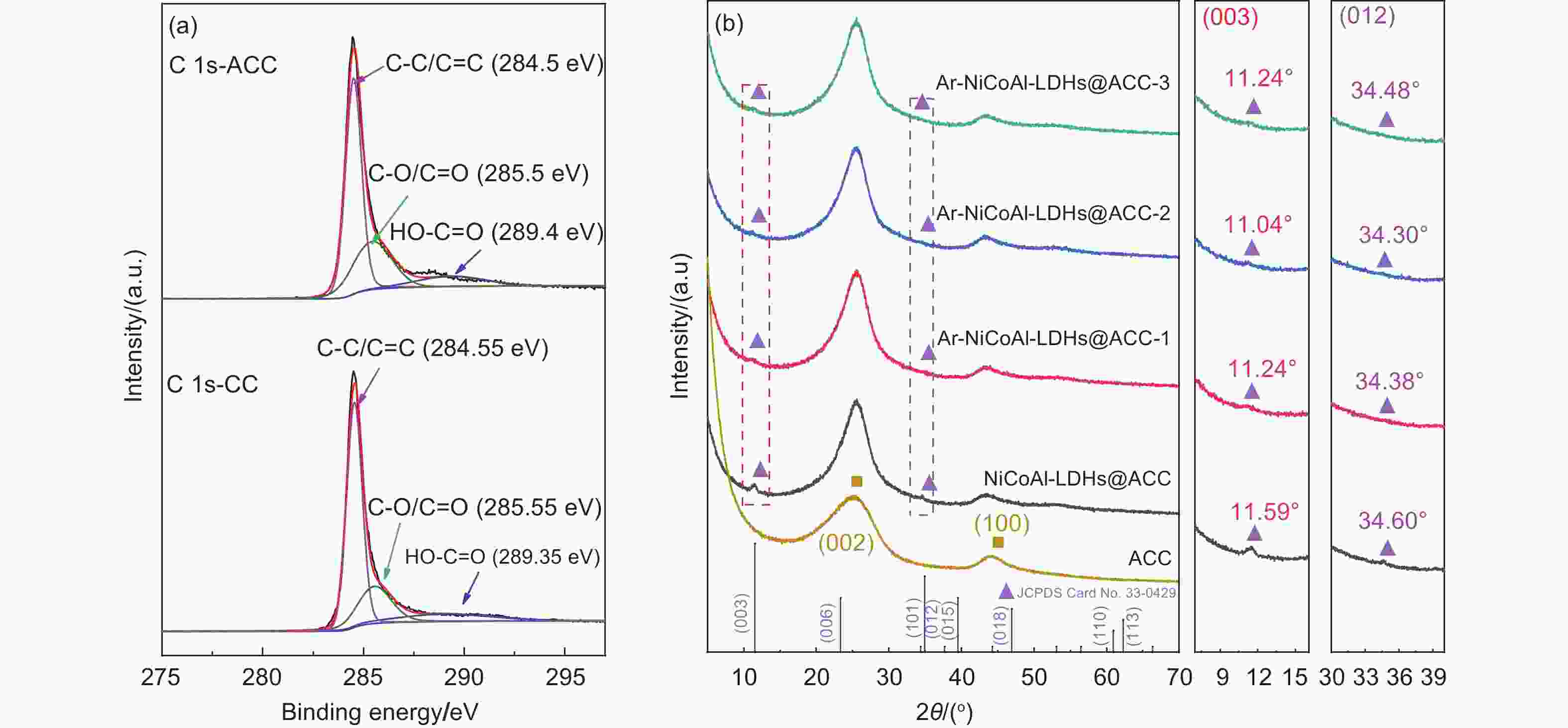
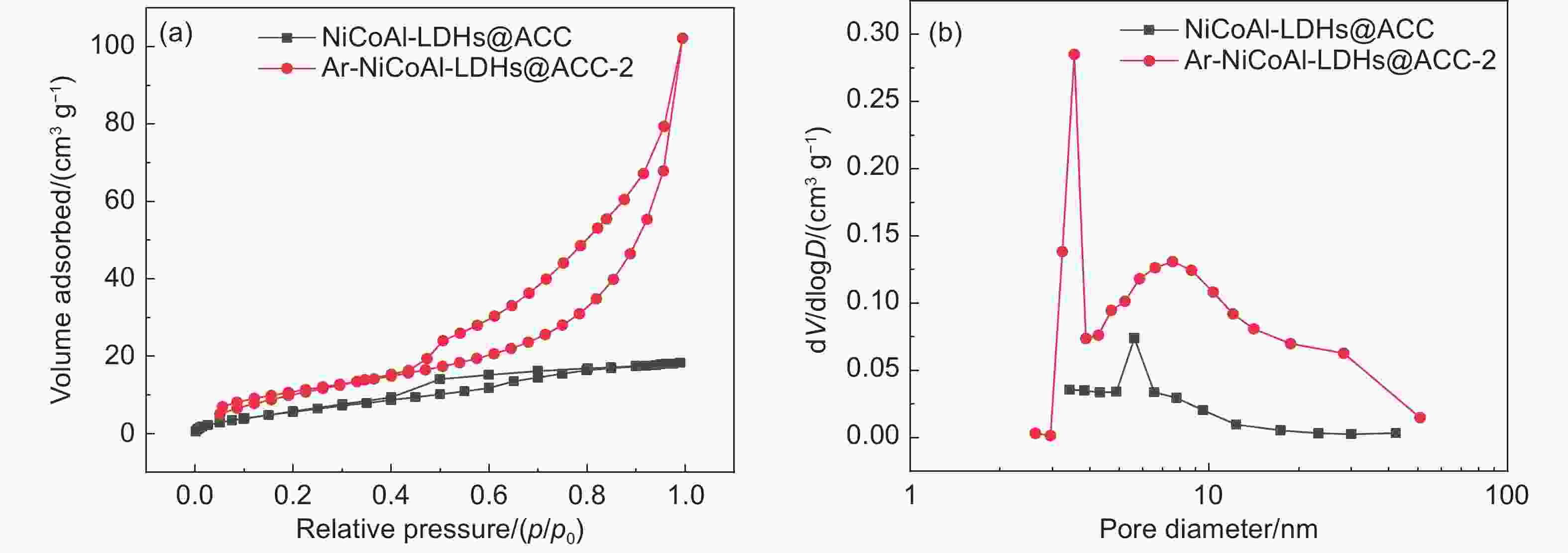
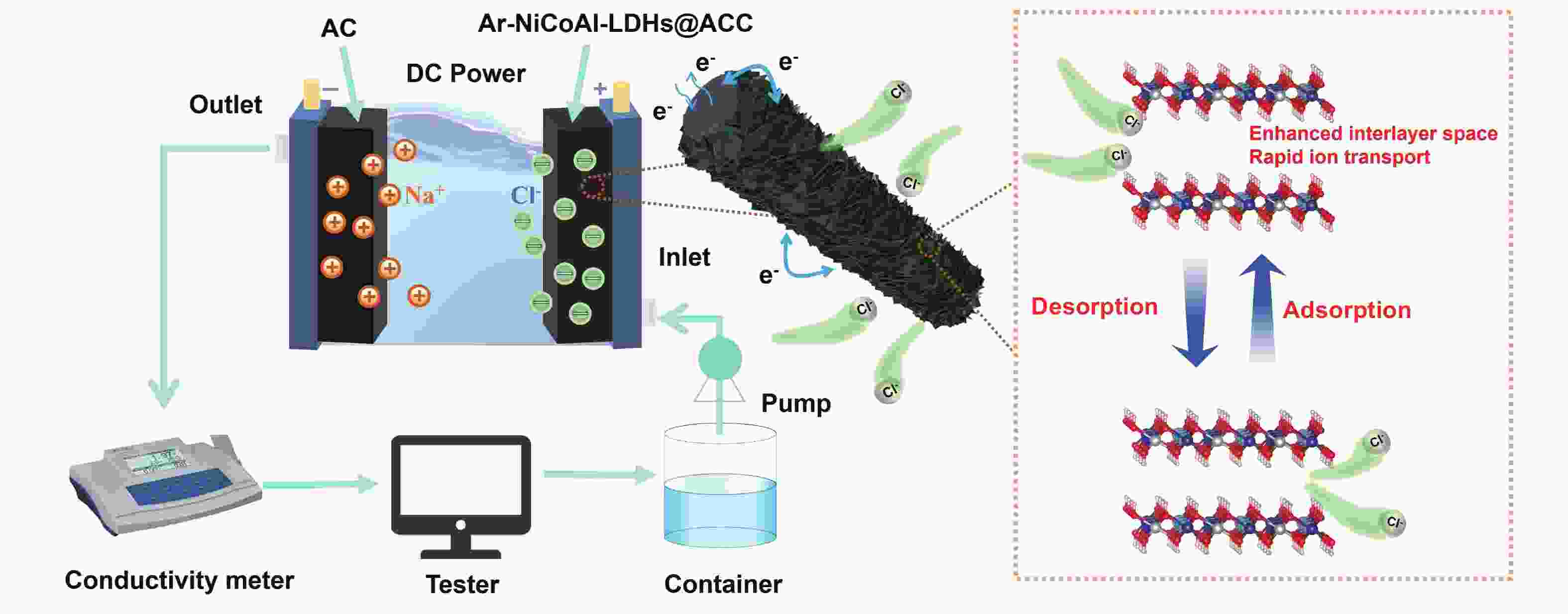
Plasma-assisted preparation of NiCoAl-LDHs with enhanced interlayer space on carbon cloth for electrochemical deionization
-
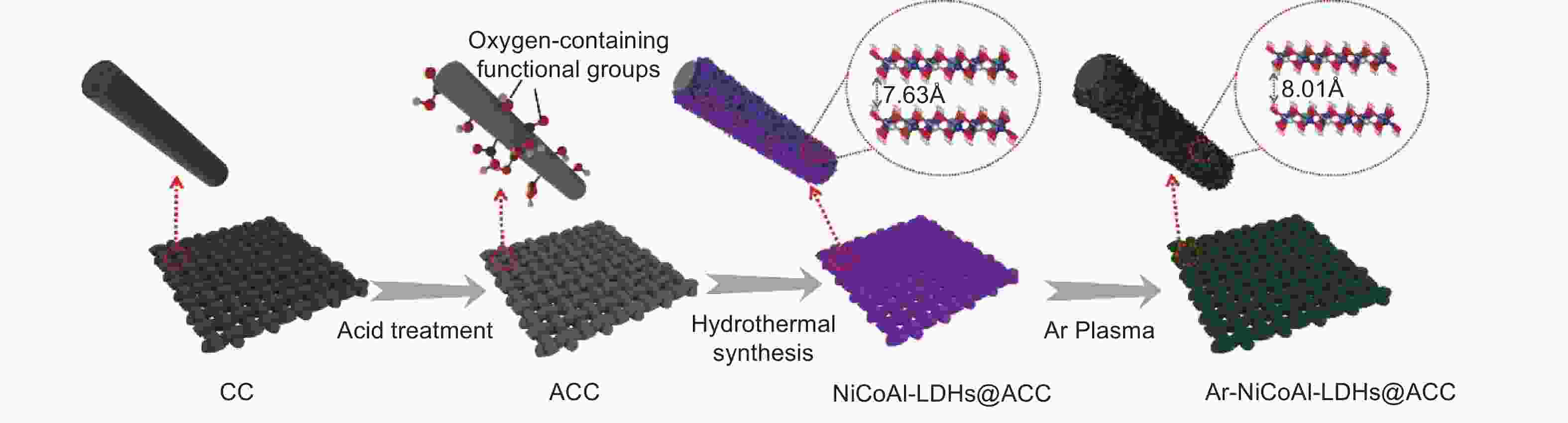
摘要: 电容去离子技术近年来被认为是一种新兴的海水淡化技术,尤其在苦咸水范围内具有经济节能的特点。然而,目前关于除氯电极的研究较少,同时缓慢的除盐动力学也制约了除氯电极的发展。本工作通过在表面酸处理后的柔性炭布上原位生长NiCoAl-LDHs纳米片阵列并进行等离子体处理,制备了具有扩大层间距的Ar-NiCoAl-LDHs@ACC材料。炭布基底抑制了NiCoAl-LDHs纳米片的团聚并提高了电导率,等离子体处理则使得NiCoAl-LDHs层间距进一步扩大并改善了亲水性,提供了快速的氯离子扩散通道,并释放了更多的层间活性位点,实现了高除盐动力学。将Ar-NiCoAl-LDHs@ACC作为除氯电极与活性碳组装了混合式电容去离子器件。在1000 mg L−1NaCl溶液及1.2 V工作电压下,除盐容量可达到93.26 mg g−1,除盐速率可达到0.27 mg g−1 s−1,电荷效率高达0.97。在300 mg L−1NaCl溶液及0.8 V工作电压下,经过100次循环后容量保持率仍在85%以上。本工作的制备策略为大层间距二维金属氢氧化物材料的可控制备和高性能电化学除氯电极的设计构建提供了新思路。
-
关键词:
- 电化学去离子 /
- 海水淡化 /
- 大层间距 /
- NiCoAl-LDHs /
- 高除盐动力学
Abstract: Capacitive deionization technology has been considered as an emerging desalination technique in recent years, especially for its economic and energy-saving characteristics within the brackish water range. However, there are currently few studies on chloride ion removal electrodes, and the slow desalination kinetics limits their development. In this work, Ar-NiCoAl-LDHs@ACC materials with enhanced interlayer space were prepared by in-situ growth of NiCoAl-LDHs nanosheets arrays on acid-treated carbon cloth and subsequent argon plasma treatment. The carbon cloth suppresses the agglomeration of NiCoAl-LDHs nanosheets and improves the electrical conductivity, while the plasma treatment further expands the interlayer space of NiCoAl-LDHs and enhances the hydrophilicity. This provides a rapid diffusion channel and more interlayer active sites for chloride ions, achieving high desalination kinetics. A hybrid capacitive deionization (HCDI) cell was assembled using the Ar-NiCoAl-LDHs@ACC as chloride ion removal electrode and activated carbon as sodium ion removal electrode. This HCDI cell achieves a high desalination capacity of 93.26 mg g−1 at 1.2 V in 1000 mg L−1 NaCl solution, remarkable desalination rate of 0.27 mg g−1 s−1, and good charge efficiency of 0.97. In 300 mg L−1 NaCl solution at 0.8 V, the capacity retention rate remains above 85% after 100 cycles. This work provides new ideas for the controllable preparation of two-dimensional metal hydroxide materials with large interlayer space and the design of high-performance electrochemical chlorine ion removal electrodes. -
图 2 (a) CC,(b) ACC,(c) NiCoAl-LDHs@ACC,(d) Ar-NiCoAl-LDHs@ACC-1,(e) Ar-NiCoAl-LDHs@ACC-2,(f) Ar-NiCoAl-LDHs@ACC-3的SEM图;(g-l)Ar-NiCoAl-LDHs@ACC-2的元素分布图
Figure 2. SEM images of (a) CC, (b) ACC, (c) NiCoAl-LDHs@ACC, (d) Ar-NiCoAl-LDHs@ACC-1, (e) Ar-NiCoAl-LDHs@ACC-2 and (f) Ar-NiCoAl-LDHs@ACC-3, (g-l) elemental mapping of Ar-NiCoAl-LDHs@ACC-2
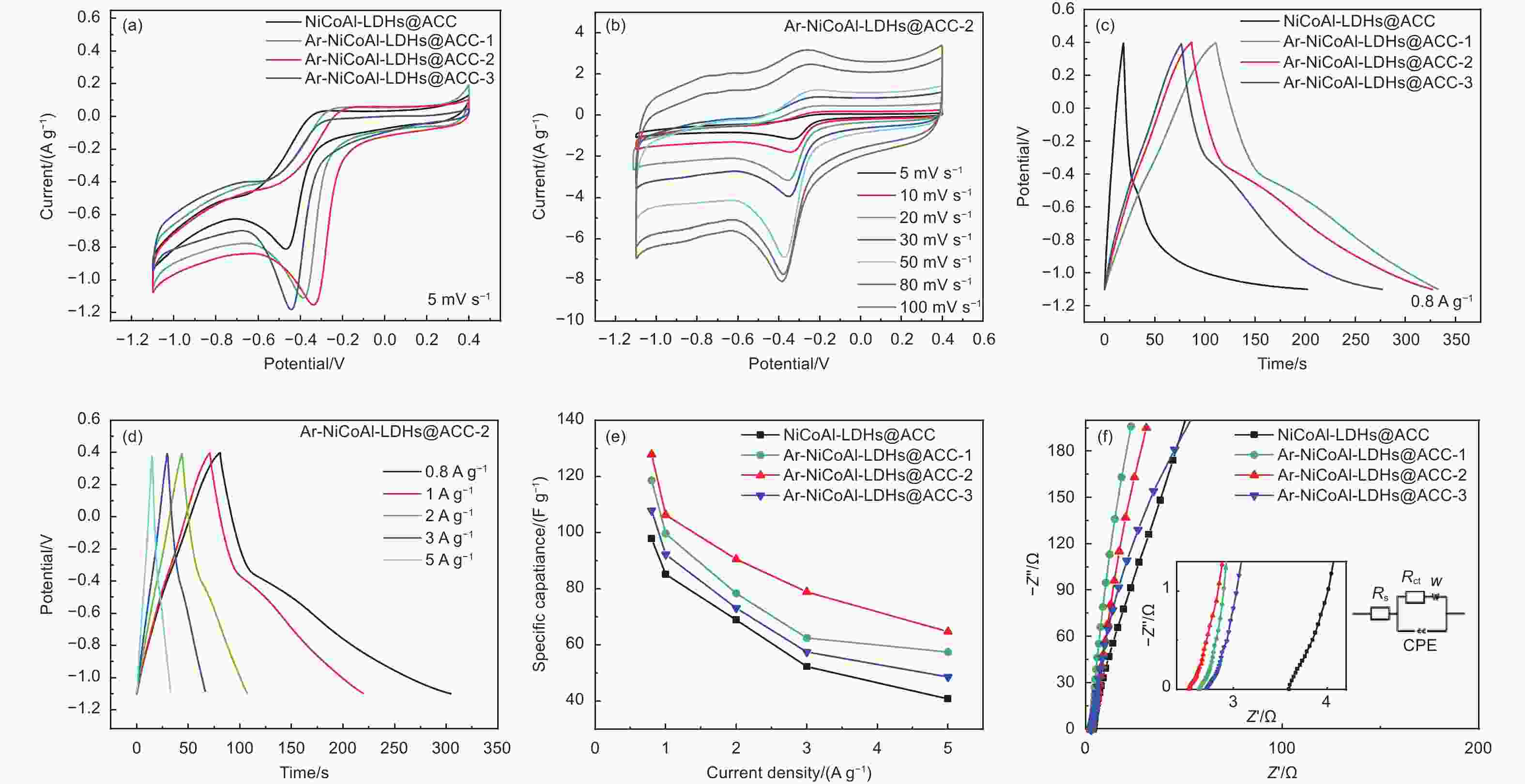
图 6 (a) NiCoAl-LDHs@ACC和Ar-NiCoAl-LDHs@ACC在5mV s−1下的CV曲线,(b) Ar-NiCoAl-LDHs@ACC-2在不同扫描速率下的CV曲线,(c) NiCoAl-LDHs@ACC和Ar-NiCoAl-LDHs@ACC在0.8 A g−1下的GCD曲线,(d) Ar-NiCoAl-LDHs@ACC-20 min在不同电流密度下的GCD曲线,(e) NiCoAl-LDHs@ACC和Ar-NiCoAl-LDHs@ACC在不同电流密度下的比电容,(f) Ar-NiCoAl-LDHs@ACC和Ar-NiCoAl-LDHs@ACC的EIS曲线
Figure 6. CV curves of (a) NiCoAl-LDHs@ACC and Ar-NiCoAl-LDHs@ACC at 5 mV s−1, and (b) Ar-NiCoAl-LDHs@ACC-2 at different scanning rates, GCD curves of (c) NiCoAl-LDHs@ACC and Ar-NiCoAl-LDHs@ACC at 0.8 A g−1, and (d) Ar-NiCoAl-LDHs@ACC-2 at different current densities, (e) the specific capacitances of NiCoAl-LDHs@ACC and Ar-NiCoAl-LDHs@ACC at different current densities, (f) EIS plots of NiCoAl-LDHs@ACC and Ar-NiCoAl-LDHs@ACC
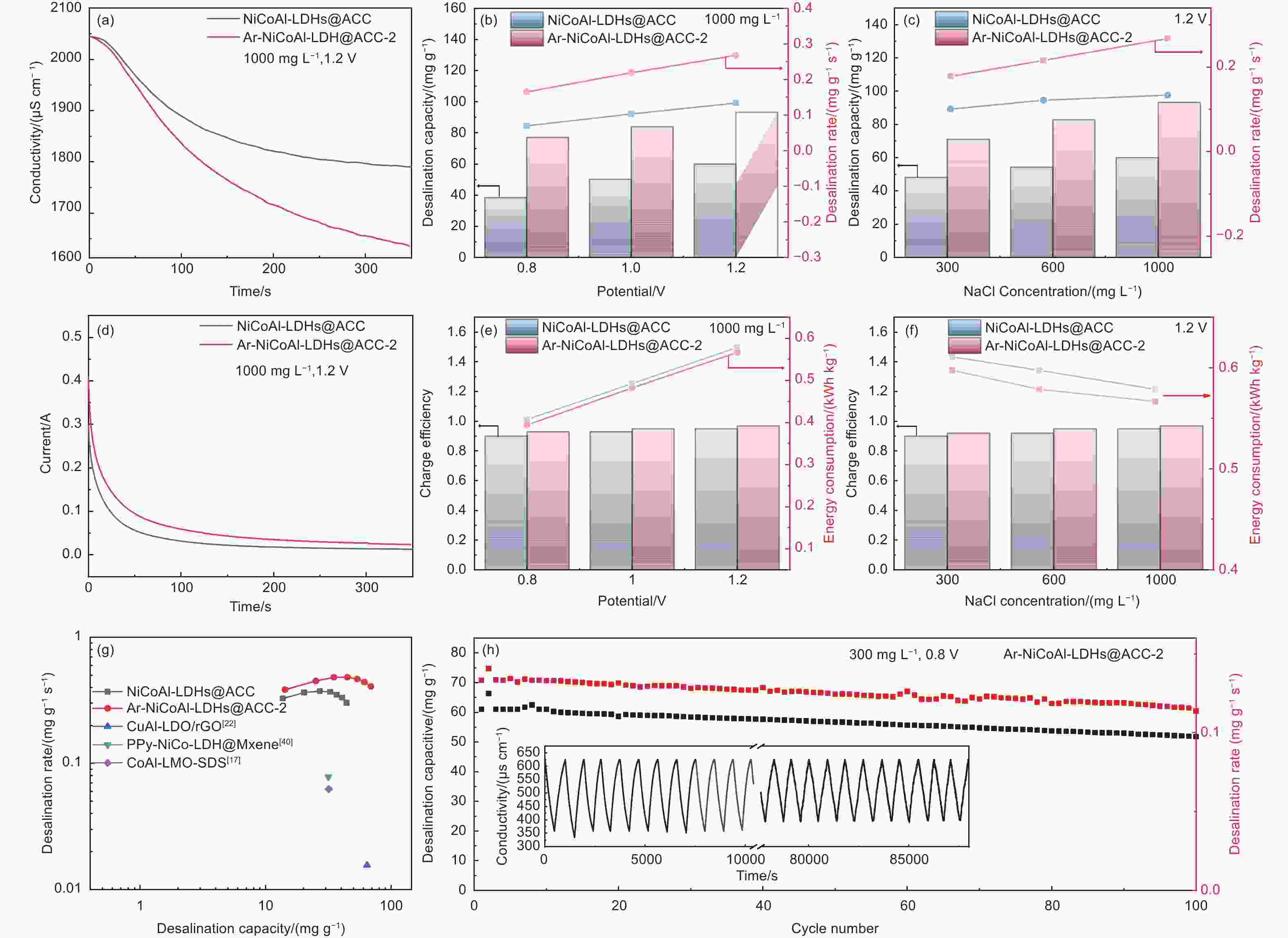
图 8 Ar-NiCoAl-LDHs@ACC-2//AC和NiCoAl-LDHs@ACC//AC系统在(a) 1.2 V下1000 mg L−1的NaCl溶液中的电导率瞬态,(b) 1000 mg L−1的NaCl溶液中在不同工作电压下的除盐容量与除盐速率,(c) 1.2 V 的工作电压下在不同NaCl溶液浓度中的除盐容量与除盐速率,(d) 1.2 V下1000 mg L−1的NaCl溶液中的电流瞬态,(e) 1000 mg L−1的NaCl溶液中在不同工作电压下的电荷效率与能耗,(f) 1.2 V 的工作电压下在不同NaCl溶液浓度中的电荷效率与能耗,(g) Ragone 图,(h) Ar-NiCoAl-LDHs@ACC-2//AC系统在300 mg L−1的NaCl溶液中1.2V下的循环性能图
Figure 8. (a) Conductivity variation of Ar-NiCoAl-LDHs@ACC-2//AC and NiCoAl-LDHs@ACC//AC at 1.2 V in 1000 mg L−1 NaCl solution, the desalination capacity and desalination rate of Ar-NiCoAl-LDHs@ACC-2//AC and NiCoAl-LDHs@ACC//AC at different (b) operational voltages and (c) NaCl concentrations, (d) current variation of Ar-NiCoAl-LDHs@ACC-2//AC and NiCoAl-LDHs@ACC//AC at 1.2 V in 1000 mg L−1 NaCl solution, the charge efficiency and energy consumption of Ar-NiCoAl-LDHs@ACC-2//AC and NiCoAl-LDHs@ACC//AC at different (e) operational voltages and (f) NaCl concentrations, (g) Ragone plots between desalination capacity and desalination rate, (h) cycling performance of the Ar-NiCoAl-LDHs@ACC-2//AC
-
[1] Werber J R, Osuji C O, Elimelech M. Materials for next-generation desalination and water purification membranes[J]. Nature Reviews Materials,2016,1(5):16018. doi: 10.1038/natrevmats.2016.18 [2] Zeng Z H, Yan L L, Li G H, et al. Development of biochar electrode materials for capacitive deionization: Preparation, performance, regeneration and other challenges[J]. New Carbon Materials,2023,38(5):837-860. doi: 10.1016/S1872-5805(23)60779-6 [3] Suss M E, Porada S, Sun X, et al. Water desalination via capacitive deionization: what is it and what can we expect from it?[J]. Energy & Environmental Science,2015,8(8):2296-2319. [4] Wang S Y, Li Z L, Wang G, et al. Freestanding Ti3C2Tx MXene/Prussian blue analogues films with superior ion uptake for efficient capacitive deionization by a dual pseudocapacitance effect[J]. ACS Nano,2022,16(1):1239-1249. doi: 10.1021/acsnano.1c09036 [5] Gamaethiralalage J G, Singh K, Sahin S, et al. Recent advances in ion selectivity with capacitive deionization[J]. Energy & Environmental Science,2021,14(3):1095-1120. [6] Inagaki M, Huang Z. Carbon materials for water desalination by capacitive deionization[J]. New Carbon Materials,2023,38(3):405-431. doi: 10.1016/S1872-5805(23)60736-X [7] Zornitta R L, Srimuk P, Lee J, et al. Charge and potential balancing for optimized capacitive deionization using lignin-derived, low-cost activated carbon electrodes[J]. Chemistry and sustainable chemistry,2018,11(13):2101-2113. doi: 10.1002/cssc.201800689 [8] Kumar R, Sen Gupta S, Katiyar S, et al. Carbon aerogels through organo-inorganic co-assembly and their application in water desalination by capacitive deionization[J]. Carbon,2016,99:375-383. doi: 10.1016/j.carbon.2015.12.004 [9] Wang L, Wang M, Huang Z H, et al. Capacitive deionization of NaCl solutions using carbon nanotube sponge electrodes[J]. Journal of Materials Chemistry,2011,21(45):18295-18299. doi: 10.1039/c1jm13105b [10] Li Z, Song B, Wu Z K, et al. 3D porous graphene with ultrahigh surface area for microscale capacitive deionization[J]. Nano Energy,2015,11:711-718. doi: 10.1016/j.nanoen.2014.11.018 [11] Wang H, Edano L, Valentino L, et al. Capacitive deionization using carbon derived from an array of zeolitic-imidazolate frameworks[J]. Nano Energy,2020,77:105304. doi: 10.1016/j.nanoen.2020.105304 [12] Li Q, Zheng Y, Xiao D J, et al. Faradaic electrodes open a new era for capacitive deionization[J]. Advanced Science,2020,7(22):2002213. doi: 10.1002/advs.202002213 [13] Sun K G, Tebyetekerwa M, Wang C, et al. Electrocapacitive deionization: mechanisms, electrodes and cell designs[J]. Advanced Functional Materials,2023,33(18):2213578. doi: 10.1002/adfm.202213578 [14] Chang J J, Li Y P, Duan F, et al. Selective removal of chloride ions by bismuth electrode in capacitive deionization[J]. Separation and Purification Technology,2020,240:116600. doi: 10.1016/j.seppur.2020.116600 [15] Hao Z W, Sun X Q, Cheng J B, et al. Recent progress and challenges in faradic capacitive desalination: From mechanism to performance[J]. Small,2023,19(33):2300253. doi: 10.1002/smll.202300253 [16] Pan Q F, Zheng F H, Deng D F, et al. Interlayer spacing regulation of NiCo-LDH nanosheets with ultrahigh specific capacity for battery-type supercapacitors[J]. ACS Applied Materials & Interfaces,2021,13(47):56692-56703. [17] Wang Y, Pan Q F, Qiao Y X, et al. Layered metal oxide nanosheets with enhanced interlayer space for electrochemical deionization[J]. Advanced Materials,2023,35(15):2210871. [18] Zhang Z H, Li H B. Promoting the uptake of chloride ions by ZnCo–Cl layered double hydroxide electrodes for enhanced capacitive deionization[J]. Environmental Science:Nano,2021,8(7):1886-1895. doi: 10.1039/D1EN00350J [19] Zhang D D, Cao J, Zhang X Y, et al. NiMn layered double hydroxide nanosheets in-situ anchored on Ti3C2 MXene via chemical bonds for auperior supercapacitors[J]. ACS Applied Energy Materials,2020,3(6):5949-5964. doi: 10.1021/acsaem.0c00863 [20] Li C Y, Zhang D D, Cao J, et al. Ti3C2 MXene-encapsulated NiFe-LDH hybrid anode for high-performance lithium-ion batteries and capacitors[J]. ACS Applied Energy Materials,2021,4(8):7821-7828. doi: 10.1021/acsaem.1c01171 [21] Wei D, Cao Y Y, Yan L J, et al. Enhanced pseudo-capacitance process in nanoarchitectural layered double hydroxide nanoarrays hollow nanocages for improved capacitive deionization performance[J]. ACS applied materials & interfaces,2023,15(20):24427-24436. [22] Xi W, Li H B. Vertically-aligned growth of CuAl-layered double oxides on reduced graphene oxide for hybrid capacitive deionization with superior performance[J]. Environmental Science-Nano,2020,7(3):764-772. doi: 10.1039/C9EN01238A [23] Lei J J, Xiong Y C, Yu F, et al. Flexible self-supporting CoFe-LDH/MXene film as a chloride ions storage electrode in capacitive deionization[J]. Chemical Engineering Journal,2022,437:135381. doi: 10.1016/j.cej.2022.135381 [24] Dou S, Tao L, Wang R L, et al. Plasma‐assisted synthesis and surface modification of electrode materials for renewable energy[J]. Advanced Materials,2018,30(21):1705850. doi: 10.1002/adma.201705850 [25] Liu Z J, Zhao Z H, Wang Y Y, et al. In situ exfoliated, edge-rich, oxygen‐functionalized graphene from carbon fibers for oxygen electrocatalysis[J]. Advanced Materials,2017,29(18):1606207. doi: 10.1002/adma.201606207 [26] Chen C, Tao L, Du S Q, et al. Advanced exfoliation strategies for layered double hydroxides and applications in energy conversion and storage[J]. Advanced Functional Materials,2020,30(14):1909832. doi: 10.1002/adfm.201909832 [27] Wang Y Y, Xie C, Zhang Z Y, et al. In situ exfoliated, N-doped, and edge-rich ultrathin layered double hydroxides Nanosheets for oxygen evolution reaction[J]. Advanced Functional Materials,2017,28(4):1703363. [28] Liu H Z, Ji X L, Guo Z H, et al. A high-current hydrogel generator with engineered mechanoionic asymmetry[J]. Nature Communications,2024,15(1):1494-1504. doi: 10.1038/s41467-024-45931-7 [29] Zhang C J, Wang D, Wang Z, et al. Boosting capacitive deionization performance of commercial carbon fibers cloth via structural regulation based on catalytic-etching effect[J]. Energy & Environmental Materials,2023,6(1):e12276. [30] Jing C, Dong B Q, Zhang Y X. Chemical modifications of layered double hydroxides in the supercapacitor[J]. Energy & Environmental Materials,2020,3(3):346-379. [31] Gao X R, Wang P K, Pan Z H, et al. Recent progress in two‐dimensional layered double hydroxides and their derivatives for supercapacitors[J]. Chemistry and sustainable chemistry,2020,13(6):1226-1254. doi: 10.1002/cssc.201902753 [32] Fan X Z, Pang Q Q, Yi S S, et al. Intrinsic-structural-modulated carbon cloth as efficient electrocatalyst for water oxidation[J]. Applied Catalysis B-Environmental,2021,292(5):120152. [33] Gao X Y, Zhao Y F, Dai K Q, et al. NiCoP nanowire@NiCo-layered double hydroxides nanosheet heterostructure for flexible asymmetric supercapacitors[J]. Chemical Engineering Journal,2020,384:123373. doi: 10.1016/j.cej.2019.123373 [34] Qiu W L, Li G R, Luo D, et al. Hierarchical micro-nanoclusters of bimetallic layered hydroxide polyhedrons as advanced sulfur reservoir for high-performance lithium-sulfur batteries[J]. Advanced Science,2021,8(7):2003400. doi: 10.1002/advs.202003400 [35] Sun H M, Miao Y L, Wu T, et al. Exfoliation of bimetallic (Ni, Co) carbonate hydroxide nanowires by Ar plasma for enhanced oxygen evolution[J]. Chemical Communications,2020,56(6):872-875. doi: 10.1039/C9CC08841E [36] Zhai C X, Yuan J H, Wang Y B, et al. Rectorite in flow-electrode capacitive deionization with three-dimensional current collector to achieve cost-effective desalination[J]. Desalination,2024,573:117217. doi: 10.1016/j.desal.2023.117217 [37] Zhang R J, Dong J D, Zhang W, et al. Synergistically coupling of 3D FeNi-LDH arrays with Ti3C2Tx-MXene nanosheets toward superior symmetric supercapacitor[J]. Nano Energy,2022,91:106633. doi: 10.1016/j.nanoen.2021.106633 [38] Wang H Y, Wei D, He Y J, et al. Carbon nanoarchitectonics with Bi nanoparticle encapsulation for improved electrochemical deionization performance[J]. ACS Applied Materials & Interfaces,2022,14(11):13177-13185. [39] Wang K, Liu Y, Ding Z B, et al. Chloride pre-intercalated CoFe-layered double hydroxide as chloride ion capturing electrode for capacitive deionization[J]. Chemical Engineering Journal,2022,433:133578. doi: 10.1016/j.cej.2021.133578 [40] Cai Y M, Wang Y, Fang R L, et al. Flexible structural engineering of PPy-NiCo-LDH@Mxene for improved capacitive deionization and efficient hard water softening process[J]. Separation and Purification Technology,2022,280:119828. doi: 10.1016/j.seppur.2021.119828 -





 下载:
下载: