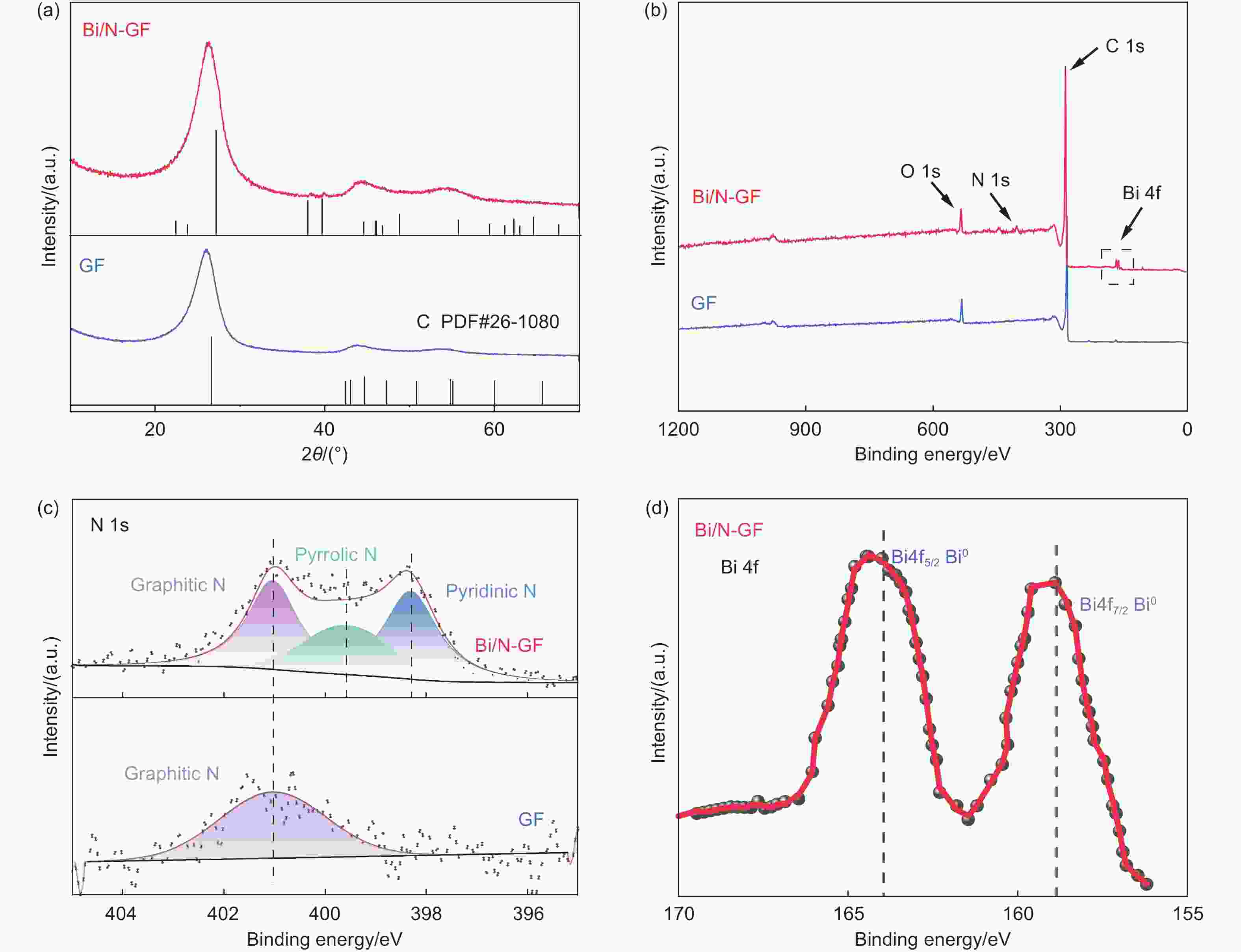
Bismuth nanoparticles anchored on N-doped graphite felts to give stable and efficient iron-chromium redox flow batteries
-
摘要: 铁铬氧化还原液流电池 (ICRFB) 是一种具有成本效益的可规模化储能系统,其利用资源丰富、低成本的铬和铁作为电解液的活性物质。然而,ICRFB存在Cr3+/Cr2+电化学活性低、负极易产生严重的析氢反应 (HER) 等问题。本文报道了一种简单的合成策略,即通过自聚合和湿化学还原方法结合煅烧处理,在氮掺杂石墨毡 (GF) 表面沉积了非晶态铋 (Bi) 纳米颗粒 (NPs),其作为ICRFB的负极材料时可展示出高效的电化学性能。生成的Bi NPs与H+形成中间体,极大地抑制了HER副反应。此外,Bi的引入和GF表面的N掺杂通过协同作用显著提高了Fe2+/Fe3+和Cr3+/Cr2+的电化学活性,降低了电荷传递电阻,提高了反应传质速率。在不同的电流密度下,经25次循环,库仑效率仍高达97.7%。在60.0 mA cm−2电流密度下,能量效率达到85.8%,超过了许多其他报道的材料。循环100次后容量达到862.7 mAh/L,约为GF的5.3倍。Abstract: Iron-chromium redox flow batteries (ICRFBs) use abundant and inexpensive chromium and iron as the active substances in the electrolyte and have great potential as a cost-effective and large-scale energy storage system. However, they are still plagued by several issues, such as the low electrochemical activity of Cr3+/Cr2+ and the occurrence of the undesired hydrogen evolution reaction (HER). We report the synthesis of amorphous bismuth (Bi) nanoparticles (NPs) immobilized on N-doped graphite felts (GFs) by a combined self-polymerization and wet-chemistry reduction strategy followed by annealing, which are used as the negative electrodes for ICRFBs. The resulting Bi NPs react with H+ to form intermediates and greatly inhibit the parasitic HER. In addition, the combined effect of Bi and N dopants on the surface of GF dramatically increases the electrochemical activity of Fe2+/Fe3+ and Cr3+/Cr2+, reduces the charge transfer resistance, and increases the mass transfer rate compared to plain GF. At the optimum Bi/N ratio of 2, a high coulombic efficiency of up to 97.7% is maintained even for 25 cycles at different current densities, the energy efficiency reaches 85.8% at 60.0 mA cm−2, exceeding many other reported materials, and the capacity reaches 862.7 mAh L−1 after 100 cycles, which is about 5.3 times that of bare GF.
-
Key words:
- Iron-chromium flow battery /
- Bi /
- Negative electrode /
- Nitrogen doping /
- Graphite felt
-
Figure 2. STEM characterisation of Bi/N-GF. (a) Low magnification bright field (BF)-STEM image. (b) Magnified view of the indicated area in (a), showing nanoparticle decoration of the graphite flake. (c) High magnification HAADF-STEM image of the particles, showing amorphous aggregation of atoms and some loose single atoms. (d) HAADF-STEM image and (e) fast Fourier transform of a nanoparticle, confirming lack of crystallinity. (f) HAADF-STEM image and (g−i) accompanying EDS mapping, confirming Bi aggregate nanoparticles dispersed on the N-doped graphite felt
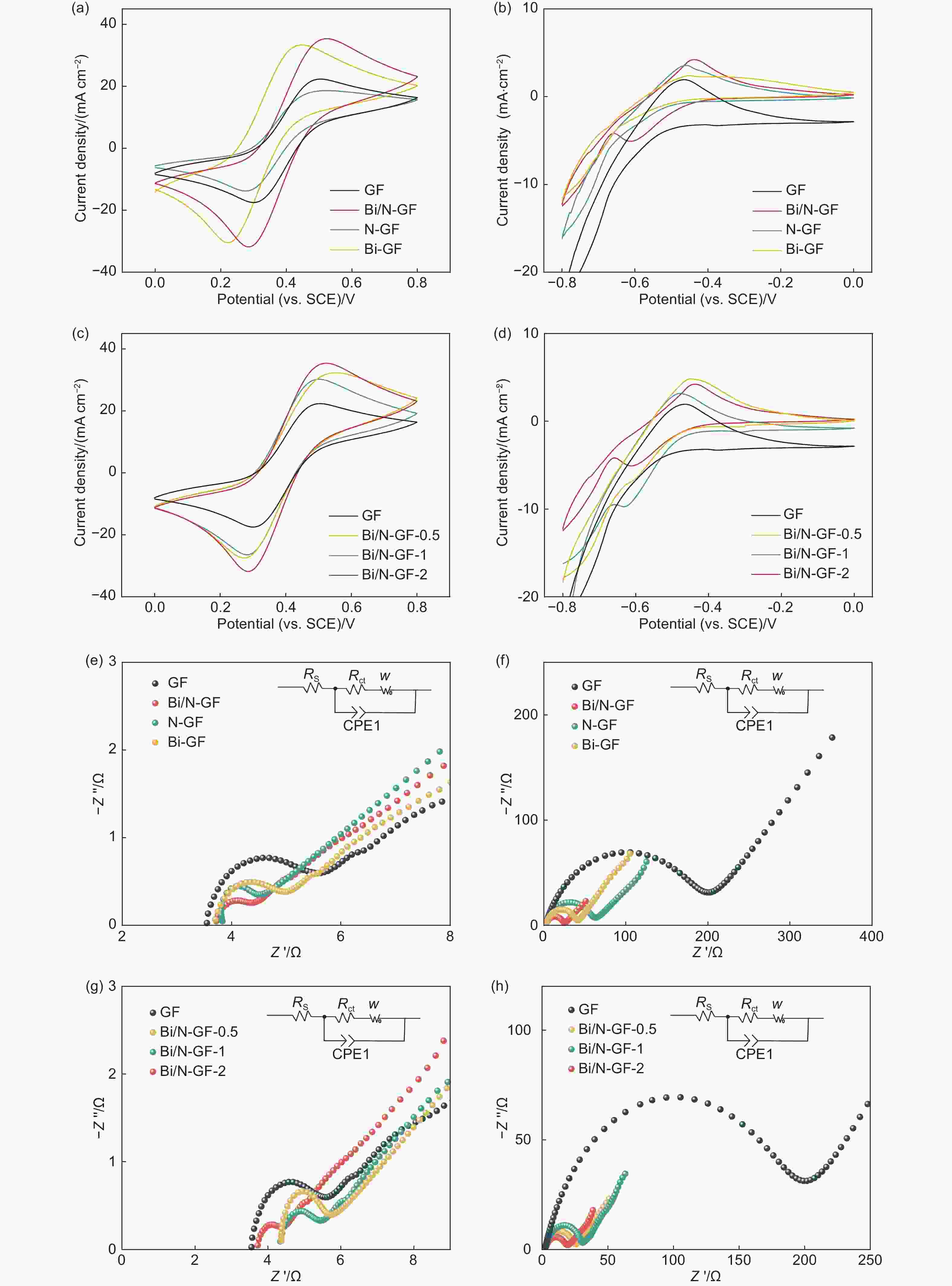
Figure 4. CV curves of GF, N-GF, Bi-GF and Bi/N-GF derived under (a) 40.0 mV s−1 and voltage range of 0−0.8 V (vs. SCE) at the positive electrode and (b) 6.0 mV s−1 and voltage range from −0.8 to 0 V at the negative electrode. CV curves of GF, Bi/N-GF-0.5, Bi/N-GF-1, and Bi/N-GF-2 obtained under (c) 40.0 mV s−1 and voltage range of 0−0.8 V at the positive electrode and (d) 6.0 mV s−1, −0.8−0 V at the negative electrode. EIS diagrams of GF, N-GF, Bi-GF, and Bi/N-GF measured under (e) 0.35 V at the positive electrode and (f) −0.5 V at the negative electrode. EIS diagrams of GF, Bi/N-GF-0.5, Bi/N-GF-1, and Bi/N-GF-2 acquired under (c) 0.35 V at the positive electrode and (d) −0.5 V at the negative electrode
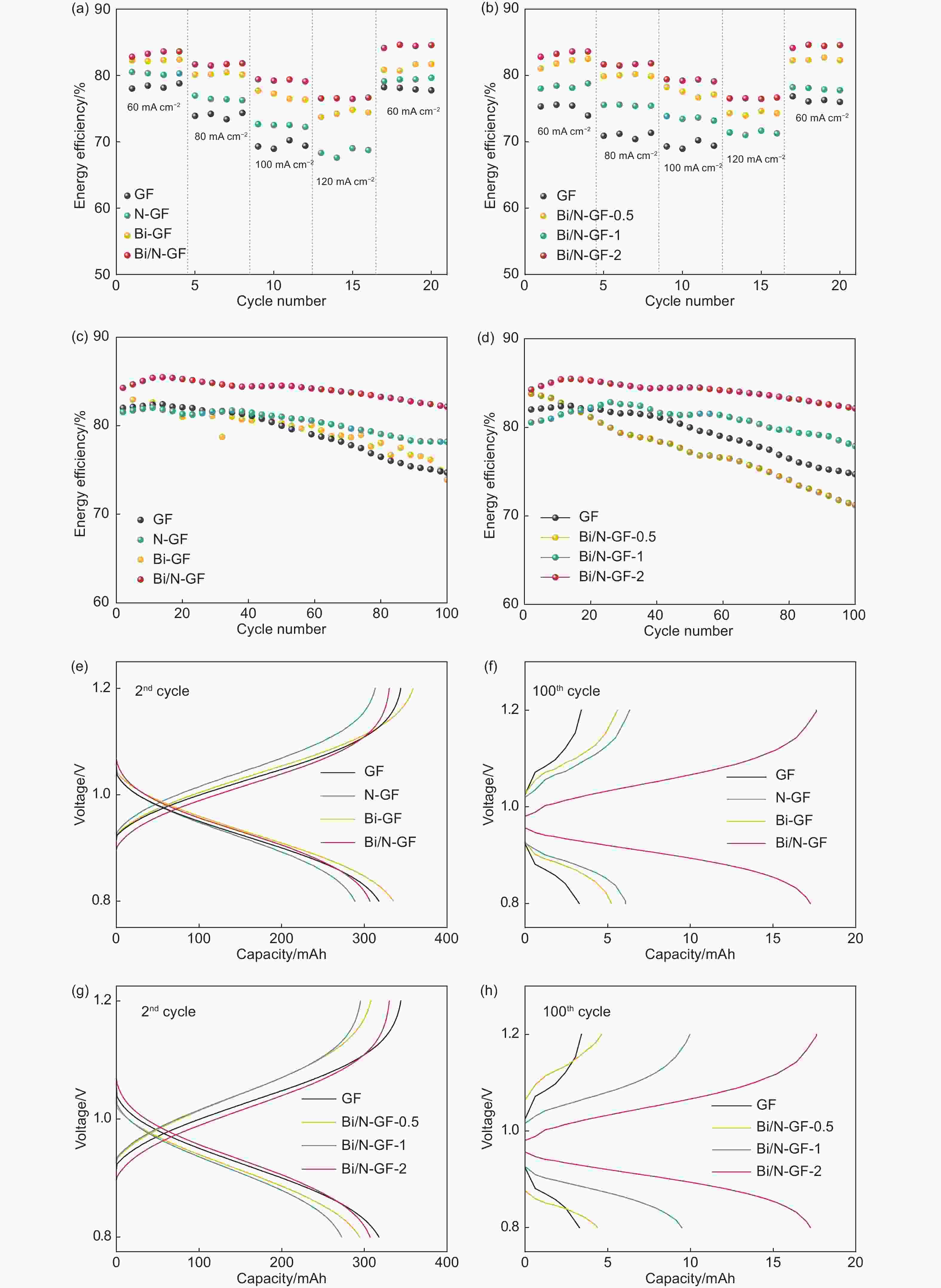
Figure 5. EEs at different current densities in ICRFB for (a) GF, N-GF, Bi-GF and Bi/N-GF, and (b) GF, Bi/N-GF-0.5, Bi/N-GF-1 and Bi/N-GF-2. EEs of (c) GF, N-GF, Bi-GF and Bi/N-GF and (d) GF, Bi/N-GF-0.5, Bi/N-GF-1 and Bi/N-GF-2 tested at 60.0 mA cm−2 and 100 cycles in ICRFB. Charge–discharge curves at the (e) 2nd and (f) 100th cycle in the voltage range of 0.8 to 1.2 V for GF, N-GF, Bi-GF and Bi/N-GF. Charge–discharge curves at the (g) 2nd and (h) 100th cycle in the voltage range of 0.8 to 1.2 V for GF, Bi/N-GF-0.5, Bi/N-GF-1 and Bi/N-GF-2
-
[1] GüR T M. Review of electrical energy storage technologies, materials and systems: Challenges and prospects for large-scale grid storage[J]. Energy Environmental Science,2018,11(10):2696-767. doi: 10.1039/C8EE01419A [2] Zheng Q, Li X F, Cheng Y H, et al. Development and perspective in vanadium flow battery modeling[J]. Applied Energy,2014,132:254-266. doi: 10.1016/j.apenergy.2014.06.077 [3] Li X R, Xiong J, Tang A, et al. Investigation of the use of electrolyte viscosity for online state-of-charge monitoring design in vanadium redox flow battery[J]. Applied Energy,2018,211:1050-1059. doi: 10.1016/j.apenergy.2017.12.009 [4] Zeng Y K, Zhao T S, An L, et al. A comparative study of all−vanadium and iron−chromium redox flow batteries for large−scale energy storage[J]. Journal of Power Sources,2015,300:438-443. doi: 10.1016/j.jpowsour.2015.09.100 [5] Yeonjoo A, Janghyuk M, Seoung E P, et al. High-performance bifunctional electrocatalyst for iron-chromium redox flow batteries[J]. Chemical Engineering Journal,2021,421:127855. doi: 10.1016/j.cej.2020.127855 [6] Wang W, Luo Q T, Li B, et al. Recent progress in redox flow battery research and development[J]. Advanced Functional Materials,2012,23(8):970-986. [7] Chakrabarti M H, Brandon N P, Hajimolana S A, et al. Application of carbon materials in redox flow batteries[J]. Journal of Power Sources,2014,253:150-166. doi: 10.1016/j.jpowsour.2013.12.038 [8] Fic K, Platek A, Piwek J, et al. Sustainable materials for electrochemical capacitors[J]. Materials Today,2018,21(4):437-454. doi: 10.1016/j.mattod.2018.03.005 [9] Ryu J, Park M, Cho J. Catalytic effects of Bi/N-co-doped porous carbon incorporated with ketjenblack nanoparticles for all-vanadium redox flow batteries[J]. Journal of The Electrochemical Society,2016,163(1):A5144. doi: 10.1149/2.0191601jes [10] Wang S L, Xu Z Y, Wu X L, et al. Excellent stability and electrochemical performance of the electrolyte with indium ion for iron-chromium flow battery[J]. Electrochimica Acta,2021,368:137524. doi: 10.1016/j.electacta.2020.137524 [11] Lopez-Atalaya M, Codina G, Perez J R, et al. Optimization studies on a Fe/Cr redox flow battery[J]. Journal of Power Sources,1992,39(2):147-154. doi: 10.1016/0378-7753(92)80133-V [12] Siva R T, Rama K H G. Synthesis, characterization and evaluation of Pb electroplated carbon felts for achieving maximum efficiency of Fe-Cr redox flow cell[J]. Journal of New Materials for Electrochemical Systems,2013,16:287-292. doi: 10.14447/jnmes.v16i4.155 [13] Xie C Y, Yan H, Song Y F, et al. Catalyzing anode Cr2+/Cr3+ redox chemistry with bimetallic electrocatalyst for high-performance iron-chromium flow batteries[J]. Journal of Power Sources,2023,564:232860. doi: 10.1016/j.jpowsour.2023.232860 [14] Yang Z F, Wei Y G, Zeng Y K, et al. Effects of in-situ bismuth catalyst electrodeposition on performance of vanadium redox flow batteries[J]. Journal of Power Sources,2021,506:230238. doi: 10.1016/j.jpowsour.2021.230238 [15] Wu C D, Scherson D A, Calvo E J, et al. A Bismuth-based electrocatalyst for the chromous−chromic couple in acid electrolytes[J]. Journal of the Electrochemical Society,1986,133:2109-2112. doi: 10.1149/1.2108351 [16] Chen N, Zhang H, Luo X D, et al. SiO2-decorated graphite felt electrode by silicic acid etching for iron-chromium redox flow battery[J]. Electrochimica Acta,2020,336:135646. doi: 10.1016/j.electacta.2020.135646 [17] Xiang Y, Daoud W A. Binary NiCoO2-modified graphite felt as an advanced positive electrode for vanadium redox flow batteries[J]. Journal of Materials Chemistry A,2019,7(10):5589-5600. doi: 10.1039/C8TA09650C [18] Ren H L, Su Y, Zhao S, et al. Research on the performance of cobalt oxide decorated graphite felt as electrode of iron-chromium flow battery[J]. ChemElectroChem,2023,10(5):e202201146. doi: 10.1002/celc.202201146 [19] Ji Y, Li J L, Li S F Y. Synergistic effect of the bifunctional polydopamine-Mn3O4 composite electrocatalyst for vanadium redox flow batteries[J]. Journal of Materials Chemistry A,2017,5(29):15154-15166. doi: 10.1039/C7TA03922K [20] Zhang K Y, Yan C W, Tang A. Interfacial co-polymerization derived nitrogen-doped carbon enables high-performance carbon felt for vanadium flow batteries[J]. Journal of Materials Chemistry A,2021,9(32):17300-17310. doi: 10.1039/D1TA03683A [21] Li B, Gu M, Nie Z M, et al. Bismuth nanoparticle decorating graphite felt as a high-performance electrode for an all-vanadium redox flow battery[J]. Nano Letters,2013,13(3):1330-1335. doi: 10.1021/nl400223v [22] Grubaˇc Z, Metikoˇs-Hukovi ́c M. Kinetics and mechanism of electrocrystallization of bismuth in oxide matrix[J]. Electrochim. Acta,1999,44(25):4559-4571. doi: 10.1016/S0013-4686(99)00174-7 [23] Zhang H, Tan Y, Luo X D, et al. Polarization effects of a rayon and polyacrylonitrile based graphite felt for iron-chromium redox flow batteries[J]. ChemElectroChem,2019,6(12):3175-3188. doi: 10.1002/celc.201900518 [24] Sun C Y, Zhang H, Luo X D, et al. A comparative study of Nafion and sulfonated poly(ether ether ketone) membrane performance for iron-chromium redox flow battery[J]. Advanced Materials Interfaces,2020,7(20):2000855. doi: 10.1002/admi.202000855 -
 0261.pdf
0261.pdf

-






 下载:
下载: