Carbon-based metal-free nanomaterials for the electrosynthesis of small-molecule chemicals: A review
-
摘要: 电催化是各种以化学品形式储存可再生电力能源技术的核心。目前,贵金属基催化剂广泛应用于提高电催化转化效率。然而,成本高、稳定性差等缺点严重阻碍了其在电合成和可持续能源器件中的大规模应用。碳基无金属催化剂(CMFCs)在提高催化性能方面展现出巨大潜力,且受到越来越多的关注。本文概述了用于电催化合成的CMFCs的最新研究进展,并讨论了其催化机理和设计策略。此外,简要总结了电催化合成过氧化氢、氨、氯以及各种碳基和氮基化合物的研究现状,并重点阐述了CMFCs目前面临的挑战和未来前景。Abstract: Electrocatalysis is a key component of many clean energy technologies that has the potential to store renewable electricity in chemical form. Currently, noble metal-based catalysts are most widely used for improving the conversion efficiency of reactants during the electrocatalytic process. However, drawbacks such as high cost and poor stability seriously hinder their large-scale use in this process and in sustainable energy devices. Carbon-based metal-free catalysts (CMFCs) have received growing attention due to their enormous potential for improving the catalytic performance. This review gives a concise comprehensive overview of recent developments in CMFCs for electrosynthesis. First, the fundamental catalytic mechanisms and design strategies of CMFCs are presented and discussed. Then, a brief overview of various electrosynthesis processes, including the synthesis of hydrogen peroxide, ammonia, chlorine, as well as various carbon- and nitrogen-based compounds is given. Finally, current challenges and prospects for CMFCs are highlighted.
-
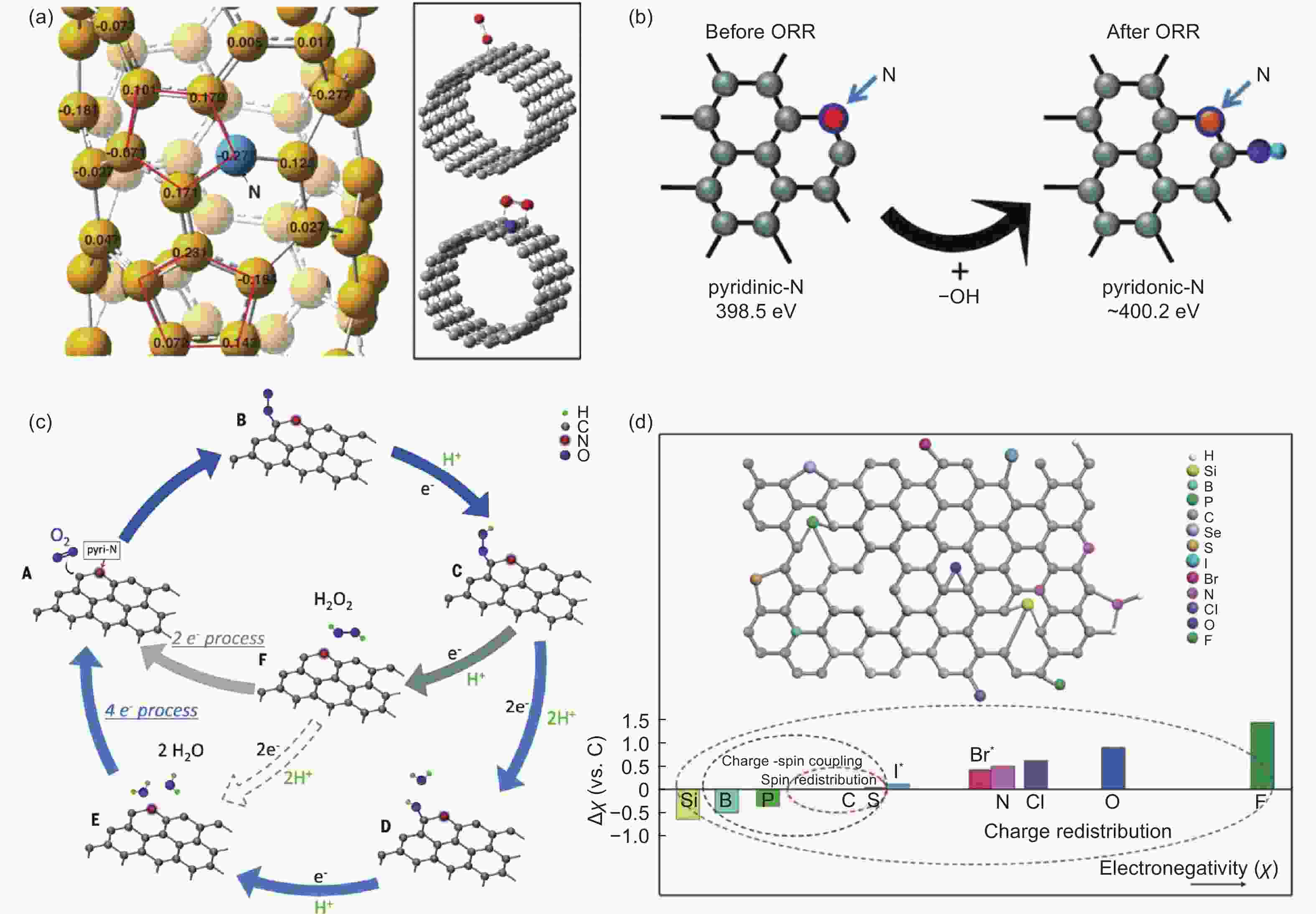
Figure 1. (a) Calculated charge density distribution of N-doped CNTs and the corresponding adsorption modes of oxygen molecule[27]. Reproduced with permission from AAAS. (b) Reaction process between pyridinic N and OH species. (c) Proposed mechanism for ORR on nitrogen-doped carbon materials[59]. Reproduced with permission from AAAS. (d) Heteroatom-doping mechanism in CMFCs[60]. Reproduced with permission from Wiley-VCH
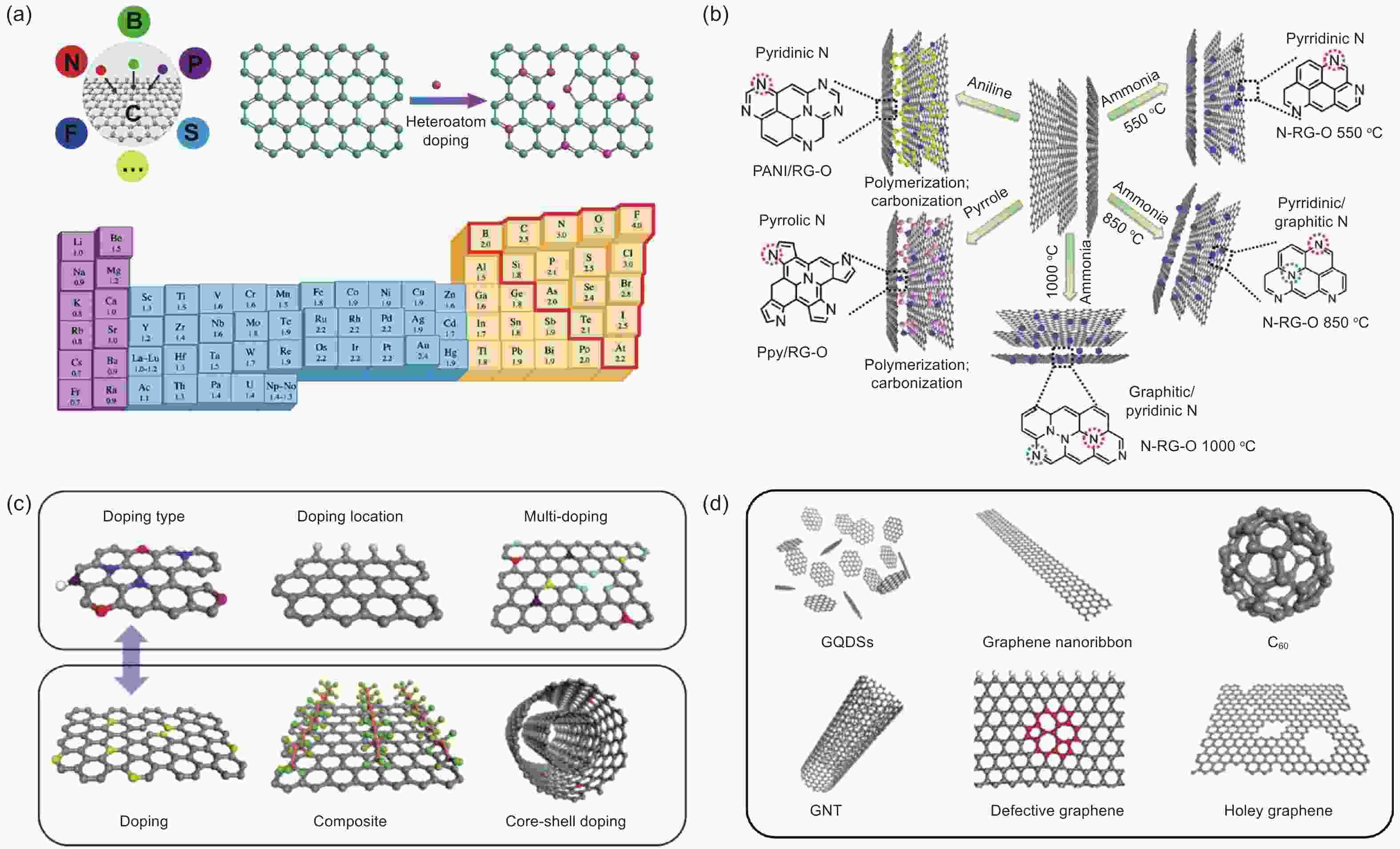
Figure 2. (a) Heteroatoms-doping strategies and the electronegativity of various elements[26]. Reproduced with permission from American Chemical Society. (b) Schematic diagram of preparation for controlled N-doped graphene[81]. Reproduced with permission from The Royal Society of Chemistry. (c, d) Design principles for various CMFCs[87]. Reproduced with permission from Wiley-VCH
Figure 3. (a, b) TEM and HRTEM images of edge-rich graphitic ordered mesoporous carbon (GOMC), (c) The stability test of o-GOMC-1 for H2O2 electrosynthesis[96]. Reproduced with permission from Wiley-VCH. (d) O 1s spectrum of O-CNTs. (e) ORR polarization curves for SP and AB-based CMFCs. (f) Calculated two-electron ORR-related volcano plot via the function of ΔGOOH*[62]. Reproduced with permission from Nature Publishing Group. (g) The mechanism of H2O2 electrosynthesis for N-doped CNH[98]. Reproduced with permission from Elsevier Inc. (h) Free energy profile of two- and four-electron ORR pathways for different N-doped CMFCs[63]. Reproduced with permission from Nature Publishing Group. (i) The possible two-electron ORR mechanism of O-DG-30[64]. Reproduced with permission from Nature Publishing Group
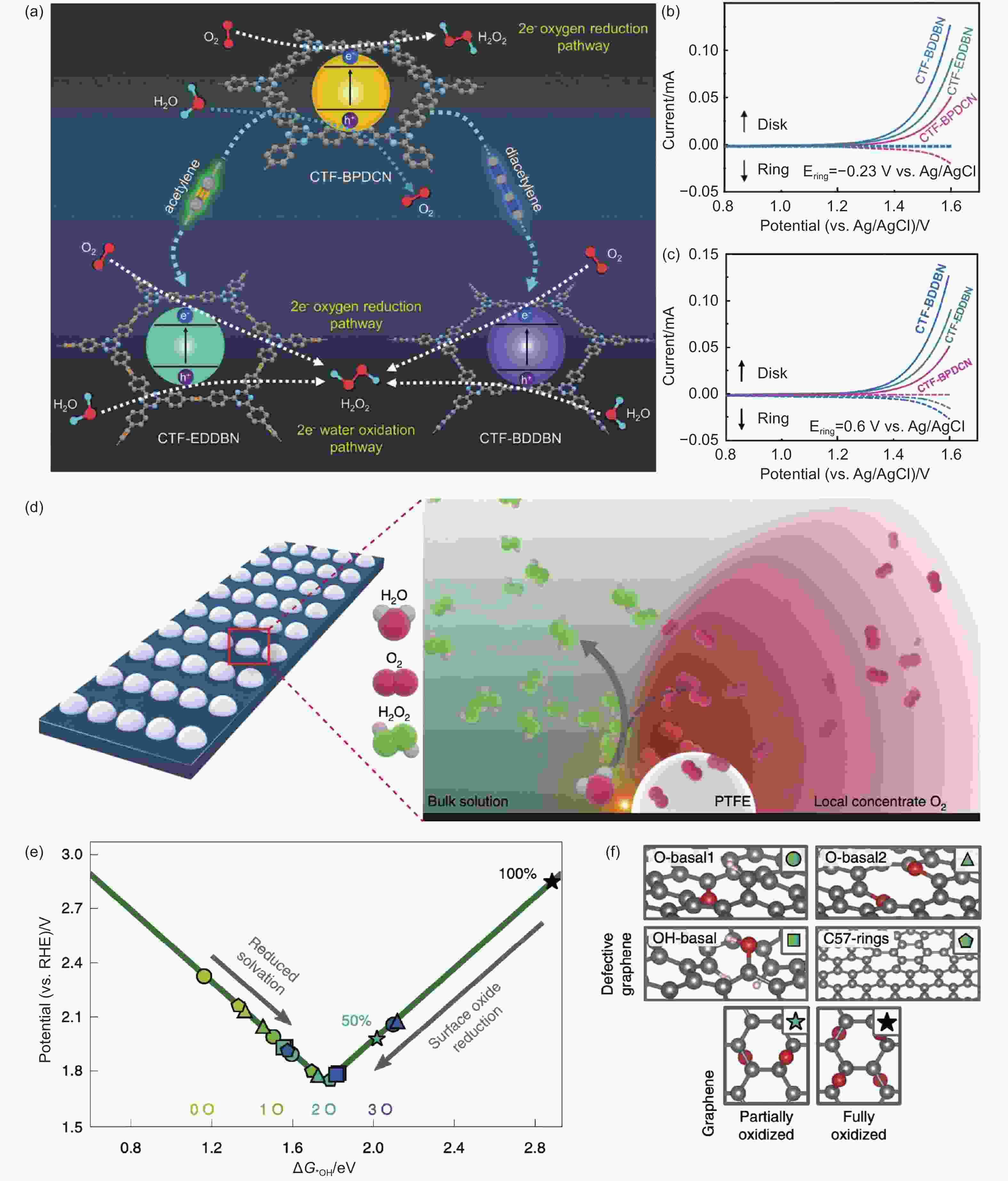
Figure 4. (a) Schematic illustration of reaction pathways for different acetylene-based polymer, (b, c) WOR polarization curves of different acetylene-based polymer, the potential of Pt ring were set to -0.23 V and 0.6 V vs. Ag/AgCl to detect O2 and H2O2, respectively[71]. Reproduced with permission from Wiley-VCH. (d) The possible reaction pathway of PTFE-coated carbon electrode. (e) The data points depict *OH binding energies on different carbon-based metal-free nanomaterials. (f) The coverages of oxygen atoms in different carbon-based defected structures surface[99]. Reproduced with permission from Nature Publishing Group
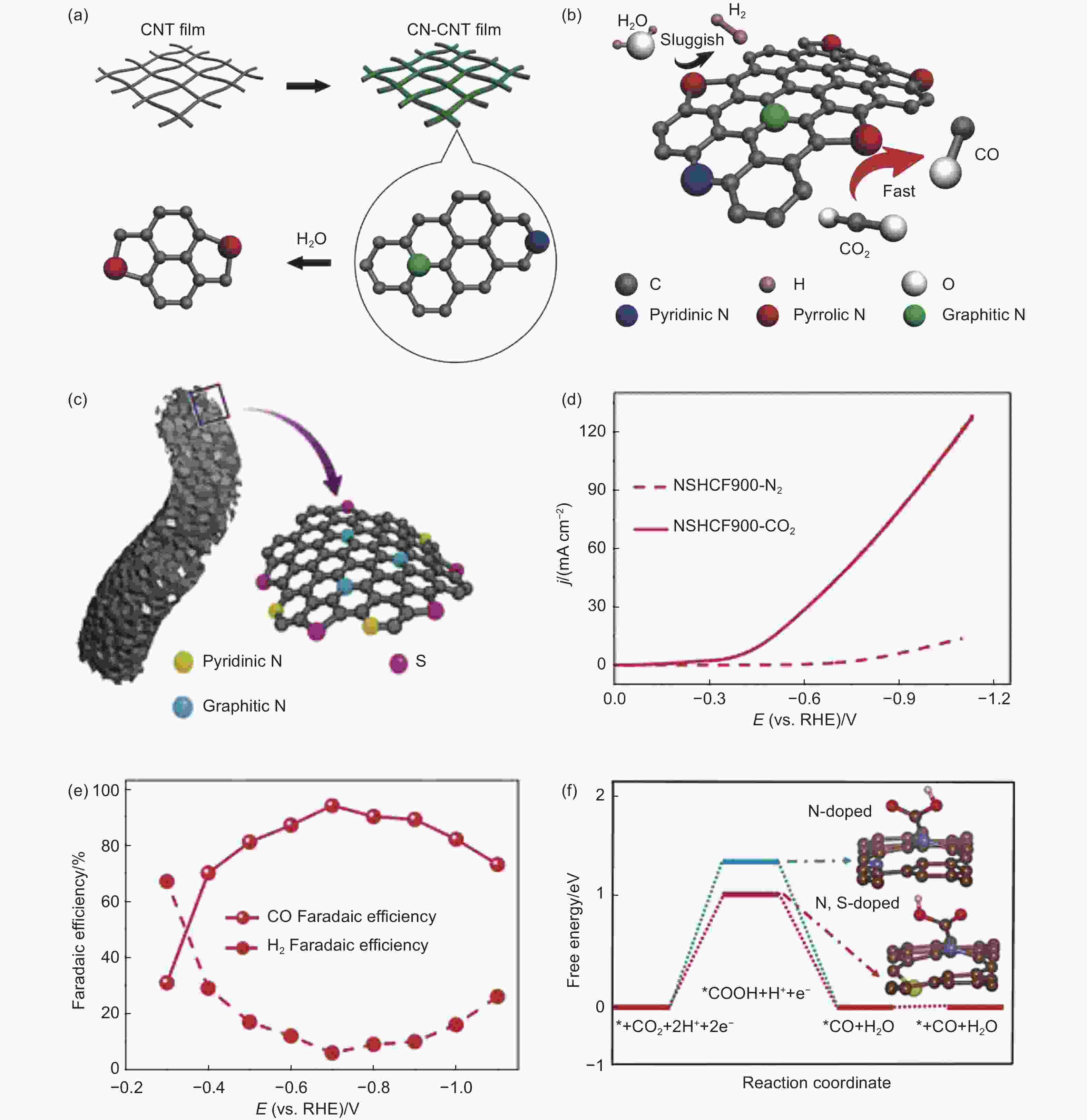
Figure 5. (a, b) The synthesis illustration of N-doped CMFCs and the corresponding catalytic ability for CO2RR[76]. Reproduced with permission from Wiley-VCH. (c) Schematic diagram of the structure of NSHCF. (d-f) Polarization curves, FE, and CO2RR free energy of NSHCF900 catalyst[102]. Reproduced with permission from Wiley-VCH
Figure 6. (a, b) Schematic illustration of synthesis procedures, the stability and corresponding formate FE of N-C61 catalyst. (c) Tafel plots comparison for N-C61-800 and pristine C61. (d) The proposed mechanism of N-C61 electrocatalysts for CO2RR[104]. Reproduced with permission from Royal Society of Chemistry. (e) TEM and HRTEM images, (f, g) FE of CH4 and
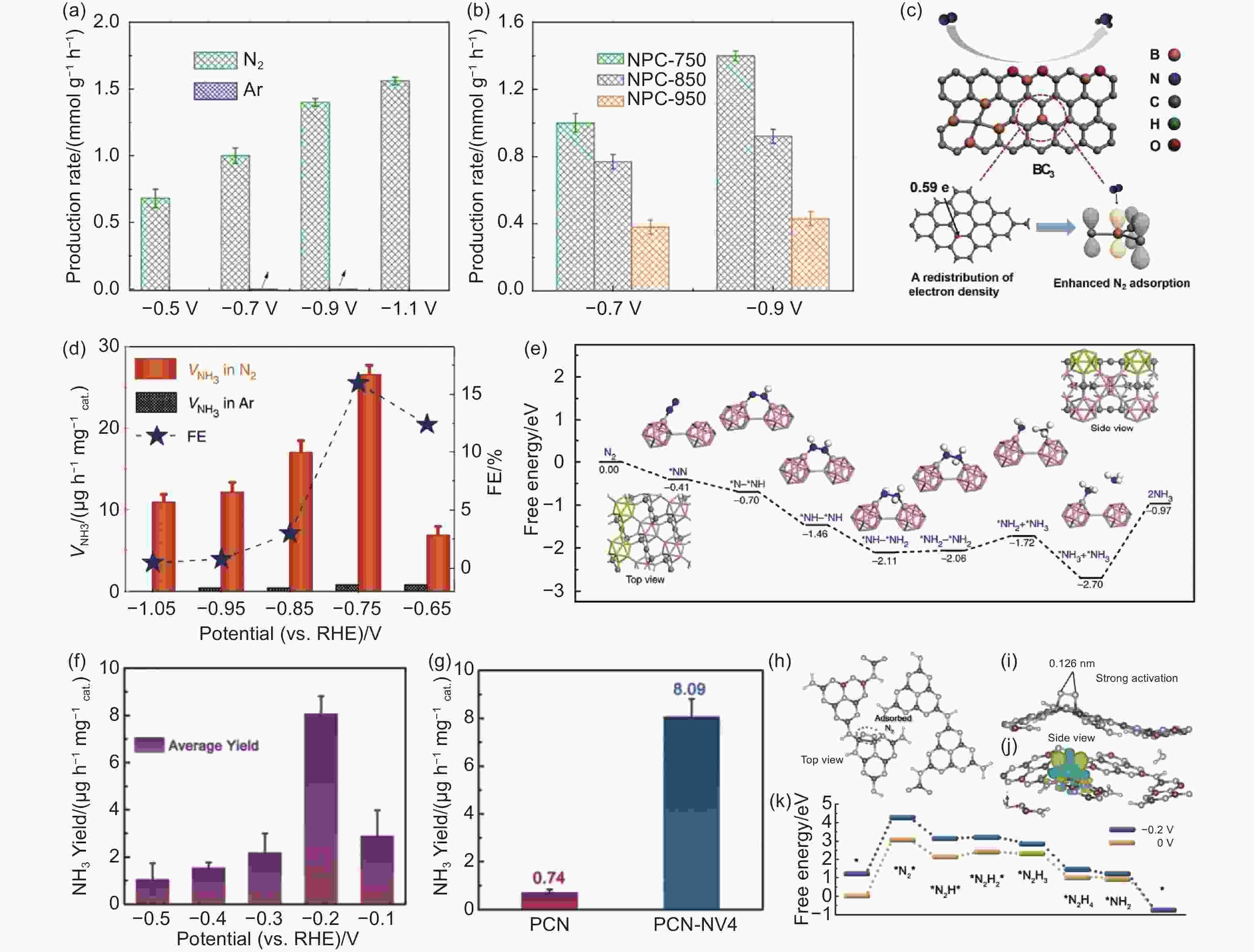
$j_{{{\rm{CH}}}_4}$ for GQD-NH2-H and GQD-NH2-L electrocatalysts[105]. Reproduced with permission from Nature Publishing GroupFigure 7. (a, b) The performance of ammonia production for different N-doped porous carbons[18]. Reproduced with permission from American Chemical Society. (c) Structural illustration of B-doped graphene and the corresponding BC3 sites for adsorbing N2[19]. Reproduced with permission from Elsevier Inc. (d, e) NH3 yields, FEs and DFT calculations for NRR on the B4C (110) surface[113]. Reproduced with permission from Nature Publishing Group. (f, g) NH3 yields of PCN-based catalysts. (h-j) N2 adsorption model and charge density difference on “N” vacancy of PCN. (k) Free energy diagram toward NRR for PCN[114]. Reproduced with permission from Wiley-VCH
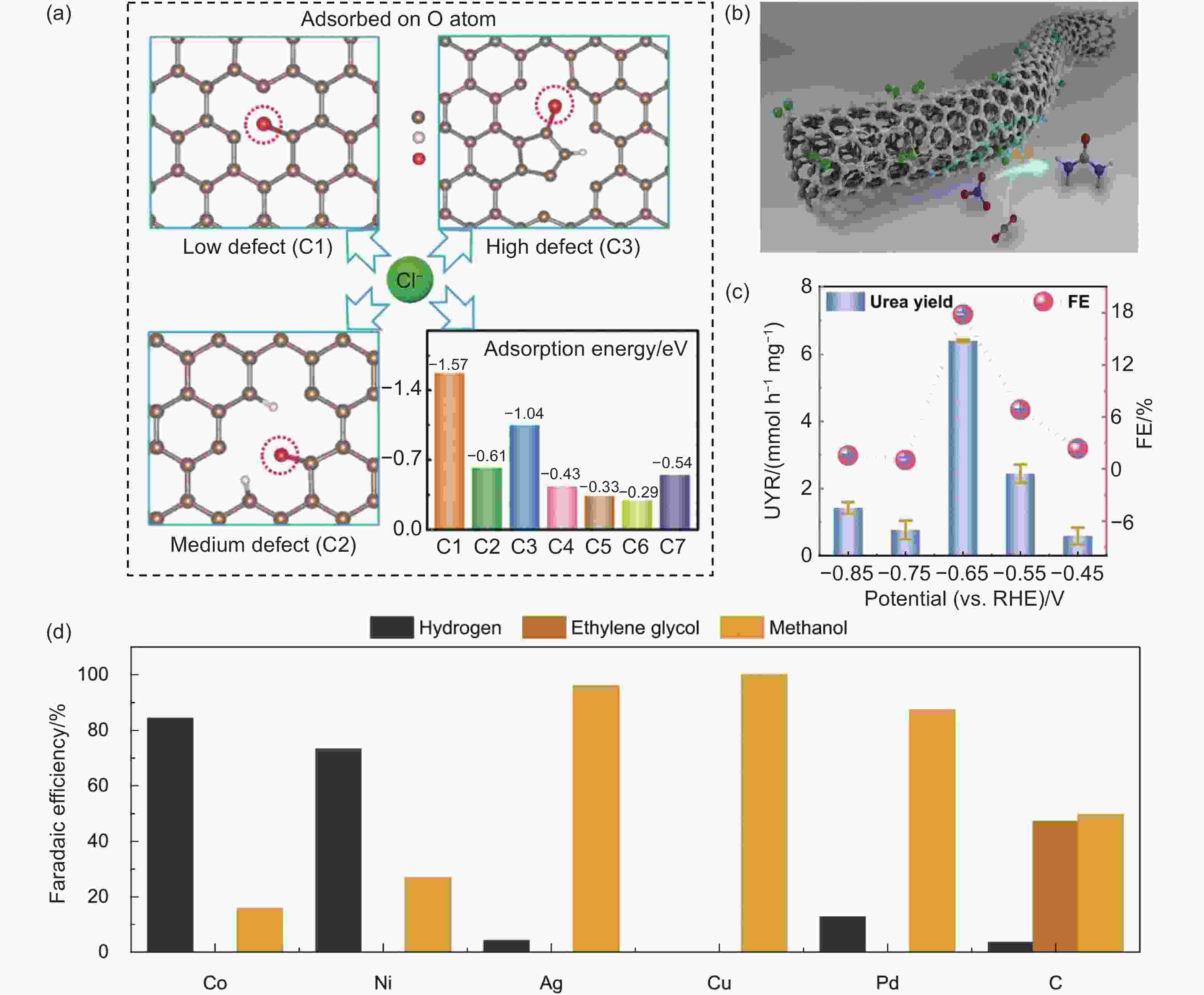
Figure 8. (a) The Cl adsorption structure at different O sites[120]. Reproduced with permission from Wiley-VCH. (b, c) Schematic illustration and performance of urea synthesis on the F-CNT[121]. Reproduced with permission from Elsevier Inc. (d) The FE of electrochemical formaldehyde reduction for various metal/metal-free catalysts[123]. Reproduced with permission from Nature Publishing Group
-
[1] Leech M, Lam K. A practical guide to electrosynthesis[J]. Nature Reviews Chemistry,2022,6:275-286. doi: 10.1038/s41570-022-00372-y [2] Qi J, Du Y, Yang Q, et al. Energy-saving and product-oriented hydrogen peroxide electrosynthesis enabled by electrochemistry pairing and product engineering[J]. Nature Communcation,2023,14:6263. doi: 10.1038/s41467-023-41997-x [3] Zhao S, Yang Y, Tang Z. Insight into structural evolution, active sites, and stability of heterogeneous electrocatalysts[J]. Angewandte Chemie International Edition,2022,61:e202110186. doi: 10.1002/anie.202110186 [4] Zhang B, Cao L, Tang C, et al. Atomically dispersed dual-site cathode with a record high sulfur mass loading for high-performance room-temperature sodium-sulfur batteries[J]. Advanced Materials,2023,35:2206828. doi: 10.1002/adma.202206828 [5] Yang Q, Qu X, Cui H, et al. Rechargeable aqueous Mn-metal battery enabled by inorganic-organic interfaces[J]. Angewandte Chemie International Edition,2022,61:e202206471. doi: 10.1002/anie.202206471 [6] Lin X, Shi L, Liu F, et al. Large-scale production of holey carbon nanosheets implanted with atomically dispersed Fe sites for boosting oxygen reduction electrocatalysis[J]. Nano Research, 2022, 15: 1926-1933. [7] Shi L, Lin X, Liu F, et al. Geometrically deformed iron-based single-atom catalysts for high-performance acidic proton exchange membrane fuel cells[J]. ACS Catalysis,2022,12:5397-5406. doi: 10.1021/acscatal.2c00915 [8] Wu K, Liu Y, Tan X, et al. Regulating electron transfer over asymmetric low-spin Co (II) for highly selective electrocatalysis[J]. Chem Catalysis,2023,2(2):372-385. [9] Liu F, Shi L, Lin X, et al. Fe/Co dual metal catalysts modulated by S-ligands for efficient acidic oxygen reduction in PEMFC[J]. Science Advances,2023,9:eadg0366. doi: 10.1126/sciadv.adg0366 [10] Wu Z, Liu X, Li H, et al. A semiconductor-electrocatalyst nano interface constructed for successive photoelectrochemical water oxidation[J]. Nature Communications,2023,14:2574. doi: 10.1038/s41467-023-38285-z [11] Shi L, Qi Z, Peng P, et al. Pyrene-based covalent organic polymers for enhanced photovoltaic performance and solar-driven hydrogen production[J]. ACS Applied Energy Materials,2018,1:7007-7013. doi: 10.1021/acsaem.8b01432 [12] Zhang S, Tan C, Yan R, et al. Constructing built-in electric field in heterogeneous nanowire arrays for efficient overall water electrolysis[J]. Angewandte Chemie International Edition,2023,135:e202302795. [13] Pei Z, Tan H, Gu J, et al. A polymeric hydrogel electrocatalyst for direct water oxidation[J]. Nature Communications,2023,14:818. doi: 10.1038/s41467-023-36532-x [14] Liu Y, Wang Y, Zhao S, et al. Metal-organic framework-based nanomaterials for electrocatalytic oxygen evolution[J]. Small Methods,2023,6:2200773. [15] Liu C, Colón B C, Ziesack M, et al. Water splitting-biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis[J]. Science,2016,352:1210-1213. doi: 10.1126/science.aaf5039 [16] Ma X, Shi L, Zhang L, et al. Tuning B-N pairs in porous carbon nanorods for electrochemical conversion of CO2 to syngas with controllable CO/H2 ratios[J]. Sustainable Energy & Fuels,2023,7:661-670. [17] Eizawa A, Arashiba K, Tanaka H, et al. Remarkable catalytic activity of dinitrogen-bridged dimolybdenum complexes bearing NHC-based PCP-pincer ligands toward nitrogen fixation[J]. Nature Communications,2017,8:14874. doi: 10.1038/ncomms14874 [18] Huang Z L, Madiha Rafiq, Abebe Reda Woldu, at al. Recent progress in electrocatalytic nitrogen reduction to ammonia (NRR)[J]. Coordination Chemistry Reviews,2023,478(1):214981. [19] Yu X, Han P, Wei Z, et al. Boron-doped graphene for electrocatalytic N2 reduction[J]. Joule,2018,2:1610-1622. doi: 10.1016/j.joule.2018.06.007 [20] Liu Y, Li C, Tan C, et al. Electrosynthesis of chlorine from seawater-like solution through single-atom catalysts[J]. Nature Communications,2023,14:2475. doi: 10.1038/s41467-023-38129-w [21] Liu X, Kumar P, Chen Q, et al. Activating lattice oxygen in layered lithium oxides through cation vacancies for enhanced urea electrolysis[J]. Angewandte Chemie International Edition, 2022, 134: e202206050. [22] Wang Z, Huang J, Wang L, et al. Cation-tuning induced d-band center modulation on Co-based spinel oxide for oxygen reduction/evolution reaction[J]. Angewandte Chemie International Edition,2022,134:e202114696. [23] Zhou Z, Kong Y, Tan H, et al. Cation-vacancy-enriched nickel phosphide for efficient electrosynthesis of hydrogen peroxides[J]. Advanced Materials,2022,34:2106541. doi: 10.1002/adma.202106541 [24] Ge R, Dong L, Hu X, et al. Intensified coupled electrolysis of CO2 and brine over electrocatalysts with ordered mesoporous transport channels[J]. Chemical Engineering Journal,2022,438:135500. doi: 10.1016/j.cej.2022.135500 [25] Cui H, Wang T, Huang Z, et al. High-voltage organic cathodes for Zinc-ion batteries through electron cloud and solvation structure regulation[J]. Angewandte Chemie International Edition,2022,134:e202203453. [26] Dai L, Xue Y, Qu L, et al. Metal-free catalysts for oxygen reduction reaction[J]. Chemical Reviews,2015,115:4823-4892. doi: 10.1021/cr5003563 [27] Gong K, Du F, Xia Z, et al. Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction[J]. Science,2009,323:760-764. doi: 10.1126/science.1168049 [28] Wang S, Zhang L, Xia Z, et al. BCN Graphene as efficient metal-free electrocatalyst for the oxygen reduction reaction[J]. Angewandte Chemie International Edition,2012,51:4209-4212. doi: 10.1002/anie.201109257 [29] Zhao Y, Yang L, Chen S, et al. Can boron and nitrogen co-doping improve oxygen reduction reaction activity of carbon nanotubes[J]. Journal of the American Chemical Society,2013,135:1201-1204. doi: 10.1021/ja310566z [30] Baptista F, Belhout S, Giordani S, et al. Recent developments in carbon nanomaterial sensors[J]. Chemical Society Reviews,2015,44:4433-4453. doi: 10.1039/C4CS00379A [31] Yang Q, Jiang N, Shao Y, et al. Functional carbon materials addressing dendrite problems in metal batteries: Surface chemistry, multi-dimensional structure engineering and defects[J]. Science China Chemistry,2022,65:2351-2368. doi: 10.1007/s11426-022-1397-2 [32] Hu C, Paul R, Dai Q, et al. Carbon-based metal-free electrocatalysts: From oxygen reduction to multifunctional electrocatalysis[J]. Chemical Society Review,2021,50:11785-11843. doi: 10.1039/D1CS00219H [33] Cui P, Zhao L, Long Y, et al. Carbon-based electrocatalysts for acidic oxygen reduction reaction[J]. Angewandte Chemie International Edition,2023,62:e202218269. doi: 10.1002/anie.202218269 [34] Higgins D, Zamani P, Yu A, et al. The application of graphene and its composites in oxygen reduction electrocatalysis: A perspective and review of recent progress[J]. Energy & Environmental Science,2016,9:357-390. [35] Qu L, Liu Y, Baek J B, et al. Nitrogen-doped graphene as efficient metal-free electrocatalyst for oxygen reduction in fuel vells[J]. ACS Nano,2010,4:1321-1326. doi: 10.1021/nn901850u [36] Zheng Y, Jiao Y, Zhu Y, et al. Hydrogen evolution by a metal-free electrocatalyst[J]. Nature Communications,2014,5:3783. doi: 10.1038/ncomms4783 [37] Zheng Y, Jiao Y, Jaroniec M, et al. Advancing the electrochemistry of the hydrogen-evolution reaction through combining experiment and theory[J]. Angewandte Chemie International Edition,2015,54:52-65. doi: 10.1002/anie.201407031 [38] Suen N, Hung S, Quan Q, et al. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives[J]. Chemical Society Reviews,2017,46:337-365. doi: 10.1039/C6CS00328A [39] Zheng Y, Jiao Y, Zhu Y, et al. Molecule-level g-C3N4 coordinated transition metals as a new class of electrocatalysts for oxygen electrode reactions[J]. Journal of the American Chemical Society,2017,139:3336-3339. doi: 10.1021/jacs.6b13100 [40] Duan X, Xu J, Wei Z, et al. Metal-free carbon materials for CO2 electrochemical reduction[J]. Advanced Materials,2017,29:1701784. doi: 10.1002/adma.201701784 [41] Liu Y, Su Y, Quan X, et al. Facile ammonia synthesis from electrocatalytic N2 reduction under ambient conditions on N-doped porous carbon[J]. ACS Catalysis,2018,8:1186-1191. doi: 10.1021/acscatal.7b02165 [42] Zhang J, Zhao Z, Xia Z, et al. A metal-free bifunctional electrocatalyst for oxygen reduction and oxygen evolution reactions[J]. Nature Nanotechnology,2015,10:444. doi: 10.1038/nnano.2015.48 [43] Yang H, Miao J, Hung S, et al. Identification of catalytic sites for oxygen reduction and oxygen evolution in N-doped graphene materials: Development of highly efficient metal-free bifunctional electrocatalyst[J]. Science Advances,2016,2:e150112. [44] Zhang J, Xia Z, Dai L. Carbon-based electrocatalysts for advanced energy conversion and storage[J]. Science Advances,2015,1:e1500564. doi: 10.1126/sciadv.1500564 [45] Ma T, Ran J, Dai S, et al. Phosphorus-doped graphitic carbon nitrides grown in situ on carbon-fiber paper: Flexible and reversible oxygen electrodes[J]. Angewandte Chemie International Edition,2015,54:4646-4650. doi: 10.1002/anie.201411125 [46] Zhang J, Qu L, Shi G, et al. N, P-codoped carbon networks as efficient metal-free bifunctional catalysts for oxygen reduction and hydrogen evolution reactions[J]. Angewandte Chemie International Edition,2016,55:2230-2234. doi: 10.1002/anie.201510495 [47] Hu C, Dai L. Carbon-based metal-free catalysts for electrocatalysis beyond the ORR[J]. Angewandte Chemie International Edition,2016,55:11736-11758. doi: 10.1002/anie.201509982 [48] Tabassum H, Zou R, Mahmood A, et al. A catalyst-free synthesis of B, N co-doped graphene nanostructures with tunable dimensions as highly efficient metal free dual electrocatalysts[J]. Journal of Materials Chemistry A,2016,4:16469-16475. doi: 10.1039/C6TA07214C [49] Mulyadi A, Zhang Z, Dutzer M, et al. Facile approach for synthesis of doped carbon electrocatalyst from cellulose nanofibrils toward high-performance metal-free oxygen reduction and hydrogen evolution[J]. Nano Energy,2017,32:336-346. doi: 10.1016/j.nanoen.2016.12.057 [50] Zhang J, Dai L. Nitrogen, phosphorus and fluorine tri-doped graphene as a multifunctional catalyst for self-powered electrochemical water splitting[J]. Angewandte Chemie International Edition,2016,128:13490-13494. [51] Hu C, Dai L. Multifunctional carbon-based metal-free electrocatalysts for simultaneous oxygen reduction, oxygen evolution, and hydrogen evolution[J]. Advanced Materials,2017,29:1604942. doi: 10.1002/adma.201604942 [52] Guo H, Feng Q C, Zh J X, et al. Cobalt nanoparticle-embedded nitrogen-doped carbon/carbon nanotube frameworks derived from a metal-organic framework for tri-functional ORR, OER and HER electrocatalysis[J]. Journal of Materials Chemistry A,2019,7:3664-3672. [53] Fan W, Zhang B, Wang X, et al. Efficient hydrogen peroxide synthesis by metal-free polyterthiophene via photoelectrocatalytic dioxygen reduction[J]. Energy & Environmental Science,2020,13:238-245. [54] Hu X, Zeng X, Liu Y, et al. Carbon-based materials for photo-and electrocatalytic synthesis of hydrogen peroxide[J]. Nanoscale,2020,12:16008-16027. doi: 10.1039/D0NR03178J [55] Shinde S, Lee C, Sami A, et al. Scalable 3D carbon nitride sponge as an efficient metal-free bifunctional oxygen electrocatalyst for rechargeable Zn-air batteries[J]. ACS Nano,2017,11:347-357. doi: 10.1021/acsnano.6b05914 [56] Zhu D, Zhao Q, Fan G, et al. Photoinduced oxygen reduction reaction boosts the output voltage of a Zinc-air battery[J]. Angewandte Chemie International Edition,2019,131:12590-12594. [57] Liu, X, Dai, L. Carbon-based metal-free catalysts[J]. Nature Reviews Materials,2016,1:16064. doi: 10.1038/natrevmats.2016.64 [58] Wang S, Yu D, Dai L, et al. Polyelectrolyte functionalized carbon nanotubes as efficient metal-free electrocatalysts for oxygen reduction[J]. Journal of the American Chemical Society 2011, 133: 5182-5185. [59] Guo D, Shibuya R, Akiba C, et al. Active sites of nitrogen-doped carbon materials for oxygen reduction reaction clarified using model catalysts[J]. Science,2016,351:361-365. doi: 10.1126/science.aad0832 [60] Gao K, Wang B, Tao L, et al. Efficient metal-free electrocatalysts from N-doped carbon nanomaterials: Mono-doping and co-doping[J]. Advanced Materials,2019,31:1805121. doi: 10.1002/adma.201805121 [61] Li L, Tang C, Zheng Y, et al. Tailoring selectivity of electrochemical hydrogen peroxide generation by tunable pyrrolic-nitrogen-carbon[J]. Advanced Energy Materials,2020,10:2000789. doi: 10.1002/aenm.202000789 [62] Lu Z, Chen G, Siahrostami S, et al. High-efficiency oxygen reduction to hydrogen peroxide catalysed by oxidized carbon materials[J]. Nature Catalysis,2018,1:156-162. doi: 10.1038/s41929-017-0017-x [63] Peng W, Liu J, Liu X, et al. Facilitating two-electron oxygen reduction with pyrrolic nitrogen sites for electrochemical hydrogen peroxide production[J]. Nature Communications,2023,14:4430. doi: 10.1038/s41467-023-40118-y [64] Wu Q, Zou H, Mao X, et al. Unveiling the dynamic active site of defective carbon-based electrocatalysts for hydrogen peroxide production[J]. Nature Communications,2023,14:6275. doi: 10.1038/s41467-023-41947-7 [65] Li M, Zhang L, Xu Q, et al. N-doped graphene as catalysts for oxygen reduction and oxygen evolution reactions: Theoretical considerations[J]. Journal of Catalysis,2014,314:66-72. doi: 10.1016/j.jcat.2014.03.011 [66] Kundu S, Malik B, Pattanayak D, et al. Role of dpecific N-containing active sites in interconnected graphene quantum dots for the enhanced electrocatalytic activity towards oxygen evolution reaction[J]. ChemistrySelect,2017,2:9943-9946. doi: 10.1002/slct.201701952 [67] Lu X, Yim W, Suryanto B, et al. Electrocatalytic oxygen evolution at surface-oxidized multiwall carbon nanotubes[J]. Journal of the American Chemical Society,2015,137:2901-2907. doi: 10.1021/ja509879r [68] El-Sawy A, Mosa I, Su D, et al. Controlling the active sites of sulfur-doped carbon nanotube-graphene nanolobes for highly efficient oxygen evolution and reduction catalysis[J]. Advanced Energy Materials,2016,6:1501966. doi: 10.1002/aenm.201501966 [69] Qu K, Zheng Y, Jiao Y, et al. Polydopamine-inspired, dual heteroatom-doped carbon nanotubes for highly efficient overall water splitting[J]. Advanced Energy Materials,2017,7:1602068. doi: 10.1002/aenm.201602068 [70] Mavrikis S, Goltz M, Perry S, et al. Effective hydrogen peroxide production from electrochemical water oxidation[J]. ACS Energy Letter,2021,6:2369-2377. doi: 10.1021/acsenergylett.1c00904 [71] Chen L, Wang W, Wan Y, et al. Acetylene and diacetylene functionalized covalent triazine frameworks as metal-free photocatalysts for hydrogen peroxide production: A new two-electron water oxidation pathway[J]. Advanced Materials,2020,32:1904433. doi: 10.1002/adma.201904433 [72] Benson E, Kubiak C, Sathrum A, et al. Electrocatalytic and homogeneous approaches to conversion of CO2 to liquid fuels[J]. Chemical Society Reviews,2009,38:89-99. doi: 10.1039/B804323J [73] Kumar B, Asadi M, Pisasale D, et al. Renewable and metal-free carbon nanofibre catalysts for carbon dioxide reduction[J]. Nature Communications,2013,4:2819. doi: 10.1038/ncomms3819 [74] Jia Y, Zhang L, Zhuang L, et al. Identification of active sites for acidic oxygen reduction on carbon catalysts with and without nitrogen doping[J]. Nature Catalysis,2019,2:688-695. doi: 10.1038/s41929-019-0297-4 [75] Wu J, Sharifi T, Zhang T, et al. Emerging carbon-based heterogeneous catalysts for electrochemical reduction of carbon dioxide into value-added chemicals[J]. Advanced Materials,2019,31:1804257. doi: 10.1002/adma.201804257 [76] Cui X, Pan Z, Zhang L, et al. Selective etching of nitrogen-doped carbon by steam for enhanced electrochemical CO2 reduction[J]. Advanced Energy Materials,2017,7:1701456. doi: 10.1002/aenm.201701456 [77] Natsui K, Iwakawa H, Ikemiya N, et al. Stable and highly efficient electrochemical production of formic acid from carbon dioxide using diamond electrodes[J]. Angewandte Chemie International Edition,2018,57:2639-2643. doi: 10.1002/anie.201712271 [78] Liu Y, Zhang Y, Cheng K, et al. Selective electrochemical reduction of carbon dioxide to ethanol on a boron-and nitrogen-Co-doped nanodiamond[J]. Angewandte Chemie International Edition,2018,127:15813-15817. [79] Shipman M, Symes M. Recent progress towards the electrosynthesis of ammonia from sustainable resources[J]. Catalysis Today,2017,286:57-68. doi: 10.1016/j.cattod.2016.05.008 [80] Kim D, Li O, Saito N. Enhancement of ORR catalytic activity by multiple heteroatom-doped carbon materials[J]. Physical Chemistry Chemical Physics,2015,17:407-413. doi: 10.1039/C4CP03868A [81] Lai L, Potts J, Zhan D, et al. Exploration of the active center structure of nitrogen-doped graphene-based catalysts for oxygen reduction reaction[J]. Energy & Environmental Science,2012,5:7936-7942. [82] Xue L, Liu X, Liu Q, et al. Zigzag carbon as efficient and stable oxygen reduction electrocatalyst for proton exchange membrane fuel cells[J]. Nature Communications,2018,9:3819. doi: 10.1038/s41467-018-06279-x [83] Jia Y, Zhang L, Du A, et al. Defect graphene as a trifunctional catalyst for electrochemical reactions[J]. Advanced Materials,2016,28:9532-9538. doi: 10.1002/adma.201602912 [84] Wang S, Iyyamperumal E, Roy A, et al. Vertically aligned BCN nanotubes as efficient metal-free electrocatalysts for the oxygen reduction reaction: A synergetic effect by co-doping with boron and nitrogen[J]. Angewandte Chemie International Edition,2011,50:11756-11760. doi: 10.1002/anie.201105204 [85] Mazánek V, Matějková S, Sedmidubský D, et al. One-step synthesis of B/N co-doped graphene as highly efficient electrocatalyst for the oxygen reduction reaction: Synergistic effect of impurities[J]. Chemistry-A European Journal,2018,24:928-936. doi: 10.1002/chem.201704515 [86] Jin J, Pan F, Jiang L, et al. Catalyst-free synthesis of crumpled boron and nitrogen Co-doped graphite layers with tunable bond structure for oxygen reduction reaction[J]. ACS Nano,2014,8:3313-3321. doi: 10.1021/nn404927n [87] Hu C, Lin Y, Connell J, et al. Carbon-based metal-free catalysts for energy storage and environmental remediation[J]. Advanced Materials,2019,31:1806128. doi: 10.1002/adma.201806128 [88] Zhao S, Yin H, Du L, et al. Three dimensional N-doped graphene/PtRu nanoparticle hybrids as high performance anode for direct methanol fuel cells[J]. Journal of Materials Chemistry A,2014,2:3719-3724. doi: 10.1039/c3ta14809b [89] Dong L, Hu X, Du Y, et al. Marked enhancement of electrocatalytic activities for gas-consuming reactions by bimodal mesopores[J]. Journal of Materials Chemistry A,2021,9:17821-17829. doi: 10.1039/D1TA05355H [90] Wang S, Yu D, Dai L, et al. Polyelectrolyte-functionalized graphene as metal-free electrocatalysts for oxygen reduction[J]. ACS Nano,2011,5:6202-6209. doi: 10.1021/nn200879h [91] Hu X, Dong L, Ren Y, et al. Surface polarity induced simultaneous enhancement of nanopore accessibility and metal utilization in metal and nitrogen co-doped carbon electrocatalysts for CO2 reduction[J]. ACS Applied Nano Materials,2023,6:9495-9505. doi: 10.1021/acsanm.3c01172 [92] Han Q, Cheng Z, Gao J, et al. Mesh-on-mesh graphitic-C3N4@graphene for highly efficient hydrogen evolution[J]. Advanced Functional Materials,2017,27:1606352. doi: 10.1002/adfm.201606352 [93] Duan J, Chen S, Jaroniec M, et al. Porous C3N4 nanolayers@N-graphene films as catalyst electrodes for highly efficient hydrogen evolution[J]. ACS Nano,2015,9:931-940. doi: 10.1021/nn506701x [94] Li Y, Zhou W, Wang H, et al. An oxygen reduction electrocatalyst based on carbon nanotube-graphene complexes[J]. Nature Nanotechnology,2012,7:394. doi: 10.1038/nnano.2012.72 [95] Campos-Martin J, Blanco-Brieva G, Fierro J. Hydrogen peroxide synthesis: An oputlook beyond the anthraquinone process[J]. Angewandte Chemie International Edition,2006,45:6962-6984. doi: 10.1002/anie.200503779 [96] Sa Y, Kim J, Joo S. Active edge-site-rich carbon nanocatalysts with enhanced electron transfer for efficient electrochemical hydrogen peroxide production[J]. Angewandte Chemie International Edition,2019,58:1100-1105. doi: 10.1002/anie.201812435 [97] Guan M, Dong L, Wu T, et al. Boosting selective oxidation of ethylene to ethylene glycol assisted by in situ generated H2O2 from O2 electroreduction[J]. Angewandte Chemie International Edition,2023,135:e202302466. [98] Iglesias D, Giuliani A, Melchionna M, et al. N-doped graphitized carbon nanohorns as a forefront electrocatalyst in highly selective O2 reduction to H2O2[J]. Chem,2018,4:106-123. doi: 10.1016/j.chempr.2017.10.013 [99] Xia C, Back S, Ringe S, et al. Confined local oxygen gas promotes electrochemical water oxidation to hydrogen peroxide[J]. Nature Catalysis,2020,3:125-134. doi: 10.1038/s41929-019-0402-8 [100] Lin S, Diercks C, Zhang Y, et al. Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water[J]. Science,2015,349:1208-1213. doi: 10.1126/science.aac8343 [101] Zhu D D, Liu J L, Qiao S Z. Recent advances in inorganic heterogeneous electrocatalysts for reduction of carbon dioxide[J]. Advanced Materials,2016,28:3423-3452. doi: 10.1002/adma.201504766 [102] Yang H, Wu Y, Lin Q, et al. Composition tailoring via N and S co-doping and structure tuning by constructing hierarchical pores: metal-free catalysts for high-performance electrochemical reduction of CO2[J]. Angewandte Chemie International Edition,2018,130:15700-15706. [103] Coskun H, Aljabour A, De Luna P, et al. Biofunctionalized conductive polymers enable efficient CO2 electroreduction[J]. Science Advances,2017,3:e1700686. doi: 10.1126/sciadv.1700686 [104] Chen Z, Mou K, Yao S, et al. Highly selective electrochemical reduction of CO2 to formate on metal-free nitrogen-doped PC61BM[J]. Journal of Materials Chemistry A,2018,6:11236-11243. doi: 10.1039/C8TA03328E [105] Zhang T, Li W, Huang K, et al. Regulation of functional groups on graphene quantum dots directs selective CO2 to CH4 conversion[J]. Nature Communications,2021,12:5265. doi: 10.1038/s41467-021-25640-1 [106] Wu J, Ma S, Sun J, et al. A metal-free electrocatalyst for carbon dioxide reduction to multi-carbon hydrocarbons and oxygenates[J]. Nature Communications,2016,7:13869. doi: 10.1038/ncomms13869 [107] Mao X, Hatton T. Recent advances in electrocatalytic reduction of carbon dioxide using metal-free catalysts[J]. Industrial & Engineering Chemistry Research,2015,54:4033-4042. [108] Liu Y, Chen S, Quan X, et al. Efficient electrochemical reduction of Carbon dioxide to acetate on nitrogen-doped nanodiamond[J]. Journal of the American Chemical Society,2015,137:11631-11636. doi: 10.1021/jacs.5b02975 [109] Singh A, Rohr B, Schwalbe J, et al. Electrochemical ammonia synthesis—the selectivity challenge[J]. ACS Catalysis,2017,7:706-709. doi: 10.1021/acscatal.6b03035 [110] Kordali V, Kyriacou G, Lambrou C, et al. Electrochemical synthesis of ammonia at atmospheric pressure and low temperature in a solid polymer electrolyte cell[J]. Chemical Communications,2000,17:1673-1674. [111] Bao D, Zhang Q, Meng F, et al. Electrochemical reduction of N2 under ambient conditions for artificial N2 fixation and renewable energy storage using N2/NH3 cycle[J]. Advanced Materials,2017,29:1604799. doi: 10.1002/adma.201604799 [112] Chen S, Perathoner S, Ampelli C, et al. Electrocatalytic synthesis of ammonia at room temperature and atmospheric pressure from water and nitrogen on a carbon-nanotube-based electrocatalyst[J]. Angewandte Chemie International Edition,2017,129:2743-2747. [113] Qiu W, Xie X, Qiu J, et al. High-performance artificial nitrogen fixation at ambient conditions using a metal-free electrocatalyst[J]. Nature Communications 2018, 9: 3485. [114] Lv C, Qian Y, Yan C, et al. Defect engineering metal-free polymeric carbon nitride electrocatalyst for effective nitrogen fixation under ambient conditions[J]. Angewandte Chemie International Edition,2018,57:10246-10250. doi: 10.1002/anie.201806386 [115] Cheng L, Ma T, Zhang B, et al. Steering the topological defects in amorphous laser-induced graphene for direct nitrate-to-ammonia electroreduction[J]. ACS Catalysis,2022,12:11639-11650. doi: 10.1021/acscatal.2c03219 [116] Huang L, Cheng L, Ma T, et al. Direct synthesis of ammonia from nitrate on amorphous graphene with near 100% efficiency[J]. Advanced Materials,2023,35:2211856. doi: 10.1002/adma.202211856 [117] Janssen L, Hoogland J. Electrolysis of acidic NaCl solution with a graphite anode-I. The graphite electrode[J]. Electrochimica Acta,1969,14:1097-1108. doi: 10.1016/0013-4686(69)80037-X [118] Ji J, Liu J, Shi L, et al. Ruthenium oxide clusters immobilized in cationic vacancies of 2D titanium oxide for chlorine evolution reaction[J]. Small Structure, 2023, 2300240. [119] Wang Y, Liu Y, Wiley D, et al. Recent advances in electrocatalytic chloride oxidation for chlorine gas production[J]. Journal of Materials Chemistry A,2021,9:18974-18993. doi: 10.1039/D1TA02745J [120] Qu H, Li B, Ma Y, et al. Defect-enriched hollow porous carbon nanocages enable highly efficient chlorine evolution reaction[J]. Advanced Materials, 2023, 35: 2301359. [121] Liu X, Kumar P, Chen Q, et al. Carbon nanotubes with fluorine-rich surface as metal-free electrocatalyst for effective synthesis of urea from nitrate and CO2[J]. Applied Catalysis B:Environmental,2022,316:121618. doi: 10.1016/j.apcatb.2022.121618 [122] Chen C, Li S, Zhu X, et al. Balancing sub-reaction activity to boost electrocatalytic urea synthesis using a metal-free electrocatalyst[J]. Carbon Energy,2023,5:e345. doi: 10.1002/cey2.345 [123] Xia R, Wang R, Hasa B, et al. Electrosynthesis of ethylene glycol from C1 feedstocks in a flow electrolyzer[J]. Nature Communications,2023,14:4570. doi: 10.1038/s41467-023-40296-9 -





 下载:
下载: