-
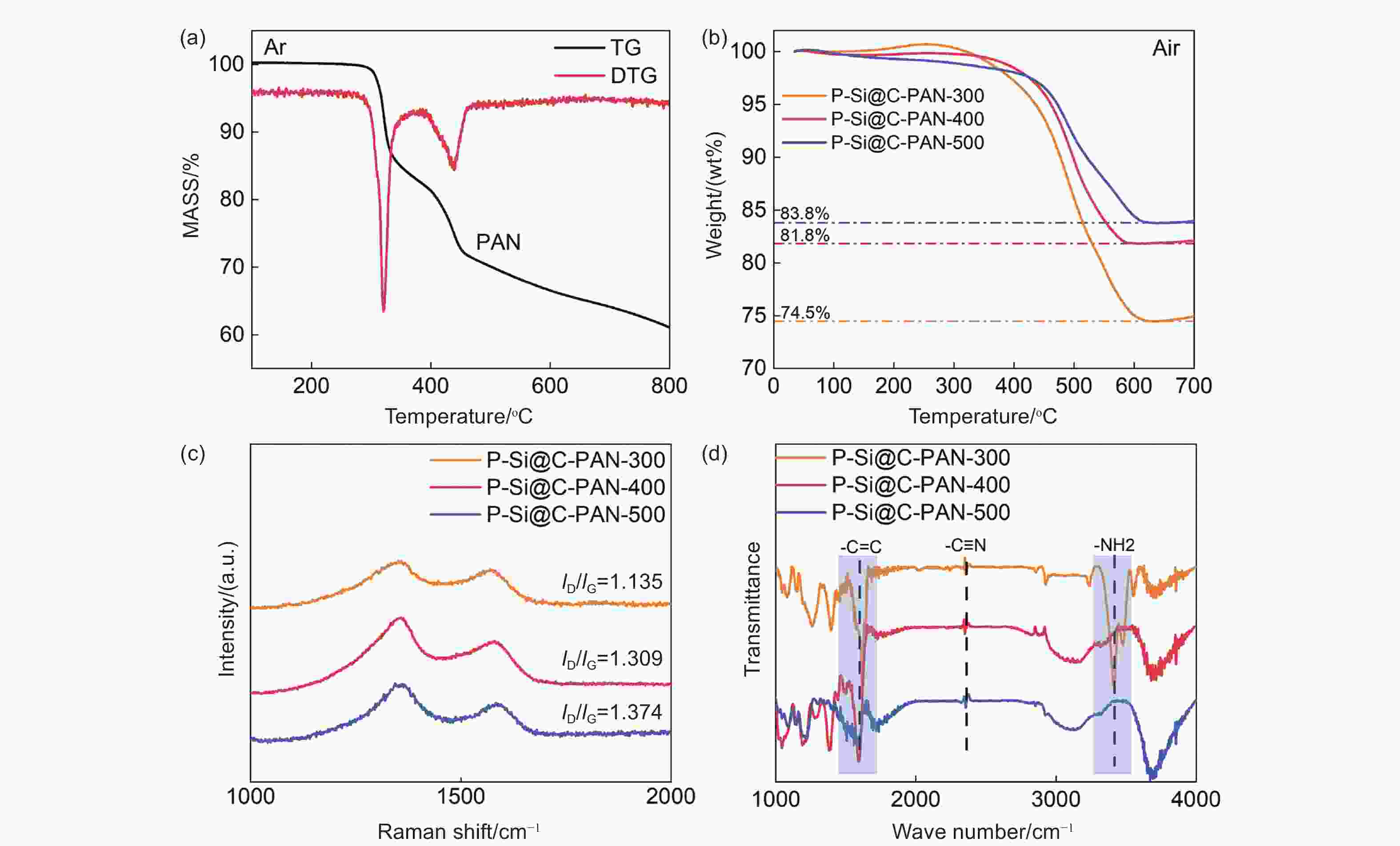
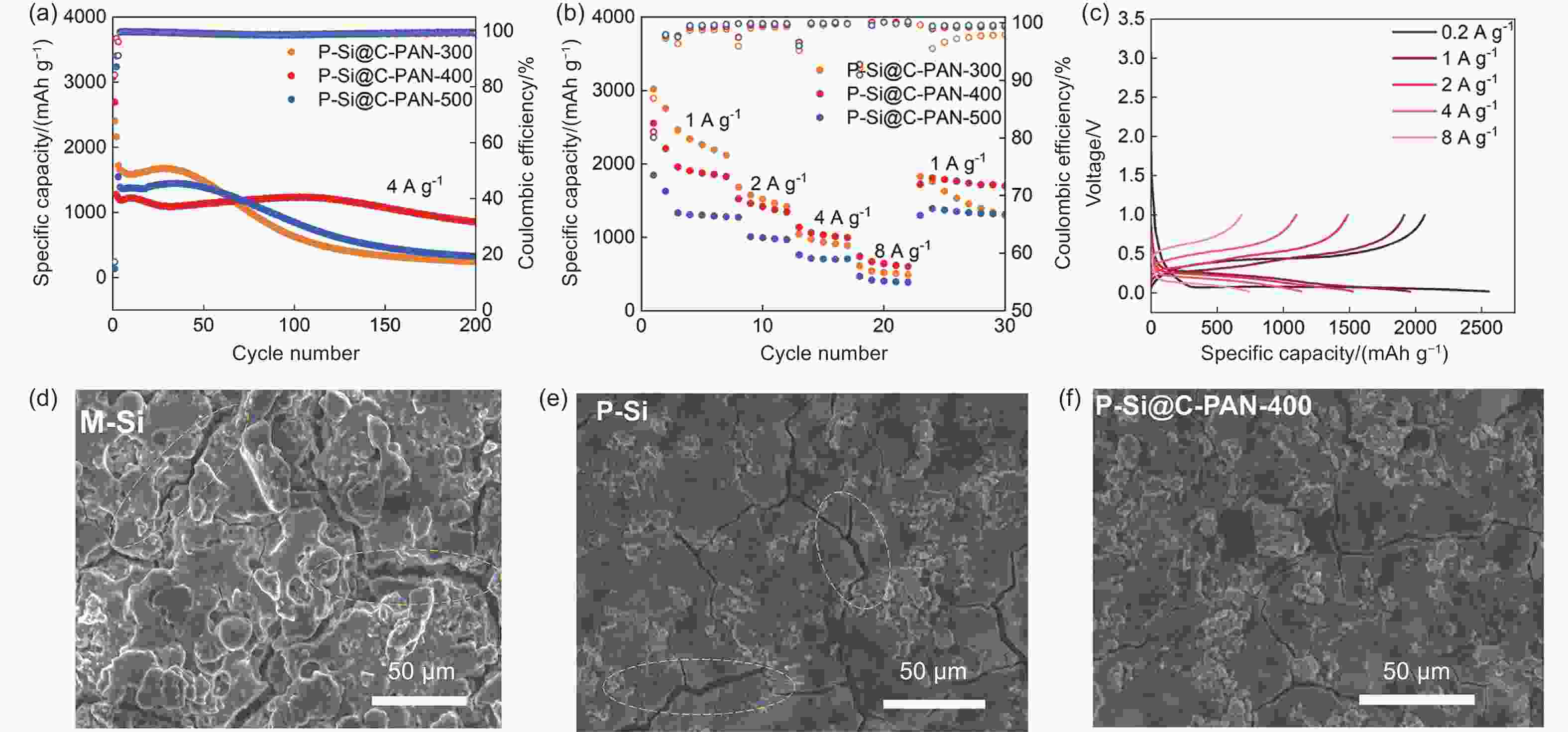
摘要: 硅负极是锂离子电池理想的候选材料。然而,其显著的体积膨胀会导致严重的材料断裂,失去电接触,从而限制了其实际应用。本研究提出了一种新的自上而下的多孔硅制备策略,并引入聚丙烯腈(PAN)作为掺氮碳涂层,旨在保持硅负极的内部空间,缓解硅负极在锂化和脱锂过程中向外膨胀的问题。随后,我们探讨了温度对PAN热转变行为和复合电极电化学行为的影响。在400 °C下处理后,PAN涂层保留了11.35 wt%的高氮掺杂含量,这明确证实了C―N和C―O键的存在,从而改善了离子电子传输特性。这种处理方法不仅保留了更完整的碳层结构,还引入了碳缺陷,即使在大电流下也能稳定循环。当以4 A g−1的电流循环时,优化后的负极在循环200次后仍能显示出857.6 mAh g−1的比容量,显示出其在高容量储能应用方面的巨大潜力。Abstract: Silicon anodes as a promising candidate for lithium-ion batteries. However, their significant volume expansion leads to severe material fracture and electrical disconnection, which limits their practical application. This study proposed a new top-down strategy for microsized porous silicon and introducing polyacrylonitrile (PAN) as nitrogen-doped carbon coating, which designed to maintain the internal space and alleviate the outward expansion of the silicon anode during the lithiation and delithiation process. Subsequently, we explored the effect of temperature on the thermal transition behavior of PAN and the electrochemical behavior of the composite electrode. After the treatment at 400 °C, the PAN coating retained a high nitrogen doping content of 11.35 wt%, which explicitly confirmed the existence of C-N and C-O bonds that improved the ionic-electronic transport properties. This treatment not only retained a more intact carbon layer structure, but also introduced carbon defects, exhibiting remarkably stable cycling even at high rates. When cycled at 4 A g−1, the optimized anode exhibited a specific capacity of 857.6 mAh g−1 even after 200 cycles, demonstrating great potential for high-capacity energy storage applications.
-
Key words:
- Porous silicon /
- Lithium-ion batteries /
- Polyacrylonitrile /
- Electrochemical behavior
-
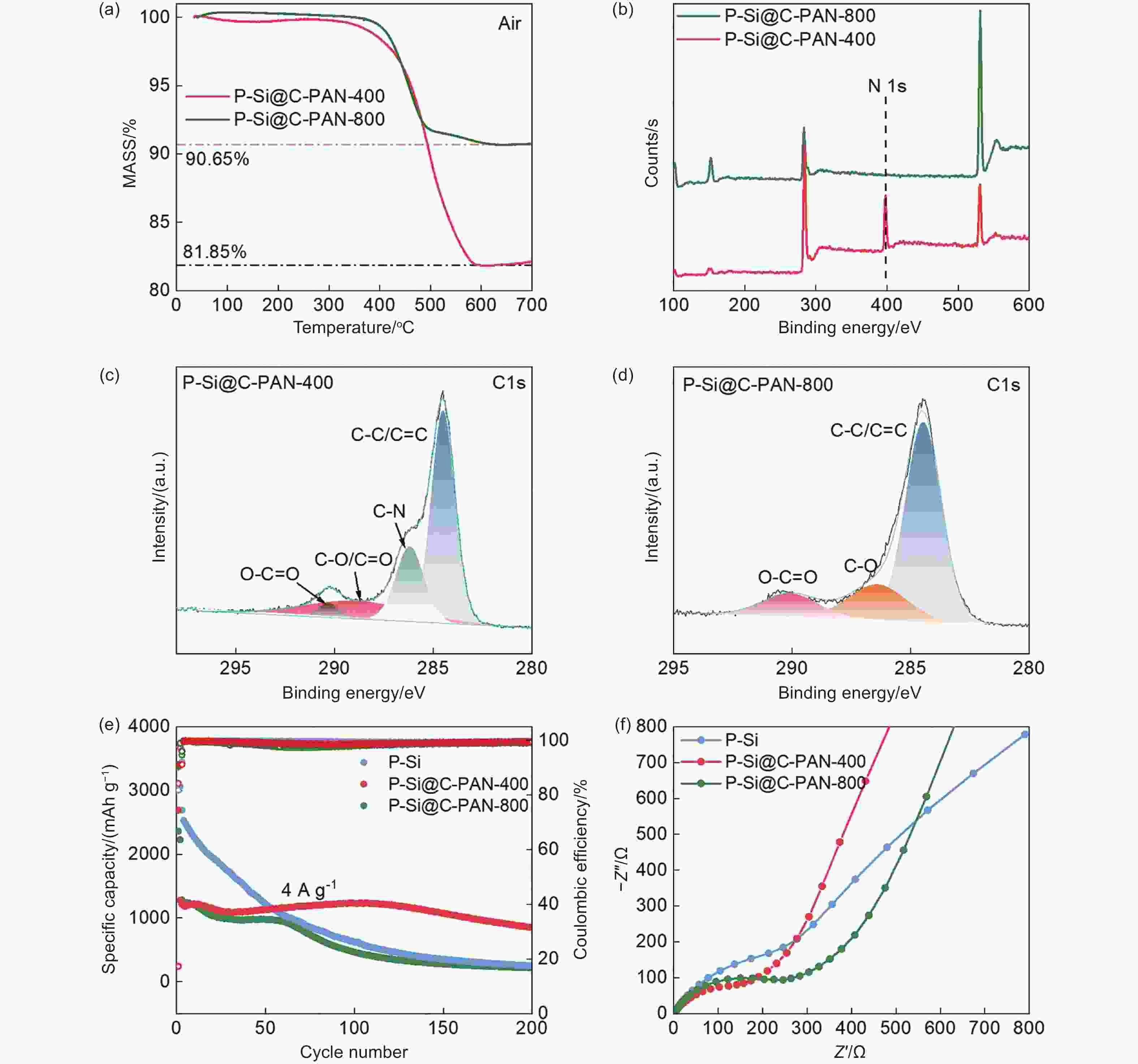
Figure 3. (a) Thermogravimetric analysis of P-Si@C-PAN-400 and P-Si@C-PAN-800, (b) XPS survey of P-Si@C-PAN-400 and P-Si@C-PAN-800. High resolution of C 1s XPS spectra of (c) P-Si@C-PAN-400 and (d) P-Si@C-PAN-800. (e) Electrochemical cycling performance of P-Si, P-Si@C-PAN-400, and P-Si@C-PAN-800. (f) EIS of P-Si, P-Si@C-PAN-400, and P-Si@C-PAN-800
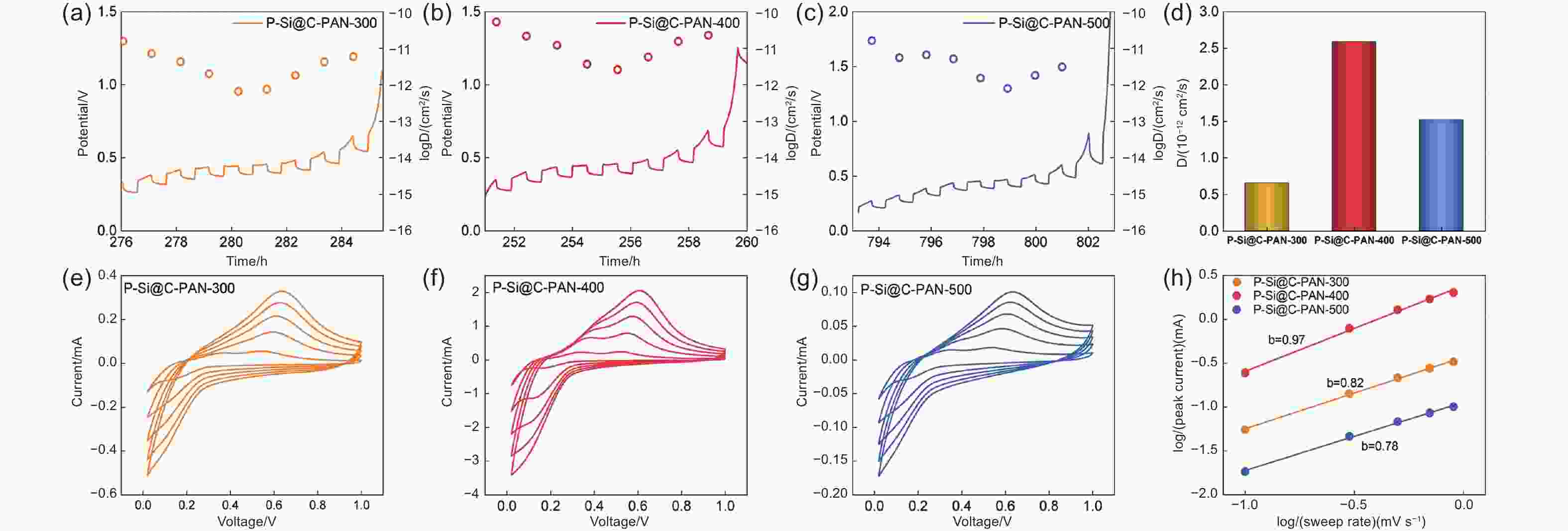
Figure 5. GITT curves of (a) P-Si@C-PAN-300, (b) P-Si@C-PAN-400, and (c) P-Si@C-PAN-500. (d) comparison for Li+ diffusion coefficients of different samples treated at different temperatures. CV curves measured at different scan rates for (e) P-Si@C-PAN-300, (f) P-Si@C-PAN-400, and (g) P-Si@C-PAN-500. (h) Linear relationship between log i (peak current) and log v (scan rate) for the three samples
-
[1] Chae S, Choi S H, Kim N, et al. Integration of graphite and silicon anodes for the commercialization of high-energy lithium-ion batteries[J]. Angewandte Chemie International Edition,2020,59(1):110-135. doi: 10.1002/anie.201902085 [2] Liu D Q, Liu Z J, Li X W, et al. Group IVA Element (Si, Ge, Sn)-based alloying/dealloying anodes as negative electrodes for full-cell lithium-ion batteries[J]. Small, 2017, 13(45). [3] Dunn B, Kamath H, Tarascon J M. Electrical energy storage for the grid: a battery of choices[J]. Science,2011,334(6058):928-935. doi: 10.1126/science.1212741 [4] Ko M, Chae S, Ma J, et al. Scalable synthesis of silicon-nanolayer-embedded graphite for high-energy lithium-ion batteries[J]. Nature Energy,2016,1(9):16113. doi: 10.1038/nenergy.2016.113 [5] Jin Y, Li S, Kushima A, et al. Self-healing SEI enables full-cell cycling of a silicon-majority anode with a coulombic efficiency exceeding 99. 9%[J]. Energy & Environmental Science,2017,10(2):580-592. [6] Zhang X W, Patil P K, Wang C S, et al. Electrochemical performance of lithium ion battery, nano-silicon-based, disordered carbon composite anodes with different microstructures[J]. Journal of Power Sources,2004,125(2):206-213. doi: 10.1016/j.jpowsour.2003.07.019 [7] Han M S, Mu Y B, Wei L, et al. Multilevel carbon architecture of subnanoscopic silicon for fast-charging high-energy-density lithium-ion batteries[J]. Carbon Energy, 2023. [8] Neiner D, Chiu H W, Kauzlarich S M. Low-temperature solution route to macroscopic amounts of hydrogen terminated silicon nanoparticles[J]. Journal of the American Chemical Society,2006,128(34):11016-11017. doi: 10.1021/ja064177q [9] Trill J-H, Tao C, Winter M, et al. NMR investigations on the lithiation and delithiation of nanosilicon-based anodes for Li-ion batteries[J]. Journal of Solid State Electrochemistry,2011,15(2):349-356. doi: 10.1007/s10008-010-1260-0 [10] Hwa Y, Kim W-S, Yu B-C, et al. Facile synthesis of Si nanoparticles using magnesium silicide reduction and its carbon composite as a high-performance anode for Li ion batteries[J]. Journal of Power Sources,2014,252:144-149. doi: 10.1016/j.jpowsour.2013.11.118 [11] Xiao Z, Lei C, Yu C, et al. Si@Si3N4@C composite with egg-like structure as high-performance anode material for lithium ion batteries[J]. Energy Storage Materials,2020,24:565-573. doi: 10.1016/j.ensm.2019.06.031 [12] Wang D, Zhou C, Cao B, et al. One-step synthesis of spherical Si/C composites with onion-like buffer structure as high-performance anodes for lithium-ion batteries[J]. Energy Storage Materials,2020,24:312-318. doi: 10.1016/j.ensm.2019.07.045 [13] Du F H, Li B, Fu W, et al. Surface binding of polypyrrole on porous silicon hollow nanospheres for Li-ion battery anodes with high structure stability[J]. Advanced Materials,2014,26(35):6145-6150. doi: 10.1002/adma.201401937 [14] Zhang J, Fang S, Qi X, et al. Preparation of high-purity straight silicon nanowires by molten salt electrolysis[J]. Journal of Energy Chemistry,2020,40:171-179. doi: 10.1016/j.jechem.2019.04.014 [15] Liu B, Huang P, Zhang Q, et al. Rational-design micro-nanostructure of porous carbon film/silicon nanowire/graphite microsphere composites for high-performance lithium-ion batteries[J]. Journal of Materials Science,2020,55(26):12165-12176. doi: 10.1007/s10853-020-04869-z [16] Zhou H, Nanda J, Martha S K, et al. Role of surface functionality in the electrochemical performance of silicon nanowire anodes for rechargeable lithium batteries[J]. Acs Applied Materials & Interfaces,2014,6(10):7607-7614. [17] Zhang Z, Han X, Li L, et al. Tailoring the interfaces of silicon/carbon nanotube for high rate lithium-ion battery anodes[J]. Journal of Power Sources,2020,450:227593. doi: 10.1016/j.jpowsour.2019.227593 [18] Wu H, Chan G, Choi J W, et al. Stable cycling of double-walled silicon nanotube battery anodes through solid-electrolyte interphase control[J]. Nature Nanotechnology,2012,7(5):309-314. [19] Ko M, Chae S, Ma J, et al. Scalable synthesis of silicon-nanolayer-embedded graphite for high-energy lithium-ion batteries (Vol 1, 16113, 2016)[J]. Nature Energy,2020,5(4):344-344. doi: 10.1038/s41560-020-0587-8 [20] Ma Y, Zhang Q, Shi L, et al. Low-cost micron-scale silicon-based anodes for high-performance lithium-ion batteries[J]. ACS Applied Energy Materials,2023,6(14):7545-7555. doi: 10.1021/acsaem.3c00951 [21] Entwistle J, Rennie A, Patwardhan S. A review of magnesiothermic reduction of silica to porous silicon for lithium-ion battery applications and beyond[J]. Journal of Materials Chemistry A,2018,6(38):18344-18356. doi: 10.1039/C8TA06370B [22] Zhang F, Zhu W Q, Li T T, et al. Advances of synthesis methods for porous silicon-based anode materials[J]. Frontiers in Chemistry, 2022, 10. [23] Ge M, Fang X, Rong J, et al. Review of porous silicon preparation and its application for lithium-ion battery anodes[J]. Nanotechnology, 2013, 24(42). [24] Kim W-J, Kang J G, Kim D-W. Fibrin biopolymer hydrogel-templated 3d interconnected si@c framework for lithium ion battery anodes[J]. Applied Surface Science,2021,551:149439. doi: 10.1016/j.apsusc.2021.149439 [25] Cheng Z L, Jiang H, Zhang X L, et al. Fundamental understanding and facing challenges in structural design of porous si-based anodes for lithium-ion batteries[J]. Advanced Functional Materials, 2023, 33(26). [26] Bao Z, Weatherspoon M R, Shian S, et al. Chemical reduction of three-dimensional silica micro-assemblies into microporous silicon replicas[J]. Nature,2007,446(7132):172-175. doi: 10.1038/nature05570 [27] Wang D H, Ma Y J, Xu W Q, et al. Controlled isotropic canalization of microsized silicon enabling stable high-rate and high-loading lithium storage[J]. Advanced Materials, 2023, 35(21). [28] Han X, Gu L H, Sun Z F, et al. Manipulating charge-transfer kinetics and a flow-domain LiF-rich interphase to enable high-performance microsized silicon-silver-carbon composite anodes for solid-state batteries[J]. Energy & Environmental Science, 2023. [29] Chae S, Xu Y B, Yi R, et al. A micrometer-sized silicon/carbon composite anode synthesized by impregnation of petroleum pitch in nanoporous silicon[J]. Advanced Materials, 2021, 33(40). [30] Yuan L-Y, Lu C-X, Lu X-X, et al. Synthesis and electrochemical properties of Nano-Si/C composite anodes for lithium-ion batteries[J]. New Carbon Materials,2023,38(5):964-975. doi: 10.1016/S1872-5805(23)60707-3 [31] Qian C, Guo P, Zhang X, et al. Nitrogen-doped mesoporous hollow carbon nanoflowers as high performance anode materials of lithium ion batteries[J]. Rsc Advances,2016,6(96):93519-93524. doi: 10.1039/C6RA21011B [32] Shaker M, Ghazvini A A S, Shahalizade T, et al. A review of nitrogen-doped carbon materials for lithium-ion battery anodes[J]. New Carbon Materials,2023,38(2):247-278. doi: 10.1016/S1872-5805(23)60724-3 [33] Mu Y, Han M, Li J, et al. Growing vertical graphene sheets on natural graphite for fast charging lithium-ion batteries[J]. Carbon,2021,173:477-484. doi: 10.1016/j.carbon.2020.11.027 [34] Han M, Chen J, Cai Y, et al. Magnetic-atom strategy enables unilamellar MoS2-C interoverlapped superstructure with ultrahigh capacity and ultrafast ion transfer capability in Li/Na/K-ion batteries[J]. Chemical Engineering Journal,2023,454:140137. doi: 10.1016/j.cej.2022.140137 [35] Chen Y X, Xi B J, Huang M, et al. Defect-selectivity and "order-in-disorder" engineering in carbon for durable and fast potassium storage[J]. Advanced Materials, 2022, 34(7). [36] Geng C, Chen Y-x, Shi L-l, et al. Design of active sites in carbon materials for electrochemical potassium storage[J]. New Carbon Materials,2022,37(3):461-483. doi: 10.1016/S1872-5805(22)60612-7 [37] Kakida H, Tashiro K. Mechanism and kinetics of stabilization reactions of polyacrylonitrile and related copolymers III. comparison among the various types of copolymers as viewed from isothermal DSC thermograms and FT-IR spectral changes[J]. Polymer Journal,1997,29(7):557-562. doi: 10.1295/polymj.29.557 [38] Jiang S, Mao M-m, Pang M-j, et al. Preparation and performance of a Graphene-(Ni-NiO)-C hybrid as the anode of a lithium-ion battery[J]. New Carbon Materials,2023,38(2):356-365. doi: 10.1016/S1872-5805(22)60647-4 -





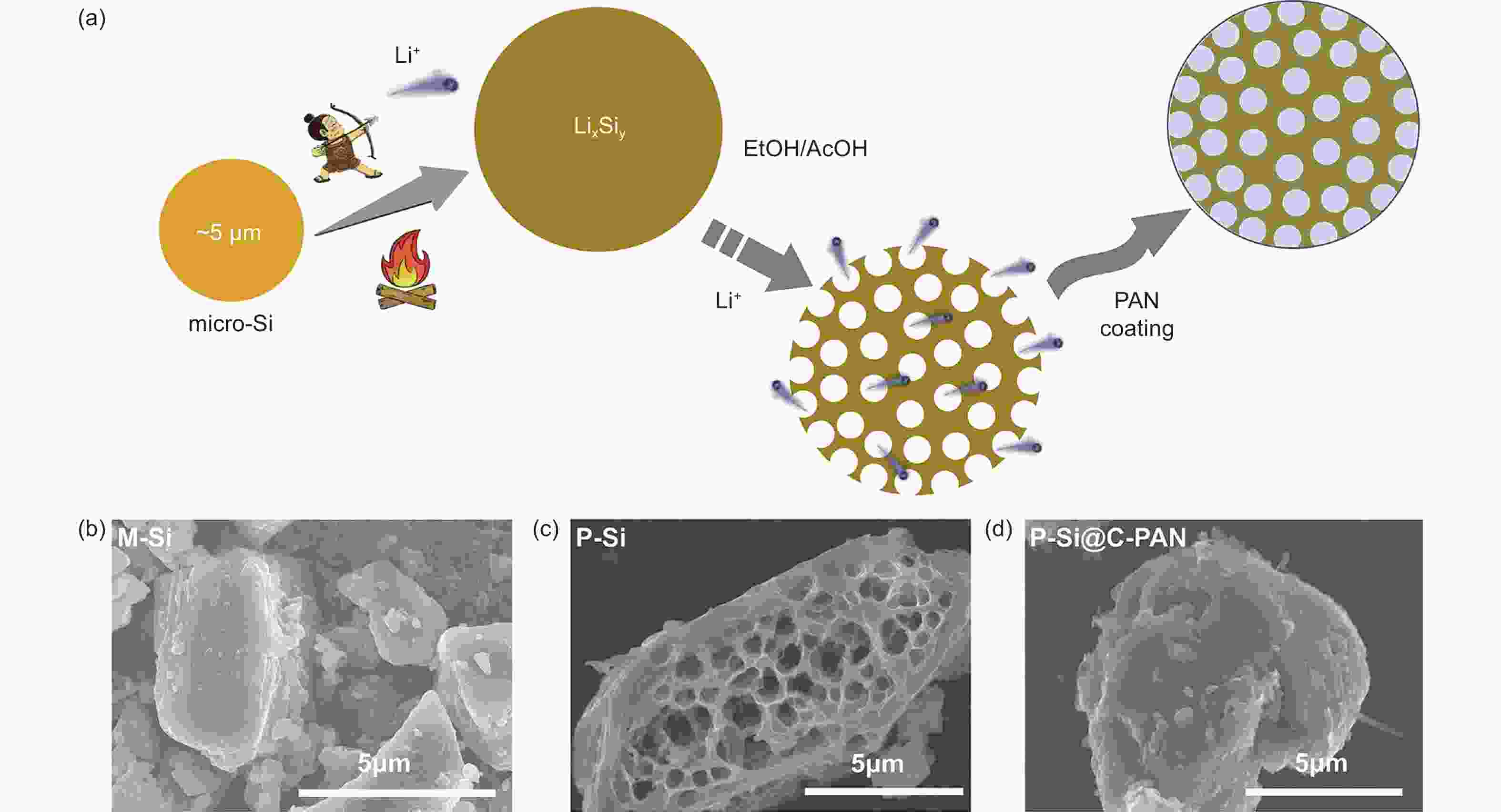
 下载:
下载: