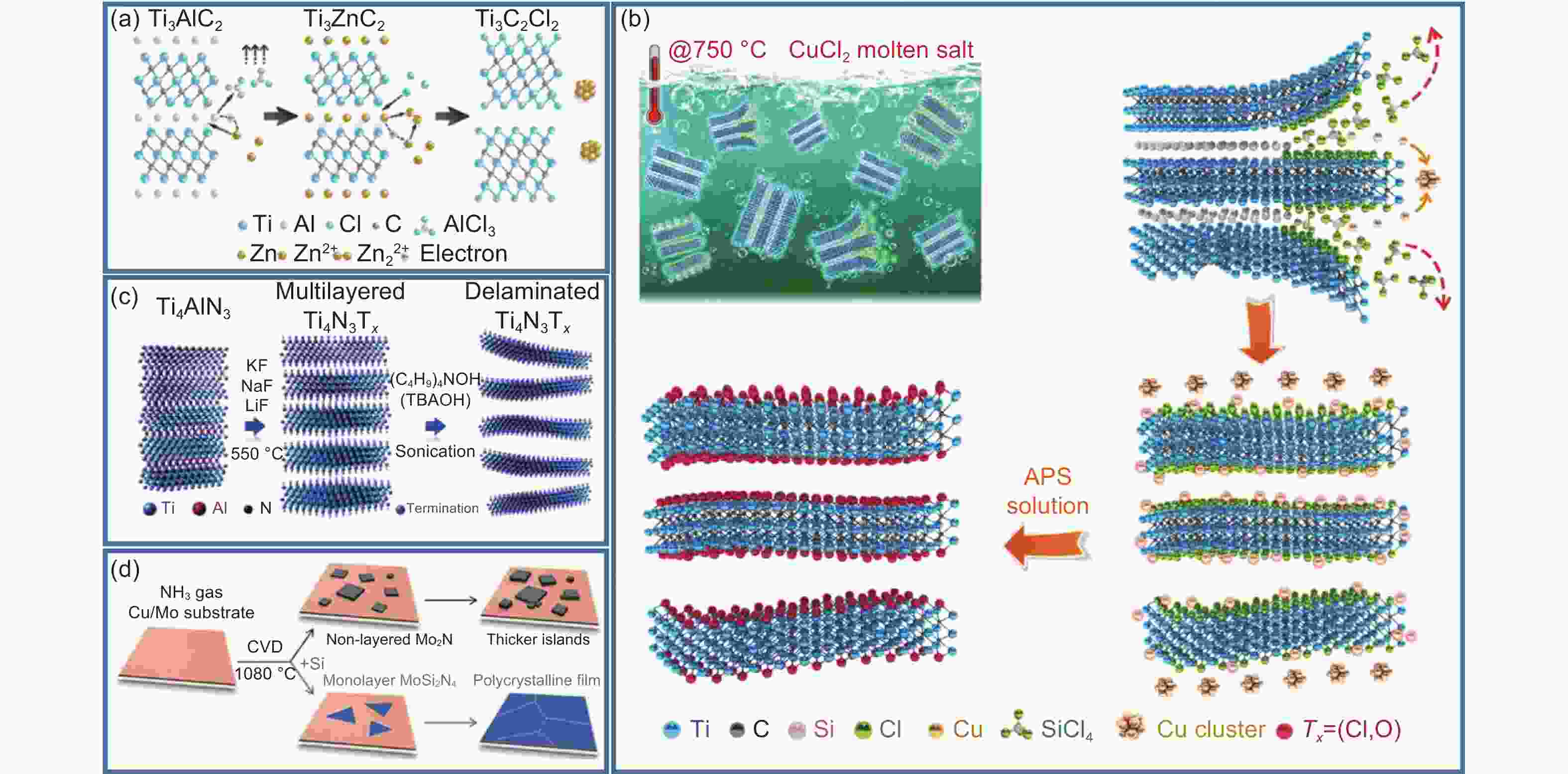
Figure 4. (a) Schematic of synthesizing Ti3AlC2 in ZnCl2 molten salt[67]. (b) Schematic of MXene preparation by oxidation-reduction method of cation coupling of element A and Lewis acid molten salt[68]. (c) Schematic of preparing Ti4N3-based MXene[69] (d) Schematic of introducing Si source to prepare single layer MoSi2N4 in CVD process[74]. (Reprinted with permission)
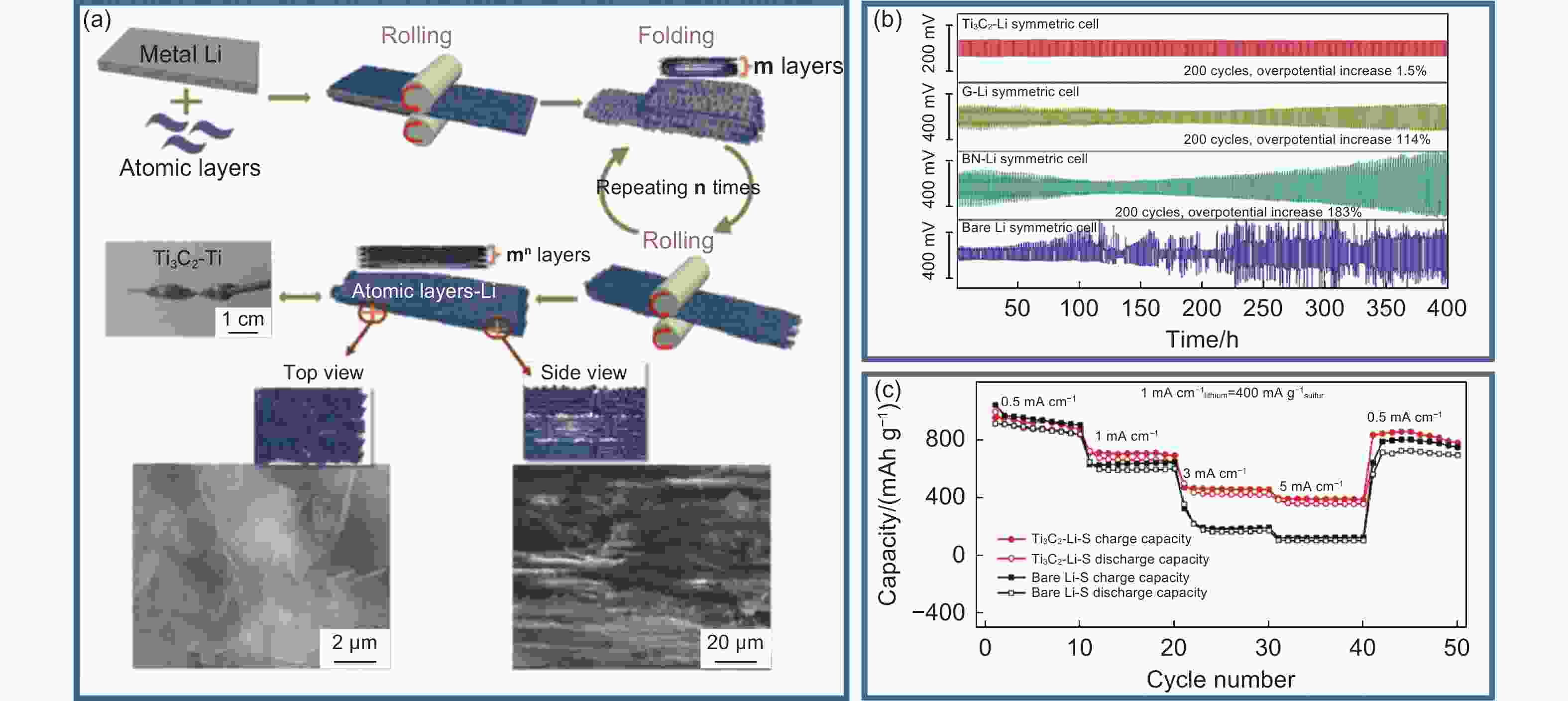
Figure 5. (a) Preparation flow chart of Ti3C2 MXene Li composite anode[41]. (b) Cyclic performance of symmetrical batteries at constant current density of 1.0 mA cm−2[41]. (c) The rate capabilities of Li-S full cells at various current densities from 0.5 to 5 mA cm−2[41]. (Reprinted with permission)
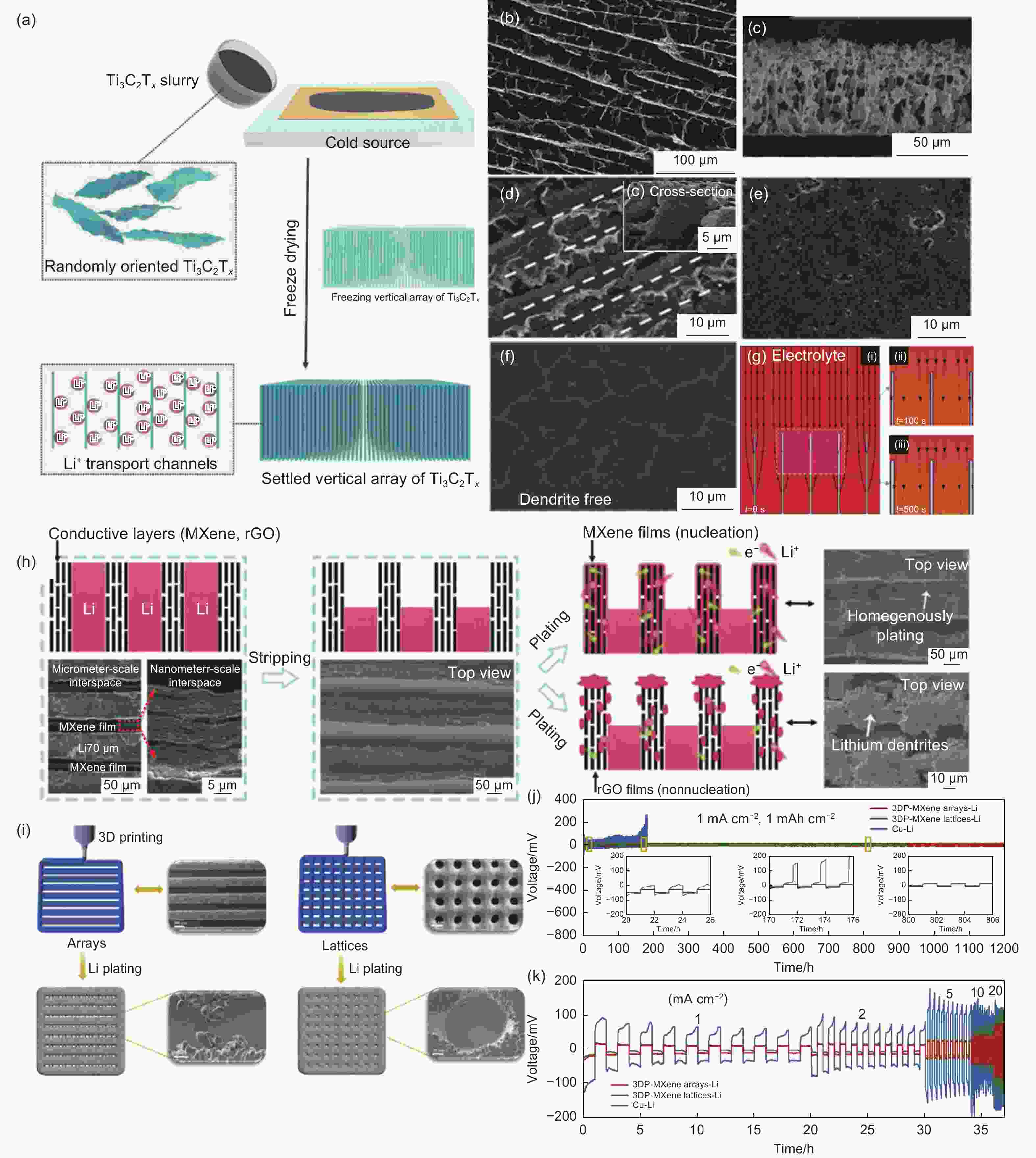
Figure 6. (a) Schematic of preparing v-Ti3C2Tx nanosheet arrays using ice template assisted blade coating method. (b) Top view and (c) cross-section SEM image of v-Ti3C2Tx electrodes; SEM characterization of v-Ti3C2Tx electrodes after plating different capacities lithium at 1.0 mA cm−2: (d) 1.0 mAh cm−2. (e) 3.0 mAh cm−2 (f) 6.0 mAh cm−2. (g) COMSOL simulation of Li deposition on v-Ti3C2Tx electrodes[78]. (h) Schematic diagram of peeling and plating states of vertical MXene Li and rGO Li arrays[79]. (j) Schematic diagram of 3D printing MXene array and lattice[80]. (k) Cycling performances of symmetric cells at 1 mA cm−2, 1 mAh cm−2[80]. (m) Rate capabilities from 1 to 20 mA cm−2[80]. (Reprinted with permission)
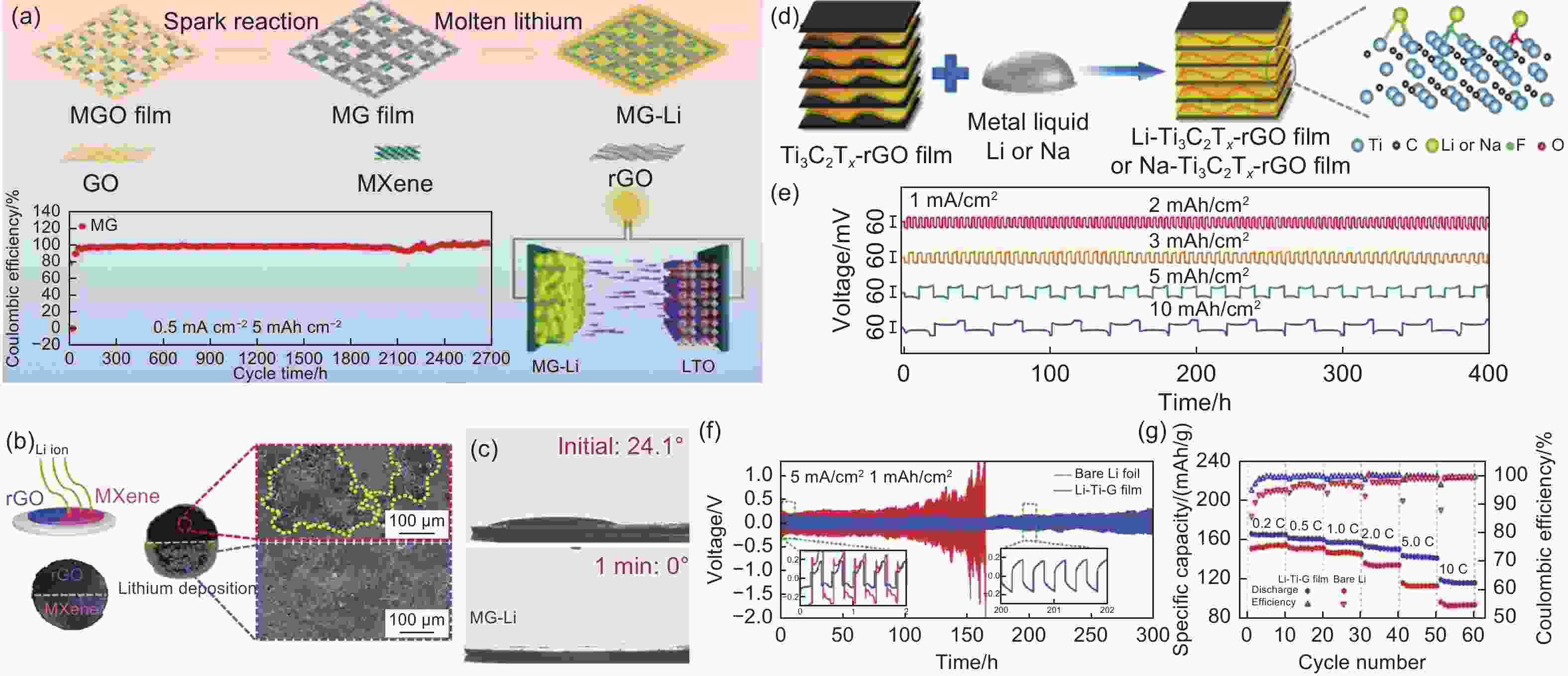
Figure 7. (a) Schematic of M/G aerogel prepared by thermal infusion strategy process and Coulombic efficiency of MG electrode at a current density of 0.5 mA cm−2 with 5 mAh cm−2[81]. (b) Photos and top SEM images of Li deposition on rGO coated MXene and uncoated MXene[81]. (c) The contact angle of electrolyte droplets on MG-Li at the initial time and after 1 min[81]. (d) Schematic illustration of Li-Ti3C2Tx-rGO preparation[82]. (e) Cycling performances for Li- Ti3C2Tx-rGO films of stripping/plating capacity of 2, 3, 5 and 10 mAh cm−2 at 1 mA cm−2 [82]. (f) Cycling of symmetric Li-Ti3C2Tx-rGO electrodes and bare Li foils for more than 300 h at a current density of 5 mA·cm−2. (g) Rate capability of LFP||Li- Ti3C2Tx-rGO and LFP||bare-Li cells from 0.2 to 10 C[82]. (Reprinted with permission)
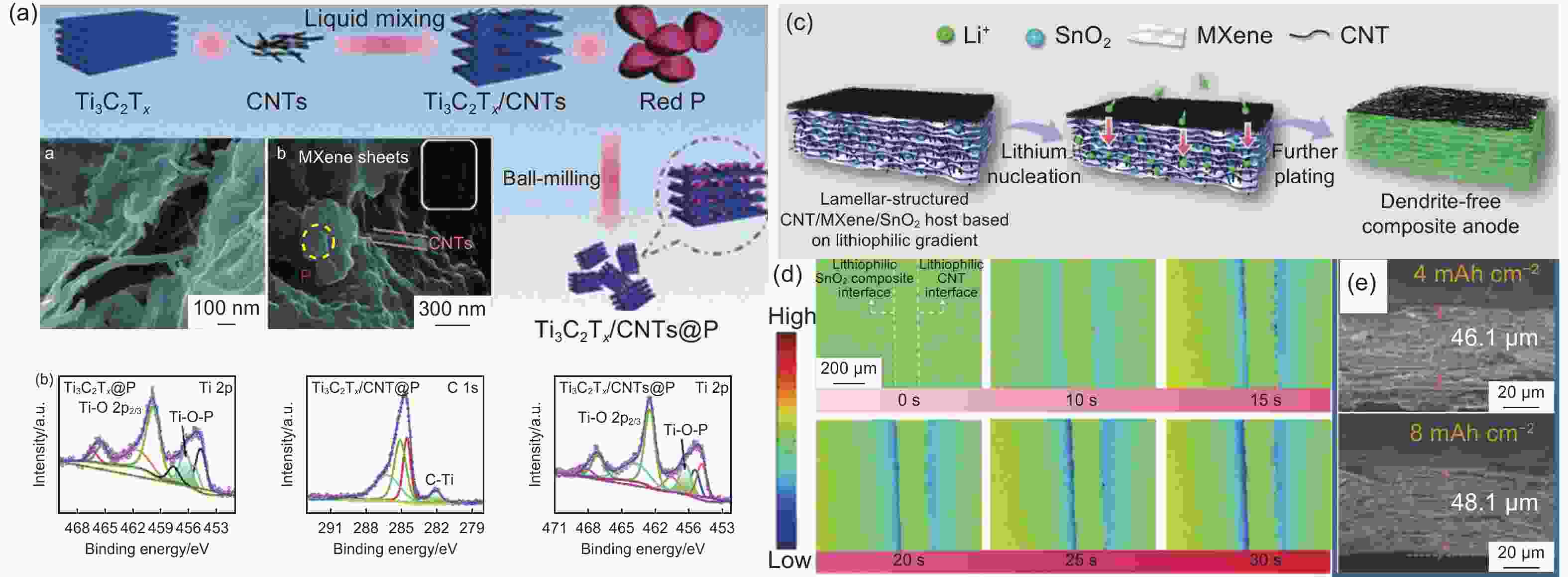
Figure 8. (a) Schematic of Ti3C2Tx/CNTs@P nanohybrid as well as SEM imagines of Ti3C2Tx/CNTs and Ti3C2Tx/CNTs@P at the bottom left[84]. (b) High resolution XPS spectra of Ti and C[84]. (c) Schematic of Li deposition in the CNT/MXene/SnO2 composite host[85]. (d) In-situ digital holographic test images of Li metal deposition process in the CNT/MXene/SnO2 at 10 mA cm−2[85]. (e) SEM morphologies of the CNT/MXene/SnO2 host with Li plating capacities of 4 mAh cm−2 and 8 mAh cm−2[85]. (Reprinted with permission)
Figure 9. TCCNFs foam[86]: (a) Preparation schematic, (b) Top-surface and (c) Cross-sectional SEM images. (d) Li deposition curves at 1 mA cm−2. (e) Multiphysics finite elemental analysis of the electric displacement field. (f) Voltage-Time profiles of symmetric cells at 1 mA cm−2, 1 mAh cm−2 and (g) 5 mA cm−2, 5 mAh cm−2; Ti3C2Tx/CMK-5 composite[87]: (h) Preparation schematic. (i) SEM and (j) TEM images. (k) Cycling stability at 1 C. (Reprinted with permission)
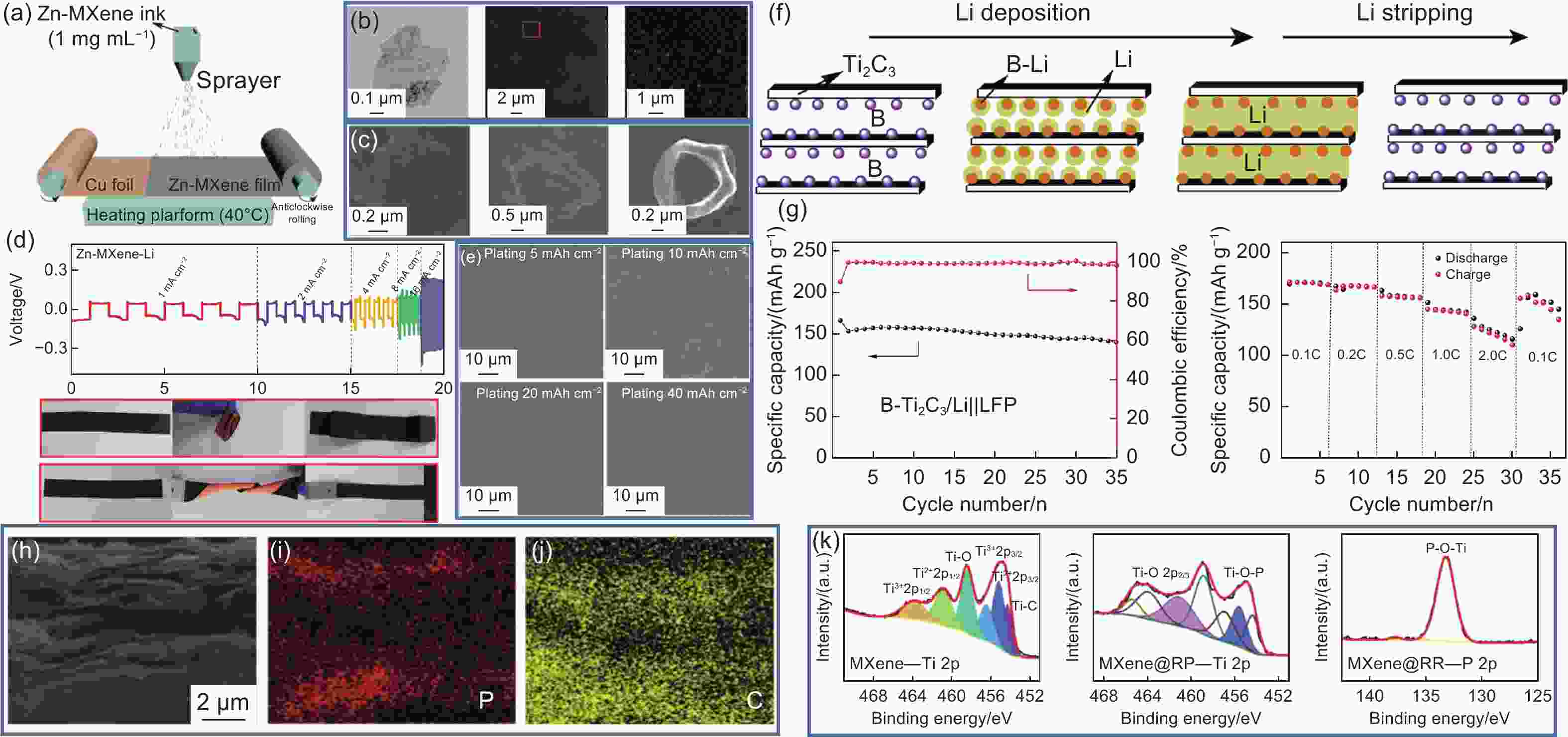
Figure 10. Zn-MXene nanosheets[88]: (a) Manufacturing schematic. (b) TEM and HAADF-STEM images. (c) SEM images after Li plating with various capacities. (d) Rate capabilities of the Zn-MXene-Li anodes at various current densities (1-16 mA cm−2) and folding and twisting tests. (e) Typical SEM images of Zn-MXene-Li anode at different Li plating levels. (f) Schematic of lithium deposition and stripping process on B-doped Ti3C2TxLi electrode[90]. (g) Cycling performance at 0.5 C and rate of full cells assembled with LFP cathode and B-doped Ti3C2Tx@Li anode[90]. (h) SEM and (i, j) corresponding EDS mapping images of MXene@RP paper. (m) High-resolution XPS spectra of MXene-Ti 2p, MXene@RP-Ti 2p and MXene@RP-P 2p[92]. (Reprinted with permission)
-
[1] Winter, M, Barnett, B, Xu, K. Before Li ion batteries[J]. Chemical Reviews,2018,118:11433-11456. doi: 10.1021/acs.chemrev.8b00422 [2] Wu F, Liu M Q, Li Y, et al. High-mass-loading electrodes for advanced secondary batteries and supercapacitors[J]. Electrochemical Energy Reviews,2021,4:382-446. doi: 10.1007/s41918-020-00093-0 [3] Zhao J, Lian J, Zhao Z, et al. A review of in-situ techniques for probing active sites and mechanisms of electrocatalytic oxygen reduction reactions[J]. Nano-Micro Letters,2023,15:19. doi: 10.1007/s40820-022-00984-5 [4] Cheng X Q, Li H J, Zhao Z X, et al. The use of in-situ Raman spectroscopy in investigating carbon materials as anodes of alkali metal-ion batteries[J]. New Carbon Materials,2021,36:93-103. doi: 10.1016/S1872-5805(21)60007-0 [5] Cheng X, Bai Q, Li H, et al. Nanoconfined SnS2 in robust SnO2 nanocrystals building heterostructures for stable sodium ion storage[J]. Chemical Engineering Journal,2022,442:136222. doi: 10.1016/j.cej.2022.136222 [6] Li H, Wang X, Zhao Z, et al. Microstructure controlled synthesis of Ni, N-codoped CoP/carbon fiber hybrids with improving reaction kinetics for superior sodium storage[J]. Journal of Materials Science & Technology,2022,99:184-192. doi: 10.1016/j.jmst.2021.05.034 [7] Chu S, Cui Y, Liu N. The path towards sustainable energy[J]. Nature Materials,2017,16:16-22. doi: 10.1038/nmat4834 [8] Trahey L, Brushett F R, Balsara N P, et al. Energy storage emerging: A perspective from the joint center for energy storage research[J]. Proceedings of the National Academy of Sciences,2020,117:12550-12557. doi: 10.1073/pnas.1821672117 [9] Qiao Y, Yang H J, Chang Z, et al. A high-energy-density and long-life initial-anode-free lithium battery enabled by a Li2O sacrificial agent[J]. Nature Energy,2021,6:653-662. doi: 10.1038/s41560-021-00839-0 [10] Tang Y X, Zhang Y Y, Li W L, et al. Rational material design for ultrafast rechargeable lithium-ion batteries[J]. Chemical Society Reviews,2015,44:5926-5940. doi: 10.1039/C4CS00442F [11] Ciez R E, Whitacre J F. The cost of lithium is unlikely to upend the price of Li-ion storage systems[J]. Journal of Power Sources,2016,320:310-313. doi: 10.1016/j.jpowsour.2016.04.073 [12] Huang B, Pan Z F, Su X Y, et al. Recycling of lithium-ion batteries: Recent advances and perspectives[J]. Journal of Power Sources,2018,399:274-286. doi: 10.1016/j.jpowsour.2018.07.116 [13] Placke T, Kloepsch R, Dühnen S, et al. Lithium ion, lithium metal, and alternative rechargeable battery technologies: The odyssey for high energy density[J]. Journal of Solid State Electrochemistry,2017,21:1939-1964. doi: 10.1007/s10008-017-3610-7 [14] Wang C Y, Liu T, Yang X G, et al. Fast charging of energy-dense lithium-ion batteries[J]. Nature,2022,611:485-490. doi: 10.1038/s41586-022-05281-0 [15] Wei C L, Tao Y, Fei H F, et al. Recent advances and perspectives in stable and dendrite-free potassium metal anodes[J]. Energy Storage Materials,2020,30:206-227. doi: 10.1016/j.ensm.2020.05.018 [16] Gao M, Li H, Xu L, et al. Lithium metal batteries for high energy density: Fundamental electrochemistry and challenges[J]. Journal of Energy Chemistry,2021,59:666-687. doi: 10.1016/j.jechem.2020.11.034 [17] Liu B, Zhang J G, Xu W. Advancing lithium metal batteries[J]. Joule,2018,2:833-845. doi: 10.1016/j.joule.2018.03.008 [18] Zhang H G, Ning H, Busbee J, et al. Electroplating lithium transition metal oxides[J]. Science Advances,2017,3:e1602427. doi: 10.1126/sciadv.1602427 [19] Meng Q, Deng B, Zhang H, et al. Heterogeneous nucleation and growth of electrodeposited lithium metal on the basal plane of single-layer graphene[J]. Energy Storage Materials,2019,16:419-425. doi: 10.1016/j.ensm.2018.06.024 [20] Yan X, Lin L, Chen Q, et al. Multifunctional roles of carbon-based hosts for Li-metal anodes: A review[J]. Carbon Energy,2021,3:303-329. doi: 10.1002/cey2.95 [21] Li C, Zhang S, Miao X, et al. Designing lithium argyrodite solid-state electrolytes for high-performance all-solid-state lithium batteries[J]. Batteries & Supercaps,2022,5:e202100288. doi: 10.1002/batt.202100288 [22] Yan C L. Realizing high performance of solid-state lithium metal batteries by flexible ceramic/polymer hybrid solid electrolyte[J]. Rare Metals,2020,39:458-459. doi: 10.1007/s12598-020-01417-1 [23] Alexander G V, Indu M S, Kamakshy S, et al. Development of stable and conductive interface between garnet structured solid electrolyte and lithium metal anode for high performance solid-state battery[J]. Electrochimica Acta,2020,332:135511. doi: 10.1016/j.electacta.2019.135511 [24] Chen K H, Wood K N, Kazyak E, et al. Dead lithium: Mass transport effects on voltage, capacity, and failure of lithium metal anodes[J]. Journal of Materials Chemistry A,2017,5:11671-11681. doi: 10.1039/C7TA00371D [25] Phattharasupakun N, Wutthiprom J, Duangdangchote S, et al. A 3D free-standing lithiophilic silver nanowire aerogel for lithium metal batteries without lithium dendrites and volume expansion: In operando X-ray diffraction[J]. Chemical Communications,2019,55:5689-5692. doi: 10.1039/C9CC01528K [26] Cheng X B, Zhang R, Zhao C Z, et al. Toward safe lithium metal anode in rechargeable batteries: A review[J]. Chemical Reviews,2017,117:10403-10473. doi: 10.1021/acs.chemrev.7b00115 [27] Le T, Liang Q, Chen M, et al. A triple-gradient host for long cycling lithium metal anodes at ultrahigh current density[J]. Small,2020,16:2001992. doi: 10.1002/smll.202001992 [28] Zhang X L, Ruan Z Q, He Q T, et al. Three-dimensional (3D) nanostructured skeleton substrate composed of hollow carbon fiber/carbon nanosheet/ZnO for stable lithium anode[J]. ACS Applied Materials & Interfaces,2021,13:3078-3088. doi: 10.1021/acsami.0c21747 [29] Qiao L, Zhang R, Li Y, et al. Super-assembled hierarchical and stable N-doped carbon nanotube nanoarrays for dendrite-free lithium metal batteries[J]. ACS Applied Energy Materials,2022,5:815-824. doi: 10.1021/acsaem.1c03259 [30] Dong P, Zhang X, Cha Y, et al. Corrigendum to “In situ surface protection of lithium metal anode in lithium-selenium disulfide batteries with ionic liquid-based electrolytes”[J]. Nano Energy,2020,72:104722. doi: 10.1016/j.nanoen.2020.104722 [31] Liu Y, Wu Y, Zheng J, et al. Silicious nanowires enabled dendrites suppression and flame retardancy for advanced lithium metal anodes[J]. Nano Energy,2021,82:105723. doi: 10.1016/j.nanoen.2020.105723 [32] Liu Y Y, Tzeng Y K, Lin D C, et al. An ultrastrong double-layer nanodiamond interface for stable lithium metal anodes[J]. Joule,2018,2:1595-1609. doi: 10.1016/j.joule.2018.05.007 [33] Zheng G Y, Lee S W, Liang Z, et al. Interconnected hollow carbon nanospheres for stable lithium metal anodes[J]. Nature Nanotechnology,2014,9:618-623. doi: 10.1038/nnano.2014.152 [34] Ma Y, Gu Y, He Y, et al. Fast-charging and dendrite-free lithium metal anode enabled by partial lithiation of graphene aerogel[J]. Nano Research,2022,15:9792-9799. doi: 10.1007/s12274-022-4261-2 [35] Qin R, Shan G, Hu M, et al. Two-dimensional transition metal carbides and/or nitrides (MXenes) and their applications in sensors[J]. Materials Today Physics,2021,21:100527. doi: 10.1016/j.mtphys.2021.100527 [36] Zhou C, Zhao X, Xiong Y, et al. A review of etching methods of MXene and applications of MXene conductive hydrogels[J]. European Polymer Journal,2022,167:111063. doi: 10.1016/j.eurpolymj.2022.111063 [37] Iqbal N, Ghani U, Liao W, et al. Synergistically engineered 2D MXenes for metal-ion/Li-S batteries: Progress and outlook[J]. Materials Today Advances,2022,16:100303. doi: 10.1016/j.mtadv.2022.100303 [38] Choi E, Lee J, Kim Y J, et al. Enhanced stability of Ti3C2Tx MXene enabled by continuous ZIF-8 coating[J]. Carbon,2022,191:593-599. doi: 10.1016/j.carbon.2022.02.036 [39] Yin X, Jin J, Chen X, et al. Ultra-wear-resistant MXene-based composite coating via in situ formed nanostructured tribofilm[J]. ACS Applied Materials & Interfaces,2019,11:32569-32576. doi: 10.1021/acsami.9b11449 [40] Ding J, Zhao H, Yu H. Structure and performance insights in carbon dots-functionalized MXene-epoxy ultrathin anticorrosion coatings[J]. Chemical Engineering Journal,2022,430:132838. doi: 10.1016/j.cej.2021.132838 [41] Li B, Zhang D, Liu Y, et al. Flexible Ti3C2 MXene-lithium film with lamellar structure for ultrastable metallic lithium anodes[J]. Nano Energy,2017,39:654-661. doi: 10.1016/j.nanoen.2017.07.023 [42] Ding L, Wei Y, Wang Y, et al. A two-dimensional lamellar membrane: MXene nanosheet stacks[J]. Angewandte Chemie International Edition,2017,56:1825-1829. doi: 10.1002/anie.201609306 [43] Ghosh K, Pumera M. MXene and MoS3− x coated 3D-printed hybrid electrode for solid-state asymmetric supercapacitor[J]. Small Methods,2021,24:2100451. doi: 10.1002/smtd.202100451 [44] Aakyiir M, Tanner B, Yap P L, et al. 3D printing interface-modified PDMS/MXene nanocomposites for stretchable conductors[J]. Journal of Materials Science & Technology,2022,117:174-182. doi: 10.1016/j.jmst.2021.11.048 [45] Zhao F, Zhai P, Wei Y, et al. Constructing artificial SEI layer on lithiophilic MXene surface for high-performance lithium metal anodes[J]. Advanced Science,2022,9:2103930. doi: 10.1002/advs.202103930 [46] Guo D, Ming F, Shinde D B, et al. Covalent assembly of two-dimensional COF-on-MXene heterostructures enables fast charging lithium hosts[J]. Advanced Functional Materials,2021,31:2101194. doi: 10.1002/adfm.202101194 [47] Wei C L, Tao Y, An Y L, et al. Recent advances of emerging 2D MXene for stable and dendrite-free metal anodes[J]. Advanced Functional Materials,2020,30(45):2004613. doi: 10.1002/adfm.202004613 [48] Zhan X X, Si C, Zhou J, et al. MXene and MXene-based composites: Synthesis, properties and environment-related applications[J]. Nanoscale Horizons,2020,5(2):235-258. doi: 10.1039/c9nh00571d [49] Ha S, Kim D, Lim H K, et al. Lithiophilic MXene-guided lithium metal nucleation and growth behavior[J]. Advanced Functional Materials,2021,31(32):2101261. doi: 10.1002/adfm.202101261 [50] Zia A, Cai Z P, Naveed A B, et al. MXene, silicene and germanene: Preparation and energy storage applications[J]. Materials Today Energy,2022,30:101144. doi: 10.1016/j.mtener.2022.101144 [51] Zhang S, Ying H, Huang P, et al. Hierarchical utilization of raw Ti3C2Tx MXene for fast preparation of various Ti3C2Tx MXene derivatives[J]. Nano Research,2022,15(3):2746-2755. doi: 10.1007/s12274-021-3847-4 [52] Lucero N, Vilcarino D, Datta D, et al. The roles of MXenes in developing advanced lithium metal anodes[J]. Journal of Energy Chemistry,2022,69:132-149. doi: 10.1016/j.jechem.2022.01.011 [53] Naguib M, Kurtoglu M, Presser V, et al. Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2[J]. Advanced Materials,2011,23(37):4248-4253. doi: 10.1002/adma.201102306 [54] Wei Y, Zhang P, Soomro R A, et al. Advances in the synthesis of 2D mxenes[J]. Advanced Materials,2021,33(39):2103148. doi: 10.1002/adma.202103148 [55] Alwarappan S, Nesakumar N, Sun D, et al. 2D metal carbides and nitrides (MXenes) for sensors and biosensors[J]. Biosensors and Bioelectronics,2022,205(2022):113943. doi: 10.1016/j.bios.2021.113943 [56] Zhang Q K, Zhang Y C, Wei C L, et al. Highly reversible lithium metal-organic battery enabled by a freestanding MXene interlayer[J]. Journal of Power Sources,2022,521:230963. doi: 10.1016/j.jpowsour.2021.230963 [57] Ghazaly A E, Ahmed H, Rezk A R, et al. Ultrafast, one-step, salt-solution-based acoustic synthesis of Ti3C2 MXene[J]. ACS Nano,2021,15:4287-4293. doi: 10.1021/acsnano.0c07242 [58] Feng A, Yu Y, Wang Y, et al. Two-dimensional MXene Ti3C2 produced by exfoliation of Ti3AlC2[J]. Materials & Design,2017,114:161-166. doi: 10.1016/j.matdes.2016.10.053 [59] Halim J, Lukatskaya M R, Cook K M, et al. Transparent conductive two-dimensional titanium carbide epitaxial thin films[J]. Chemistry of Materials,2014,26:2374-2381. doi: 10.1021/cm500641a [60] Liu F F, Zhou A G, Chen J F, et al. Preparation of Ti3C2 and Ti2C MXenes by fluoride salts etching and methane adsorptive properties[J]. Applied Surface Science,2017,416:781-789. doi: 10.1016/j.apsusc.2017.04.239 [61] Alhabeb M, Maleski K, Anasori B, et al. Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene)[J]. Chemistry of Materials,2017,29:7633-7644. doi: 10.1021/acs.chemmater.7b02847 [62] Alhabeb M, Maleski K, Mathis T S, et al. Selective etching of silicon from Ti3SiC2 (MAX) to obtain 2D titanium carbide (MXene)[J]. Angewandte Chemie-International Edition,2018,57:5444-5448. doi: 10.1002/anie.201802232 [63] Zhou J, Zha X H, Chen F Y, et al. A two-dimensional zirconium carbide by selective etching of Al3C3 from nanolaminated Zr3Al3C5[J]. Angewandte Chemie-International Edition,2016,55:5008-5013. doi: 10.1002/anie.201510432 [64] Mashtalir O, Naguib M, Mochalin V N, et al. Intercalation and delamination of layered carbides and carbonitrides[J]. Nature Communications,2013,4:1716. doi: 10.1038/ncomms2664 [65] Yang S, Zhang P P, Wang F X, et al. Fluoride-free synthesis of two-dimensional titanium carbide (MXene) using a binary aqueous system[J]. Angewandte Chemie-International Edition,2018,57:15491-15495. doi: 10.1002/anie.201809662 [66] Li T F, Yao L L, Liu Q L, et al. Fluorine-free synthesis of high-purity Ti3C2Tx (T=OH, O) via alkali treatment[J]. Angewandte Chemie-International Edition,2018,57:6115-6119. doi: 10.1002/anie.201800887 [67] Li M, Lu J, Luo K, et al. Element replacement approach by reaction with lewis acidic molten salts to synthesize nanolaminated MAX phases and MXenes[J]. Journal of the American Chemical Society,2019,141:4730-4737. doi: 10.1021/jacs.9b00574 [68] Li Y, Shao H, Lin Z, et al. A general Lewis acidic etching route for preparing MXenes with enhanced electrochemical performance in non-aqueous electrolyte[J]. Nature Materials,2020,19:894-899. doi: 10.1038/s41563-020-0657-0 [69] Urbankowski P, Anasori B, Makaryan T, et al. Synthesis of two-dimensional titanium nitride Ti4N3 (MXene)[J]. Nanoscale,2016,8:11385-11391. doi: 10.1039/C6NR02253G [70] Kamysbayev V, Filatov A S, Hu H C, et al. Covalent surface modifications and superconductivity of two-dimensional metal carbide MXenes[J]. Science,2020,369:979-983. doi: 10.1126/science.aba8311 [71] Xu C, Wang L, Liu Z, et al. Large-area high-quality 2D ultrathin Mo2C superconducting crystals[J]. Nature Materials,2015,14:1135-1141. doi: 10.1038/nmat4374 [72] Wang Z, Kochat V, Pandey P, et al. Metal immiscibility route to synthesis of ultrathin carbides, borides and nitrides[J]. Advanced Materials,2017,29:1700364. doi: 10.1002/adma.201700364 [73] Wang L, Xu C, Liu Z, et al. Magnetotransport properties in high-quality ultrathin two-dimensional superconducting Mo2C crystals[J]. ACS Nano,2016,10:4504-4510. doi: 10.1021/acsnano.6b00270 [74] Hong Y L, Liu Z B, Wang L, et al. Chemical vapor deposition of layered two-dimensional MoSi2N4 materials[J]. Science,2020,369:670-674. doi: 10.1126/science.abb7023 [75] Mei J, Ayoko G A, Hu C, et al. Two-dimensional fluorine-free mesoporous Mo2C MXene via UV-induced selective etching of Mo2Ga2C for energy storage[J]. Sustainable Materials and Technologies,2020,25:e00156. doi: 10.1016/j.susmat.2020.e00156 [76] Ljubek G, Kralj M, Kraljić Roković M. Fluorine-free mechanochemical synthesis of MXene[J]. Materials Science and Technology,2023:1-5. doi: 10.1080/02670836.2023.2178173 [77] L. Liu, H. Zschiesche, M. Antonietti, et al. In situ synthesis of MXene with tunable morphology by electrochemical etching of MAX phase prepared in molten salt[J]. Advanced Energy Materials,2023,13:2203805. doi: 10.1002/aenm.202203805 [78] Chen Q, Wei Y, Zhang X, et al. Vertically aligned MXene nanosheet arrays for high-rate lithium metal anodes[J]. Advanced Energy Materials,2022,12:2200072. doi: 10.1002/aenm.202200072 [79] Cao Z J, Zhu Q, Wang S, et al. Perpendicular MXene arrays with periodic interspaces toward dendrite-free lithium metal anodes with high-rate capabilities[J]. Advanced Functional Materials,2020,30:1908075. doi: 10.1002/adfm.201908075 [80] Shen K, Li B, Yang S. 3D printing dendrite-free lithium anodes based on the nucleated MXene arrays[J]. Energy Storage Materials,2020,24:670-675. doi: 10.1016/j.ensm.2019.08.015 [81] Shi H, Zhang C J, Lu P, et al. Conducting and lithiophilic MXene/graphene framework for high-capacity, dendrite-free lithium-metal anodes[J]. ACS Nano,2019,13:14308-14318. doi: 10.1021/acsnano.9b07710 [82] Fang Y, Zhang Y, Zhu K, et al. Lithiophilic three-dimensional porous Ti3C2Tx-rGO membrane as a stable scaffold for safe alkali metal (Li or Na) anodes[J]. ACS Nano,2019,13:14319-14328. doi: 10.1021/acsnano.9b07729 [83] Zheng W, Zhang P, Chen J, et al. In situ synthesis of CNTs@Ti3C2 hybrid structures by microwave irradiation for high-performance anodes in lithium ion batteries[J]. Journal of Materials Chemistry A,2018,6:3543-3551. doi: 10.1039/C7TA10394H [84] Zhang S, Liu H, Cao B, et al. An MXene/CNTs@P nanohybrid with stable Ti-O-P bonds for enhanced lithium ion storage[J]. Journal of Materials Chemistry A,2019,7:21766-21773. doi: 10.1039/C9TA07357D [85] Liu Y, Sun C, Lu Y, et al. Lamellar-structured anodes based on lithiophilic gradient enable dendrite-free lithium metal batteries with high capacity loading and fast-charging capability[J]. Chemical Engineering Journal,2023,451:138570. doi: 10.1016/j.cej.2022.138570 [86] Tao M, Li W, Luo P, et al. High utilization lithium metal anode constructed by allocatable MXene foam container[J]. Journal of Power Sources,2022,551:232089. doi: 10.1016/j.jpowsour.2022.232089 [87] Pourali Z, Sovizi M R, Yaftian M R. Two-dimensional Ti3C2Tx/CMK-5 nanocomposite as high performance anodes for lithium batteries[J]. Journal of Alloys and Compounds,2018,738:130-137. doi: 10.1016/j.jallcom.2017.12.116 [88] Gu J, Zhu Q, Shi Y, et al. Single zinc atoms immobilized on MXene (Ti3C2Clx) layers toward dendrite-free lithium metal anodes[J]. ACS Nano,2020,14:891-898. doi: 10.1021/acsnano.9b08141 [89] Qian Y, Wei C, Tian Y, et al. Constructing ultrafine lithiophilic layer on MXene paper by sputtering for stable and flexible 3D lithium metal anode[J]. Chemical Engineering Journal,2021,421:129685. doi: 10.1016/j.cej.2021.129685 [90] Wu N, Zhang Q Y, Guo Y J, et al. Boron-doped three-dimensional MXene host for durable lithium-metal anode[J]. Rare Metals,2022,41:2217-2222. doi: 10.1007/s12598-021-01944-5 [91] Shang M, Shovon O G, Wong F E Y, et al. A BF3-doped MXene dual-layer interphase for a reliable lithium-metal anode[J]. Advanced Materials,2023,35:2210111. doi: 10.1002/adma.202210111 [92] Na Z, Li W, Li L, et al. Conductive iodine-doped red phosphorus enabled dendrite-free lithium deposition on MXene matrix[J]. Small,2022,18:2204341. doi: 10.1002/smll.202204341 -





 下载:
下载: