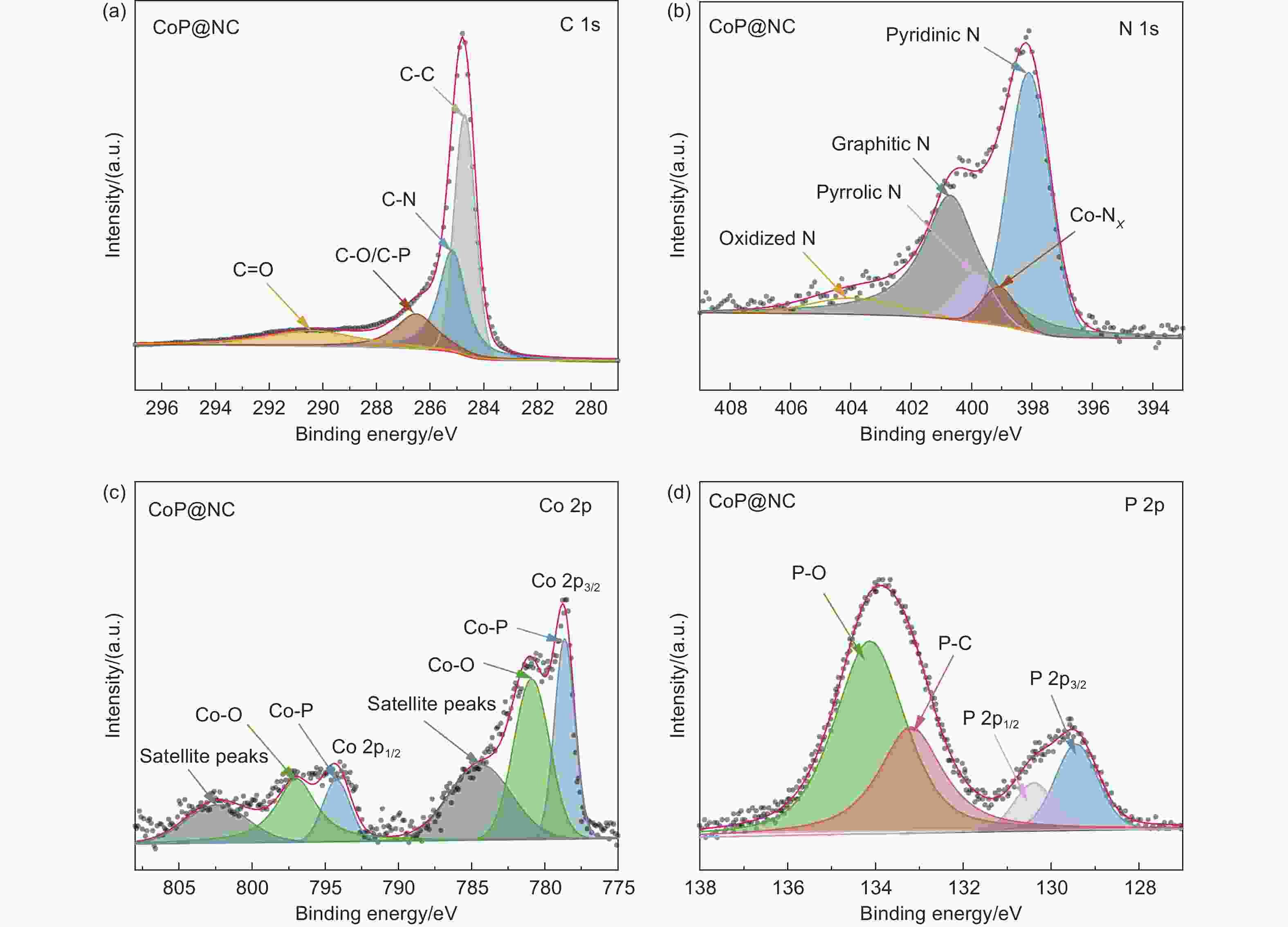
Controllable construction of CoP nanoparticles anchored on a nitrogen-doped porous carbon as an electrocatalyst for highly efficient oxygen reduction in Zn-air batteries
-
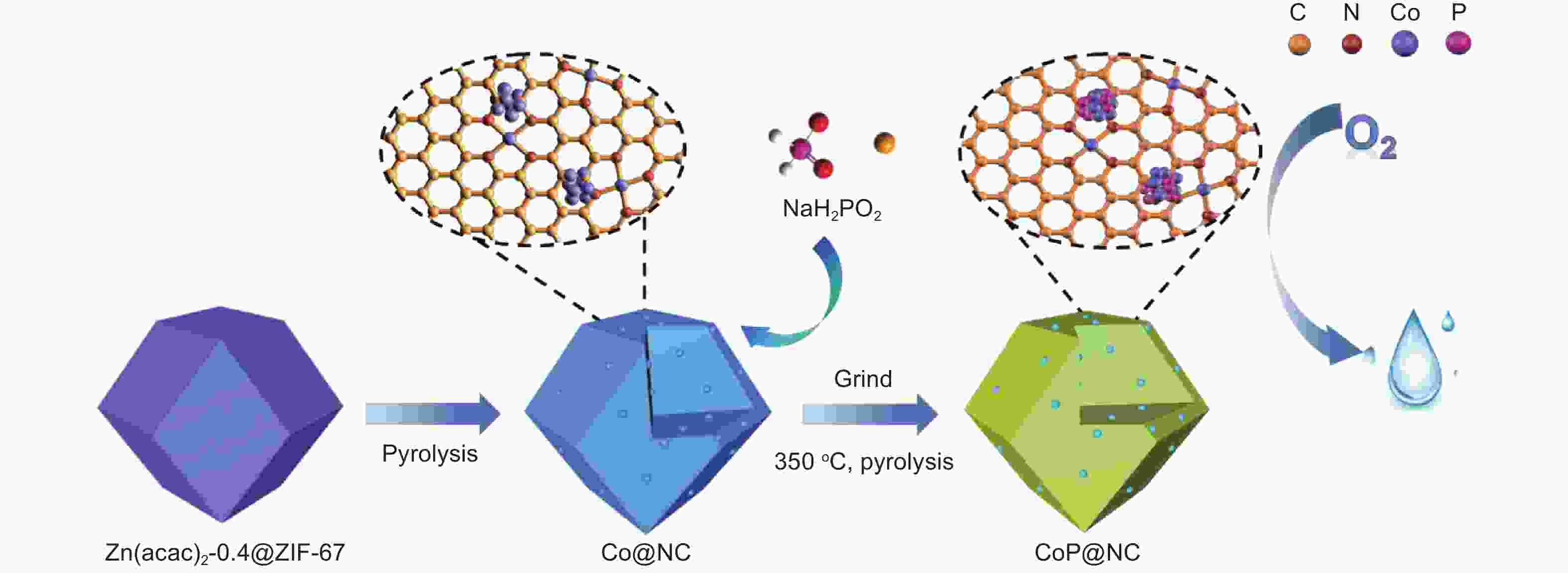
摘要: 为可持续能源设备中的氧还原反应(ORR)探索具有成本效益和高效率的无贵金属催化剂仍然是一项巨大的挑战。在掺杂杂原子的碳上支撑的过渡金属磷化物(TMPs)因其可调的电子结构和更高的催化性能,具有替代贵金属的潜力。本文采用磷化策略(350 °C下二次热解),在分层多孔碳框架(CoP@NC)上构建由掺氮碳壳包裹的CoP纳米颗粒。在二次热解过程中,Co纳米颗粒在NaH2PO2生成的PH3气体下原位转化为CoP纳米颗粒,而载体的十二面体结构没有发生改变。在碱性条件下,CoP@NC电催化剂显示出显著的ORR活性,半波电位高达0.92 V,这归功于分散良好的CoP纳米颗粒、掺氮碳壳之间的协同耦合以及通过多孔结构的高效质量传输。此外,使用CoP@NC电催化剂的锌-空气电池显示1.51 V的高开路电压和210.1 mW cm−2的功率密度。这项研究为开发具有优异ORR性能的低成本催化剂提供了一种新策略,并为燃料电池和金属空气电池的实际应用提供了机会。Abstract: Exploring cost-efficient and high-efficient noble metal-free catalysts for oxygen reduction reactions (ORRs) involved in sustainable energy devices still remains a great challenge. Transition-metal phosphides supported on heteroatom-doped carbons have presented a potential as alternative candidates of precious metals due to their tunable electronic structures and boosted catalytic performance. Herein, phosphating was adopted to construct CoP nanoparticles (NPs) anchored on a nitrogen-doped porous carbon framework (CoP@NC) from Co NPs loaded on NC using PH3 gas released from NaH2PO2 during heat treatment. The dodecahedral structure of Co NPs is retained in their transformation to CoP NPs. The CoP@NC electrocatalyst shows remarkable ORR activity with a half-wave potential up to 0.92 V under alkaline conditions, which is attributed to the synergistic coupling between the well dispersed CoP nanoparticles on the nitrogen-doped carbon support and the efficient mass transport in the porous structure. Zinc-air batteries assembled with the CoP@NC electrocatalyst as an cathode displays a high open-circuit voltage of 1.51 V and power density of 210.1 mW cm−2. This work provides a novel strategy to develop low-cost catalysts with excellent ORR performance to promote their practical application in metal-air batteries.
-
Key words:
- Electrocatalysts /
- Co-based catalysts /
- Metal phosphides /
- ORR /
- Zinc-air battery
-
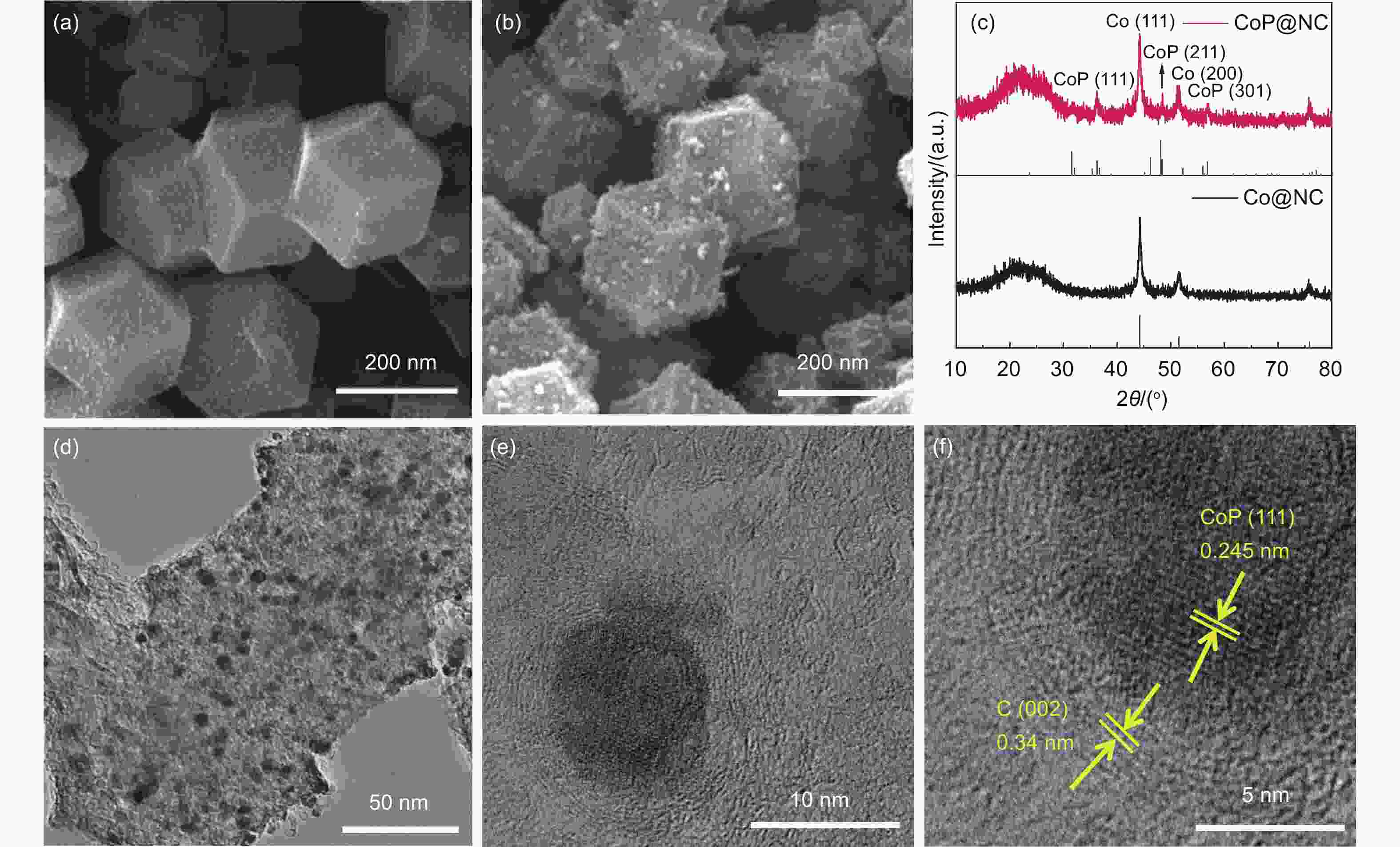
Figure 1. SEM micrographs of (a) Co@NC[37] and (b) CoP@NC electrocatalysts. (c) Corresponding XRD patterns. (d-f) TEM images of CoP@NC catalyst
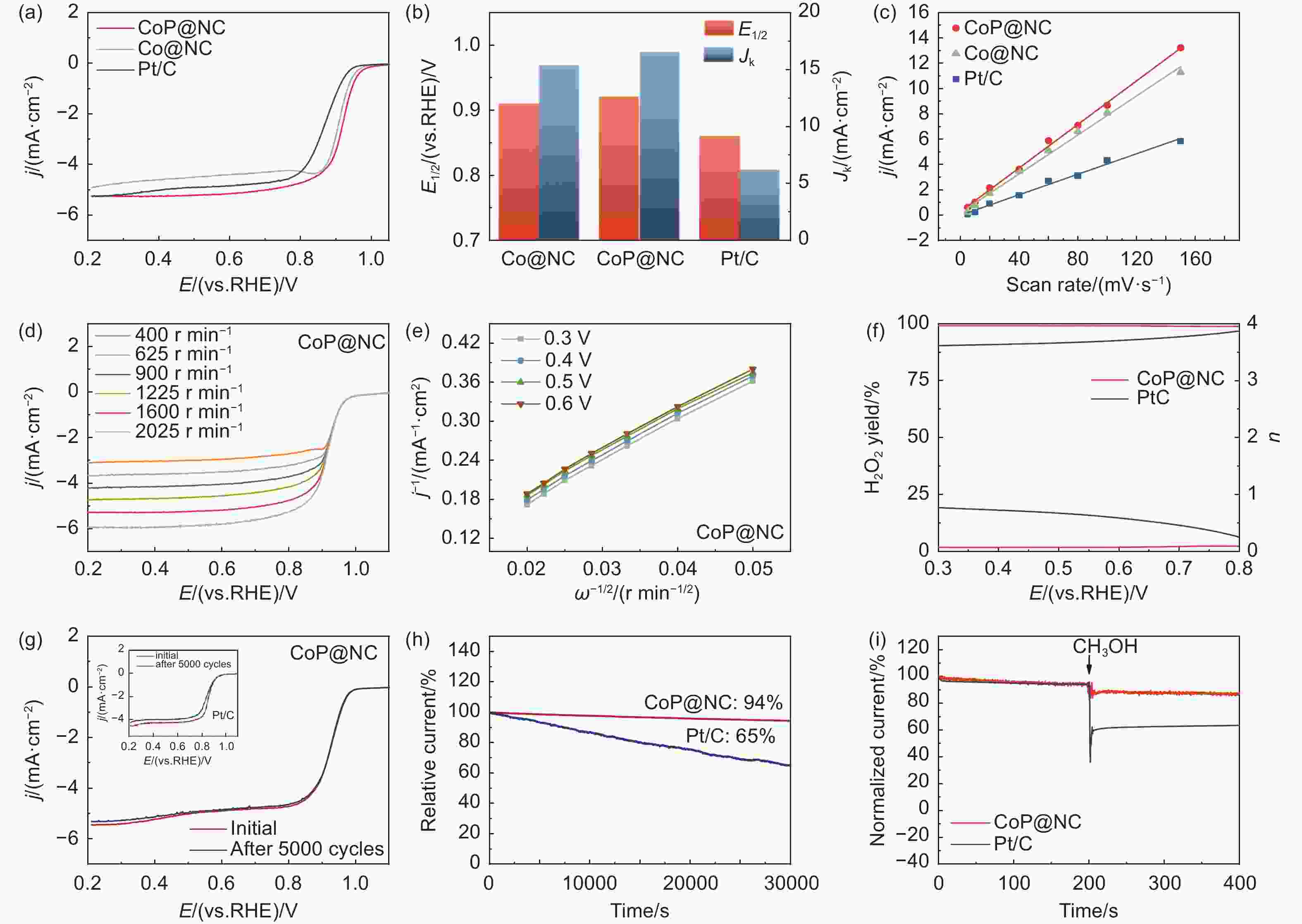
Figure 3. (a) The LSV curves of CoP@NC, Co@NC and Pt/C catalysts. (b) E1/2 and Jk (0.85 V) of different catalysts. (c) Cdl values of each catalyst versus the scanning speed, and the results are drawn as shown. (d) The LSV curves of CoP@NC obtained at rotational speeds of 400-2025 r min−1. (e) K-L plots of CoP@NC. (f) The electron transfer number (n) and the H2O2 selectivity of the CoP@NC and Pt/C catalysts measured according to RRDE. (g) LSV curves before and after 5000 potentiostatic cycles of CoP@NC and Pt/C. (h) chronoamperometric tests of CoP@NC and Pt/C catalys at 0.60 V (vs. RHE). (i) Methanol antitoxicity testing of CoP@NC and Pt/C catalysts.
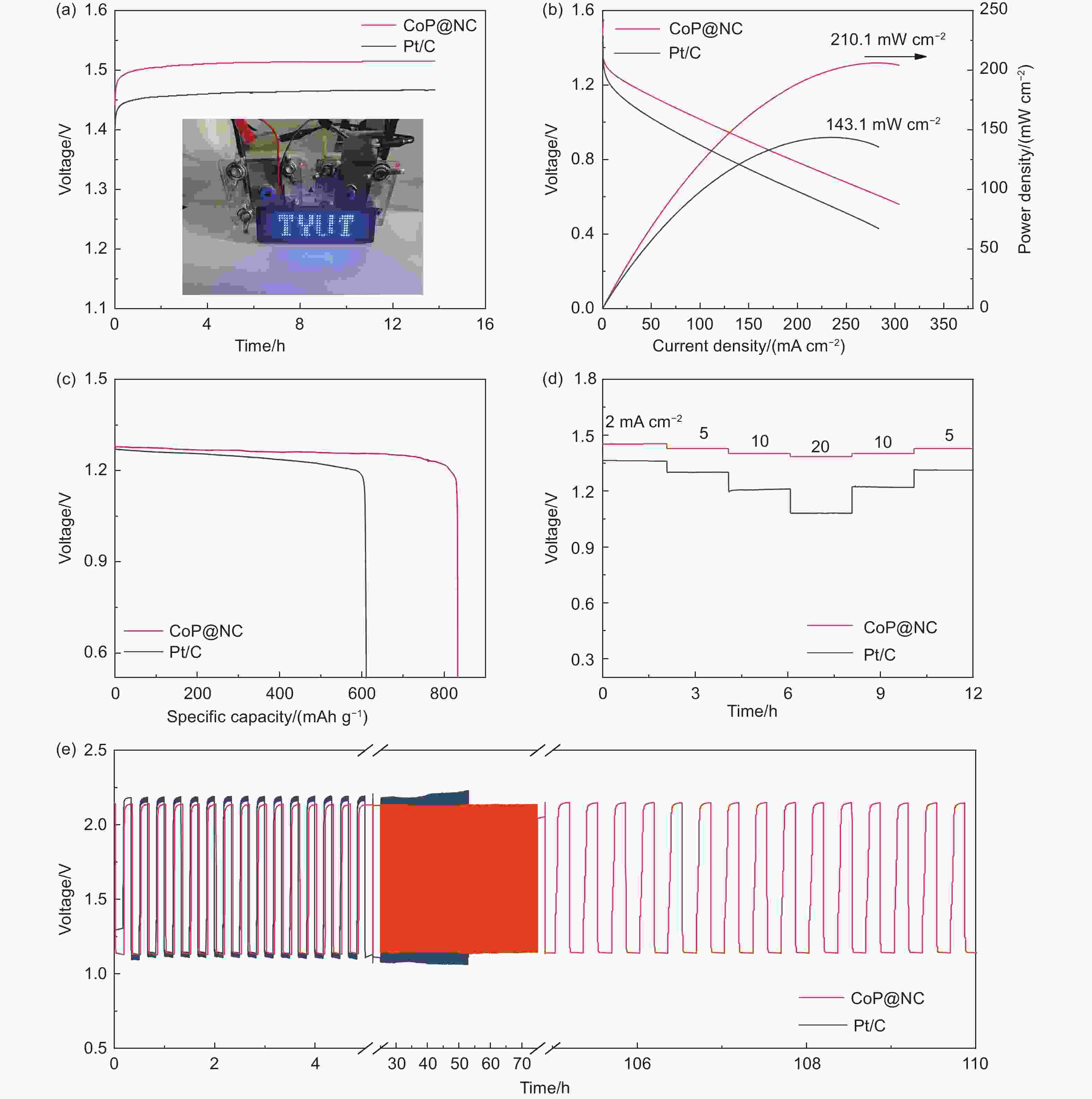
Figure 4. (a) Open-circuit voltage profile of ZABs under alkaline solution assembled using CoP@NC and Pt/C catalysts as air cathode materials. (Inset) Photograph of connecting two ZABs lighting up a LED. (b) Polarization curves and power density curves of the of ZABs of the CoP@NC and Pt/C catalysts. (c) Discharging capacity curves normalized by the consumed Zn of CoP@NC and Pt/C . (d) Rate performance of ZABs with CoP@NC and Pt/C (20%) as air cathode at 2-20 mA cm−2, respectively. (e) Charge-discharge cycle curves of ZABs using CoP@NC and Pt/C catalysts.
-
[1] Wang Q, Liang H, Zhou J, et al. Boosting oxygen reduction catalysis by introducing Fe bridging atoms between Pt nanoparticles and N-doped graphene[J]. Chemical Engineering Journal,2023,467:143482. doi: 10.1016/j.cej.2023.143482 [2] Zhang Y, Zhang H, Sha W, et al. N-doped graphene nanoribbons intertwined on 3D graphene skeleton as superior metal-free electrocatalyst for oxygen reduction[J]. Colloids and Surfaces A:Physicochemical and Engineering Aspects,2022,652:129832. doi: 10.1016/j.colsurfa.2022.129832 [3] An F, Bao X Q, Deng X Y, et al. Carbon-based metal-free oxygen reduction reaction electrocatalysts: past, present and future[J]. New Carbon Materials,2022,37(2):338-354. doi: 10.1016/S1872-5805(22)60590-0 [4] Shi J, Lin N, Lin H B, et al. A N-doped rice husk-based porous carbon as an electrocatalyst for the oxygen reduction reaction[J]. New Carbon Materials,2020,35(4):401-409. doi: 10.1016/S1872-5805(20)60497-8 [5] Li H, Zhang H X, Yan X L, et al. Carbon-supported metal single atom catalysts[J]. New Carbon Materials,2018,33(1):1-11. doi: 10.1016/S1872-5805(18)60322-1 [6] Song Y, Zhang T, Zhou G, et al. Cu nanoclusters on N-doped carbon nanotubes as efficient electrocatalyst for oxygen reduction reaction[J]. Applied Surface Science,2022,589:153022. doi: 10.1016/j.apsusc.2022.153022 [7] Li P, Wang H L. Recent advances in carbon-supported iron group electrocatalysts for the oxygen reduction reaction[J]. New Carbon Materials,2021,36(4):665-682. doi: 10.1016/S1872-5805(21)60072-0 [8] Ren X P, Hu Q W, Ling F, et al. Mott-Schottky heterojunction formation between Co and MoSe2 on carbon nanotubes for superior hydrogen evolution[J]. New Carbon Materials,2023,38(6):1059-1069. doi: 10.1016/S1872-5805(23)60782-6 [9] Guo J, Mao Z, Yan X, et al. Ultrasmall tungsten carbide catalysts stabilized in graphitic layers for high-performance oxygen reduction reaction[J]. Nano Energy,2016,28:261-268. doi: 10.1016/j.nanoen.2016.08.045 [10] Tu H, Zhang H, Song Y, et al. Electronic asymmetry engineering of Fe―N―C electrocatalyst via adjacent carbon vacancy for boosting oxygen reduction reaction[J]. Advanced Science,2023,10(32):2305194. doi: 10.1002/advs.202305194 [11] Wang X R, Liu J Y, Liu Z W, et al. Identifying the key role of pyridinic-N―Co bonding in synergistic electrocatalysis for reversible ORR/OER[J]. Advanced Materials,2018,30(23):1800005. doi: 10.1002/adma.201800005 [12] Xu X X, Zhang N C, Wang J Y, et al. The synthesis of iron-nitrogen sites embedded in electrospun carbon nanofibers with an excellent oxygen reduction reaction activity in alkaline/acidic media[J]. New Carbon Materials,2023,38(1):154-160. doi: 10.1016/S1872-5805(22)60649-8 [13] Zhang Y T, Li S Y, Zhang N N, et al. A carbon catalyst doped with Co and N derived from the metal-organic framework hybrid (ZIF-8@ZIF-67) for efficient oxygen reduction reaction[J]. New Carbon Materials,2023,38(1):200-209. doi: 10.1016/S1872-5805(22)60609-7 [14] Zhang H, Zhang Z, Zhang Z, et al. Highly dispersed ultrasmall iron phthalocyanine molecule clusters confined by mesopore-rich N-doped hollow carbon nanospheres for efficient oxygen reduction reaction and Zn-air battery[J]. Chemical Engineering Journal,2023,469:143996. doi: 10.1016/j.cej.2023.143996 [15] Liu W, Hu E, Jiang H, et al. A highly active and stable hydrogen evolution catalyst based on pyrite-structured cobalt phosphosulfide[J]. Nature Communications,2016,7(1):10771. doi: 10.1038/ncomms10771 [16] He X F, Chang L B, Han P F, et al. Highly efficient Co−N−C electrocatalysts with a porous structure for the oxygen reduction reaction[J]. New Carbon Materials,2023,38(5):976-988. doi: 10.1016/S1872-5805(23)60735-8 [17] Zhang J, Song L, Zhao C, et al. Co, N co-doped porous carbons as high-performance oxygen reduction electrocatalysts[J]. New Carbon Materials,2021,36(1):209-218. doi: 10.1016/S1872-5805(21)60016-1 [18] Tang C, Titirici M M, Zhang Q. A review of nanocarbons in energy electrocatalysis: Multifunctional substrates and highly active sites[J]. Journal of Energy Chemistry,2017,26(6):1077-1093. doi: 10.1016/j.jechem.2017.08.008 [19] Li X, Jiang Q, Dou S, et al. ZIF-67-derived Co-NC@CoP-NC nanopolyhedra as an efficient bifunctional oxygen electrocatalyst[J]. Journal of Materials Chemistry A,2016,4(41):15836-15840. doi: 10.1039/C6TA06434E [20] Zhang D, Ding R, Tang Y, et al. Stable Co/N-doped carbon nanotubes as catalysts for oxygen reduction[J]. ACS Applied Nano Materials,2022,5(7):10026-10035. doi: 10.1021/acsanm.2c02453 [21] Li M, Shi J, Xu B, et al. Size-controlled Co/CoO heterogeneous nanoparticles confined in N-doped mesoporous carbon for efficient oxygen reduction in zinc-air batteries[J]. Journal of Colloid and Interface Science,2024,653:1317-1325. doi: 10.1016/j.jcis.2023.09.176 [22] Liu H, Liu Z H, Zhang J Q, et al. Boron and nitrogen co-doped carbon dots for boosting electrocatalytic oxygen reduction[J]. New Carbon Materials,2021,36(3):585-593. doi: 10.1016/S1872-5805(21)60043-4 [23] Ye X W, Hu L B, Liu M C, et al. Improved oxygen reduction performance of a N, S co-doped graphene-like carbon prepared by a simple carbon bath method[J]. New Carbon Materials,2020,35(5):531-539. doi: 10.1016/S1872-5805(20)60506-6 [24] Rao P, Wu D, Wang T J, et al. Single atomic cobalt electrocatalyst for efficient oxygen reduction reaction[J]. eScience,2022,2(4):399-404. doi: 10.1016/j.esci.2022.05.004 [25] Shi Q, Liu Q, Ma Y, et al. High-performance trifunctional electrocatalysts based on FeCo/Co2P Hybrid Nanoparticles for Zinc–air battery and self-powered overall water splitting[J]. Advanced Energy Materials,2020,10(10):1903854. doi: 10.1002/aenm.201903854 [26] Zhu C, Zhao S, Fan Z, et al. Confinement of CoP nanoparticles in nitrogen-doped yolk-shell porous carbon polyhedron for ultrafast catalytic oxidation[J]. Advanced Functional Materials,2020,30(49):2003947. doi: 10.1002/adfm.202003947 [27] Li Y, Dong Z, Jiao L. Multifunctional transition metal-based phosphides in energy-related electrocatalysis[J]. Advanced Energy Materials,2020,10(11):1902104. doi: 10.1002/aenm.201902104 [28] Tang C, Wang B, Wang H F, et al. Defect engineering toward atomic Co―Nx―C in hierarchical graphene for rechargeable flexible solid Zn-air batteries[J]. Advanced Materials,2017,29(37):1703185. doi: 10.1002/adma.201703185 [29] Liu J, Guo Y, Fu X Z, et al. Strengthening absorption ability of Co–N–C as efficient bifunctional oxygen catalyst by modulating the d band center using MoC[J]. Green Energy & Environment,2023,8(2):459-469. [30] Lin Y, Yang L, Zhang Y, et al. Defective carbon–CoP nanoparticles hybrids with interfacial charges polarization for efficient bifunctional oxygen electrocatalysis[J]. Advanced Energy Materials,2018,8(18):1703623. doi: 10.1002/aenm.201703623 [31] Liu J, Zhang C, Yuan S, et al. CoP-decorated N, P-doped necklace-like carbon for highly efficient oxygen reduction and Al-air batteries[J]. Chemical Engineering Journal,2022,428:131326. doi: 10.1016/j.cej.2021.131326 [32] Li J, Liu G, Liu B, et al. An extremely facile route to Co2P encased in N, P-codoped carbon layers: Highly efficient bifunctional electrocatalysts for ORR and OER[J]. International Journal of Hydrogen Energy,2018,43(3):1365-1374. doi: 10.1016/j.ijhydene.2017.11.102 [33] Wang Q, Fan Y, Wang K, et al. Hierarchical tubular structures composed of CoPx and carbon nanotubes: Highly effective electrocatalyst for oxygen reduction[J]. Carbon,2018,130:241-249. doi: 10.1016/j.carbon.2018.01.005 [34] Huang X, Xu X, Li C, et al. Vertical CoP nanoarray wrapped by N, P-doped carbon for hydrogen evolution reaction in both acidic and alkaline conditions[J]. Advanced Energy Materials,2019,9(22):1803970. doi: 10.1002/aenm.201803970 [35] Akula S, Mooste M, Kozlova J, et al. Transition metal (Fe, Co, Mn, Cu) containing nitrogen-doped porous carbon as efficient oxygen reduction electrocatalysts for anion exchange membrane fuel cells[J]. Chemical Engineering Journal,2023,458:141468. doi: 10.1016/j.cej.2023.141468 [36] Li H, Li Q, Wen P, et al. Retracted: Colloidal cobalt phosphide nanocrystals as trifunctional electrocatalysts for overall water splitting powered by a Zinc-air battery[J]. Advanced Materials,2018,30(9):1705796. doi: 10.1002/adma.201705796 [37] Zhou G, Yan X, Zhang T, et al. MOFs-derived hierarchical porous carbon supported Co@NC nanocapsules for pH universal oxygen reduction reaction and Zn-air batteries[J]. Applied Surface Science,2023,621:156906. doi: 10.1016/j.apsusc.2023.156906 [38] Wang Y, Wu M, Li J, et al. In situ growth of CoP nanoparticles anchored on (N, P) co-doped porous carbon engineered by MOFs as advanced bifunctional oxygen catalyst for rechargeable Zn-air battery[J]. Journal of Materials Chemistry A,2020,8(36):19043-19049. doi: 10.1039/D0TA06435A [39] Fu G, Liu Y, Chen Y, et al. Robust N-doped carbon aerogels strongly coupled with iron–cobalt particles as efficient bifunctional catalysts for rechargeable Zn-air batteries[J]. Nanoscale,2018,10(42):19937-19944. doi: 10.1039/C8NR05812A [40] Liu W, Zhou Z, Li Z, et al. Cobalt phosphide embedded N-doped carbon nanopolyhedral as an efficient cathode electrocatalyst in microbial fuel cells[J]. Journal of Environmental Chemical Engineering,2021,9(1):104582. doi: 10.1016/j.jece.2020.104582 [41] Yang T, Wang Z, Li K, et al. Surface-oxidized cobalt phosphide used as high efficient electrocatalyst in activated carbon air-cathode microbial fuel cell[J]. Journal of Power Sources,2017,363:87-94. doi: 10.1016/j.jpowsour.2017.06.088 [42] Chen T, Ma J, Chen S, et al. Construction of heterostructured CoP/CN/Ni: Electron redistribution towards effective hydrogen generation and oxygen reduction[J]. Chemical Engineering Journal,2021,415:129031. doi: 10.1016/j.cej.2021.129031 [43] Chen J, Huang J, Wang H, et al. Phase-mediated cobalt phosphide with unique core-shell architecture serving as efficient and bifunctional electrocatalyst for hydrogen evolution and oxygen reduction reaction[J]. Chinese Chemical Letters,2022,33(8):3752-3756. doi: 10.1016/j.cclet.2021.11.063 [44] Liu B, Wang R, Yao Y, et al. Hollow-structured CoP nanotubes wrapped by N-doped carbon layer with interfacial charges polarization for efficiently boosting oxygen reduction/evolution reactions[J]. Chemical Engineering Journal,2022,431:133238. doi: 10.1016/j.cej.2021.133238 [45] Go H W, Nguyen T T, Ngo Q P, et al. Tailored heterojunction active sites for oxygen electrocatalyst promotion in Zinc-air batteries[J]. Small,2023,19(10):2206341. doi: 10.1002/smll.202206341 [46] Li M, Pan X, Jiang M, et al. Interface engineering of oxygen-vacancy-rich CoP/CeO2 heterostructure boosts oxygen evolution reaction[J]. Chemical Engineering Journal,2020,395:125160. doi: 10.1016/j.cej.2020.125160 -





 下载:
下载: